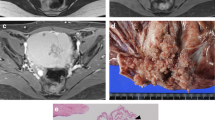
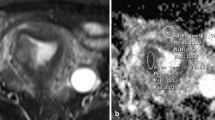
Abstract
Purpose
To investigate the feasibility of whole-tumor apparent diffusion coefficient (ADC) histogram analysis for improving the differentiation of endometriosis-related tumors: seromucinous borderline tumor (SMBT), clear cell carcinoma (CCC) and endometrioid carcinoma (EC).
Methods
Clinical features, solid component ADC (ADCSC) and whole-tumor ADC histogram-derived parameters (volume, the ADCmean, 10th, 50th and 90th percentile ADCs, inhomogeneity, skewness, kurtosis and entropy) were compared among 22 SMBTs, 42 CCCs and 21 ECs. Statistical analyses were performed using chi-square test, one-way ANOVA or Kruskal–Wallis test, and receiver operating characteristic curves.
Results
A significantly higher ADCSC and smaller volume were associated with SMBT than with CCC/EC. The ADCmean was significantly higher in CCC than in EC. The 10th percentile ADC was significantly lower in EC than in SMBT/CCC. The 50th and 90th percentile ADCs were significantly higher in CCC than in SMBT/EC. For differentiating SMBT from CCC, AUCs of the ADCSC, volume, and 50th and 90th percentile ADCs were 0.97, 0.86, 0.72 and 0.81, respectively. For differentiating SMBT from EC, AUCs of the ADCSC, volume and 10th percentile ADC were 0.97, 0.71 and 0.72, respectively. For differentiating CCC from EC, AUCs of the ADCmean and 10th, 50th and 90th percentile ADCs were 0.79, 0.72, 0.81 and 0.85, respectively.
Conclusion
Whole-tumor ADC histogram analysis was valuable for differentiating endometriosis-related tumors, and the 90th percentile ADC was optimal in differentiating CCC from EC.
Graphical abstract




Similar content being viewed by others
References
Kurata Y, Kido A, Moribata Y, et al. Diagnostic performance of MR imaging findings and quantitative values in the differentiation of seromucinous borderline tumour from endometriosis-related malignant ovarian tumour. Eur Radiol. 2017, 27: 1695-1703.
Maeda D, Shih I. Pathogenesis and the role of ARID1A mutation in endometriosis-related ovarian neoplasms. Adv Anat Pathol. 2013, 20: 45-52.
Mary A W, Ie-Ming S, James H S, et al. ancer Implications for Patients with Endometriosis. Semin Reprod Med. 2017, 35: 110-116.
Yoshikawa H, Jimbo H, Okada S et al. Prevalence of endometriosis in ovarian cancer. Gynecol Obstet Invest. 2000, 50 Suppl 1:11-7.
Nagamine M, Mikami Y. Ovarian seromucinous tumors: pathogenesis, morphologic spectrum, and clinical issues. Diagnostics (Basel). 2020, 10: 77.
Karpathiou G, Chauleur C, Corsini T, et al. Seromucinous ovarian tumor a comparison with the rest of ovarian epithelial tumors. Annals of Diagnostic Pathology. 2017, 27: 28-33.
NCCN Clinical practice guidelines in oncology: ovarian cancer including fallopian tube cancer and primary peritoneal cancer. Version 1.2021.
Li HM, Feng F, Qiang JW, et al. Quantitative dynamic contrast-enhanced MR imaging for differentiating benign, borderline, and malignant ovarian tumors. Abdom Radiol (NY). 2018, 43: 3132-3141.
Guiu MX, Stewart CJR. Endometriosis-associated ovarian neoplasia. Pathology. 2018, 50: 190-204.
Li M, Tan J, Zhang Y, et al. Assessing CT imaging features combined with CEA and CA125 levels to identify endometriosis-associated ovarian cancer. Abdom Radiol (NY). 2021, 46: 2367-2375..
Han J W, Kim K A, Chang H Y, et al. Newly categorized seromucinous tumor of the ovary: magnetic resonance imaging findings. J Comput Assist Tomogr. 2019, 43: 119-127.
Kurata Y, Kido A, Moribata Y, et al. Differentiation of seromucinous borderline tumor from serous borderline tumor on MR imaging. Magn Reson Med Sci. 2018, 17: 211-217.
Liu T, Sumida D, Wada T, et al. A diagnostic challenge of seromucinous borderline tumor: a case report. Medicine (Baltimore). 2019, 98: e15707.
Morioka S, Kawaguchi R, Yamada Y, et al. Magnetic resonance imaging findings for discriminating clear cell carcinoma and endometrioid carcinoma of the ovary. J Ovarian Res. 2019, 12: 20.
Yumiko O T, Satoshi O, Takako Y, et al. MRI of endometriotic cysts in association with ovarian carcinoma. AJR Am J Roentgenol. 2010, 194: 355-361.
