Abstract
Purpose
To investigate the intra-examination agreement between multi-echo gradient echo (MEGE) and confounder-corrected chemical shift-encoded (CSE) sequences for liver T2*/R2* estimations in a wide range of T2*/R2* and proton density fat fraction (PDFF) values. Exploratorily, to search for the T2*/R2* value where the agreement line breaks and examine differences between regions of low and high agreement.
Methods
Consecutive patients at risk for liver iron overload who underwent MEGE and CSE sequences within the same exam at 1.5 T were retrospectively selected. Regions of interest were drawn in the right and one in the left liver lobes on post-processed images for R2*(sec−1) and PDFF (%) estimation. Agreement between MEGE-R2* and CSE-R2* was evaluated using intra-class correlation coefficient (ICC) and Bland–Altman analysis. 95% confidence intervals (CI) were computed. Segment-and-regression analysis was performed to find the point where the agreement between sequences is interrupted. Regions of low and high agreement were examined using tree-based partitioning analyses.
Results
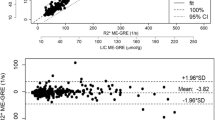
49 patients were included. Mean MEGE-R2* was 94.2 s−1 (range: 31.0–737.1) and mean CSE-R2* 87.7 (29.7–748.1). Mean CSE-PDFF was 9.12% (0.1–43.3). Agreement was strong for R2* estimations (ICC: 0.992,95%CI 0.987,0.996), but the relation was nonlinear and possibly heteroskedastic. Lower agreement occurred when MEGE-R2* > 235 s−1, with MEGE-R2* values consistently lower than CSE-R2*. Higher agreement was observed when PDFF < 14%.
Conclusion
MEGE-R2* and CSE-R2* strongly agree, though at higher iron content, MEGE-R2* is consistently lower than CSE-R2*. In this preliminary dataset, a breaking point for agreement was found at R2* > 235. Lower agreement was observed in patients with moderate to severe liver steatosis.





Similar content being viewed by others
Abbreviations
- MEGE:
-
Multi-echo gradient echo
- CSE:
-
Confounder-corrected chemical shift encoded
- PDFF:
-
Proton density fat fraction
References
Pietrangelo A. Iron and the liver. Liver Int. 2016 Jan;36 Suppl 1:116-23. doi: https://doi.org/10.1111/liv.13020.
Brittenham GM, Badman DG; National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK)Workshop. Noninvasive measurement of iron: report of an NIDDK workshop. Blood. 2003 Jan 1;101(1):15–9. doi: https://doi.org/10.1182/blood-2002-06-1723. Epub 2002 Aug 29. PMID: 12393526.
Labranche R, Gilbert G, Cerny M, Vu KN, Soulières D, Olivié D, et al. Liver Iron Quantification with MR Imaging: A Primer for Radiologists. Radiographics. 2018 Mar-Apr;38(2):392–412. doi: https://doi.org/10.1148/rg.2018170079.
Henninger B, Plaikner M, Zoller H, Viveiros A, Kannengiesser S, Jaschke W, Kremser C. Performance of different Dixon-based methods for MR liver iron assessment in comparison to a biopsy-validated R2* relaxometry method. Eur Radiol. 2021 Apr;31(4):2252-2262. doi: https://doi.org/10.1007/s00330-020-07291-w.
Bhimaniya S, Arora J, Sharma P, Zhang Z, Khanna G. Liver iron quantification in children and young adults: comparison of a volumetric multi-echo 3-D Dixon sequence with conventional 2-D T2* relaxometry. Pediatr Radiol. 2022 Jul;52(8):1476-1483. doi: https://doi.org/10.1007/s00247-022-05352-4. Epub 2022 Apr 6. PMID: 35384483.
Hernando D, Cook RJ, Qazi N, Longhurst CA, Diamond CA, Reeder SB. Complex confounder-corrected R2* mapping for liver iron quantification with MRI. Eur Radiol. 2021 Jan;31(1):264-275. doi: https://doi.org/10.1007/s00330-020-07123-x.
Cunha GM, Thai TT, Hamilton G, Covarrubias Y, Schlein A, Middleton MS, et al. Accuracy of common proton density fat fraction thresholds for magnitude- and complex-based chemical shift-encoded MRI for assessing hepatic steatosis in patients with obesity. Abdom Radiol (NY). 2020 Mar;45(3):661-671. doi: https://doi.org/10.1007/s00261-019-02350-3.
Landis JR, Koch GG. The measurement of observer agreement for categorical data. Biometrics. 1977 Mar;33(1):159-74.
Muggeo VM. Estimating regression models with unknown break-points. Stat Med. 2003 Oct 15;22(19):3055-71. doi: https://doi.org/10.1002/sim.1545.