Lu J, Li H M, Cai S Q, et al. Prediction of platinum-based chemotherapy response in advanced high-grade serous ovarian cancer: ADC histogram analysis of primary tumors. Acad Radiol. 2020: S1076-6332(20)30052-0.
He M, Song Y, Li H, et al. Histogram analysis comparison of monoexponential, advanced diffusion-weighted imaging, and dynamic contrast-enhanced MRI for differentiating borderline from malignant epithelial ovarian tumors. J Magn Reson Imaging. 2020, 52: 257-268..
Li H M, Zhang R, Gu W Y, et al. Whole solid tumour volume histogram analysis of the apparent diffusion coefficient for differentiating high-grade from low-grade serous ovarian carcinoma: correlation with Ki-67 proliferation status. Clin Radiol. 2019, 74: 918-925.
Xu Y S, Liu H F, Xi D L, et al. Whole-lesion histogram analysis metrics of the apparent diffusion coefficient: a correlation study with histological grade of hepatocellular carcinoma. Abdom Radiol (NY). 2019, 44: 3089-3098.
Xiao Z, Tang Z, Zhang J, et al. Whole-tumor histogram analysis of monoexponential and advanced diffusion-weighted imaging for sinonasal malignant tumors: Correlations with histopathologic features. J Magn Reson Imaging. 2020, 51: 273-285.
Zhang Z, Song C, Zhang Y, et al. Apparent diffusion coefficient (ADC) histogram analysis: differentiation of benign from malignant parotid gland tumors using readout-segmented diffusion-weighted imaging. Dentomaxillofac Radiol. 2019, 48: 20190100.
Pope W B, Kim H J, Huo J, et al. Recurrent glioblastoma multiforme: ADC histogram analysis predicts response to bevacizumab treatment. Radiology. 2009, 252: 182-189.
Kyriazi S, Collins D J, Messiou C, et al. Metastatic ovarian and primary peritoneal cancer: assessing chemotherapy response with diffusion-weighted MR imaging--value of histogram analysis of apparent diffusion coefficients. Radiology. 2011, 261: 182-192.
Zhou L, Yao L, Dai L, et al. Ovarian endometrioid carcinoma and clear cell carcinoma: A 21-year retrospective study. J Ovarian Res. 2021, 14:63.
Kim H S, Kim T H, Chung H H, et al. Risk and prognosis of ovarian cancer in women with endometriosis: a meta-analysis. Br J Cancer. 2014, 110:1878-1890.
Zhang Y D, Wu C J, Wang Q, et al. Comparison of utility of histogram apparent diffusion coefficient and R2* for differentiation of low-grade from high-grade clear cell renal cell carcinoma. AJR Am J Roentgenol. 2015, 205: W193-W201.
Ono T, Kishimoto K, Tajima S, et al. Apparent diffusion coefficient (ADC) values of serous, endometrioid, and clear cell carcinoma of the ovary: pathological correlation. Acta Radiol. 2020, 61: 992-1000.
Schob S, Meyer H, Dieckow J, et al. Histogram analysis of diffusion weighted imaging at 3T is useful for prediction of lymphatic metastatic spread, proliferative activity, and cellularity in thyroid cancer. Int J Mol Sci. 2017, 18: 821.
Wang F, Wang Y X, Zhou Y, et al. Apparent diffusion coefficient histogram analysis for assessing tumor staging and detection of lymph node metastasis in epithelial ovarian cancer: correlation with p53 and Ki-67 expression. Mol Imaging Biol. 2019, 21: 731-739.
Moteki T, Horikoshi H, Endo K. Relationship between apparent diffusion coefficient and signal intensity in endometrial and other pelvic cysts. Magn Reson Imaging. 2002, 20: 463-470.
Kato N, Sasou S, Motoyama T. Expression of hepatocyte nuclear factor-1beta (HNF-1beta) in clear cell tumors and endometriosis of the ovary. Mod Pathol. 2006, 19: 83-89.
Xiao W B, Awadallah A, Xin W. Loss of ARID1A/BAF250a expression in ovarian endometriosis and clear cell carcinoma. Int J Clin Exp Pathol. 2012, 5: 642-650.
Funding
This work was supported by the National Natural Science Foundation of P.R. China [No. 81971579], Shanghai Municipal Commission of Science and Technology [No. 19411972000], Shanghai Municipal Health Commission [No. ZK2019B01] and Shanghai Jinshan District Health Commission [No.JSYQ201901].
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lu, J., Zhao, S., Ma, F. et al. Whole-tumor ADC histogram analysis for differentiating endometriosis-related tumors: seromucinous borderline tumor, clear cell carcinoma and endometrioid carcinoma. Abdom Radiol 48, 724–732 (2023). https://doi.org/10.1007/s00261-022-03742-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-022-03742-8