Riazi K, Azhari H, Charette JH, Underwood FE, King JA, Afshar EE, Swain MG, Congly SE, Kaplan GG, Shaheen AA. The prevalence and incidence of NAFLD worldwide: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol. 2022 Sep;7(9):851-861. doi: https://doi.org/10.1016/S2468-1253(22)00165-0.
Henninger B, Zoller H, Kannengiesser S, Zhong X, Jaschke W, Kremser C. 3D Multiecho Dixon for the Evaluation of Hepatic Iron and Fat in a Clinical Setting. J Magn Reson Imaging. 2017 Sep;46(3):793-800. doi: https://doi.org/10.1002/jmri.25630.
Serai SD, Smith EA, Trout AT, Dillman JR. Agreement between manual relaxometry and semi-automated scanner-based multi-echo Dixon technique for measuring liver T2* in a pediatric and young adult population. Pediatr Radiol. 2018 Jan;48(1):94-100. doi: https://doi.org/10.1007/s00247-017-3990-y.
Colgan TJ, Zhao R, Roberts NT, Hernando D, Reeder SB. Limits of Fat Quantification in the Presence of Iron Overload. J Magn Reson Imaging. 2021 Oct;54(4):1166-1174. doi: https://doi.org/10.1002/jmri.27611.
Hamer OW, Aguirre DA, Casola G, Lavine JE, Woenckhaus M, Sirlin CB. Fatty liver: imaging patterns and pitfalls. Radiographics. 2006 Nov-Dec;26(6):1637–53. doi: https://doi.org/10.1148/rg.266065004. PMID: 17102041.
Henninger B, Alustiza J, Garbowski M, Gandon Y. Practical guide to quantification of hepatic iron with MRI. Eur Radiol. 2020 Jan;30(1):383-393. doi: https://doi.org/10.1007/s00330-019-06380-9.
Krafft AJ, Loeffler RB, Song R, Bian X, McCarville MB, Hankins JS, Hillenbrand CM. Does fat suppression via chemically selective saturation affect R2*-MRI for transfusional iron overload assessment? A clinical evaluation at 1.5T and 3T. Magn Reson Med. 2016 Aug;76(2):591–601. doi: https://doi.org/10.1002/mrm.25868.
Wood JC, Zhang P, Rienhoff H, Abi-Saab W, Neufeld EJ. Liver MRI is more precise than liver biopsy for assessing total body iron balance: a comparison of MRI relaxometry with simulated liver biopsy results. Magn Reson Imaging. 2015 Jul;33(6):761-7. doi: https://doi.org/10.1016/j.mri.2015.02.016.
Aigner E, Theurl I, Theurl M, Lederer D, Haufe H, Dietze O, Strasser M, Datz C, Weiss G. Pathways underlying iron accumulation in human nonalcoholic fatty liver disease. Am J Clin Nutr. 2008 May;87(5):1374-83. doi: https://doi.org/10.1093/ajcn/87.5.1374
Maliken BD, Nelson JE, Klintworth HM, Beauchamp M, Yeh MM, Kowdley KV. Hepatic reticuloendothelial system cell iron deposition is associated with increased apoptosis in nonalcoholic fatty liver disease. Hepatology. 2013 May;57(5):1806-13. doi: https://doi.org/10.1002/hep.26238.
Eskreis-Winkler S, Corrias G, Monti S, Zheng J, Capanu M, Krebs S, et al. IDEAL-IQ in an oncologic population: meeting the challenge of concomitant liver fat and liver iron. Cancer Imaging. 2018 Dec 12;18(1):51. doi: https://doi.org/10.1186/s40644-018-0167-3.
Starekova J, Hernando D, Pickhardt PJ, Reeder SB. Quantification of Liver Fat Content with CT and MRI: State of the Art. Radiology. 2021 Nov;301(2):250-262. doi: https://doi.org/10.1148/radiol.2021204288. Epub 2021 Sep 21.
Funding
The current work was carried out without any external funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
One of the authors (DH) is an employee of Philips Health Care. This author provided support to imaging acquisition but was not involved in image or data analysis nor study results interpretation. One author (O.K.) receives research support from Philips Healthcare unrelated to the matter of this manuscript. All other authors of this manuscript declare no conflict of interest regarding to the content of this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Moura Cunha, G., Kolokythas, O., Chen, W. et al. Intra-examination agreement between multi-echo gradient echo and confounder-corrected chemical shift-encoded MR sequences for R2* estimation as a biomarker of liver iron content in patients with a wide range of T2*/R2* and proton density fat fraction values. Abdom Radiol 48, 2302–2310 (2023). https://doi.org/10.1007/s00261-023-03902-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-023-03902-4