Abstract
Purpose
To assess response to programmed death-1 (PD-1) monotherapy (nivolumab) in hepatocellular carcinoma (HCC) patients using RECIST1.1, modified RECIST (mRECIST), and immune RECIST (iRECIST). A secondary objective was to identify clinicolaboratory and imaging variables predictive of progressive disease (PD) and overall survival (OS).
Methods
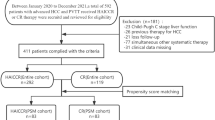
Patients with HCC treated with nivolumab at a single institution from 5/2016 to 12/2019 with MRI or CT performed ≥ 4 weeks post treatment were retrospectively assessed. Patients who received concurrent locoregional, radiation, or other systemic therapies were excluded. Response was assessed by 2 observers in consensus using RECIST1.1, mRECIST, and iRECIST at 3/6/9/12-month time points. Time to progression (TTP) and OS were recorded. Clinicolaboratory and imaging variables were evaluated as predictors of PD and OS using uni-/multivariable and Cox regression analyses.
Results
Fifty-eight patients (42M/16F) were included. 118 target lesions (TL) were identified before treatment. Baseline mean TL size was 49.1 ± 43.5 mm (range 10–189 mm) for RECIST1.1/iRECIST and 46.3 ± 42.3 mm (range 10–189 mm) for mRECIST. Objective response rate (ORR) was 21% for mRECIST/iRECIST/RECIST1.1, with no cases of pseudoprogression. Median OS and median TTP were 717 days and 127 days for RECIST1.1/mRECIST/iRECIST-iUPD (unconfirmed PD). Older age, MELD/Child–Pugh scores, AFP, prior transarterial radioembolization (TARE), and larger TL size were predictive of PD and/or poor OS using mRECIST/iRECIST. The strongest predictor of PD (HR = 2.49, 95% CI 1.29–4.81, p = 0.007) was TARE. The strongest predictor of poor OS was PD by mRECIST/iRECIST at 3 months (HR = 2.26, 95% CI 1.00–5.10, p = 0.05) with borderline significance.
Conclusion
Our results show ORR of 21%, equivalent for mRECIST, iRECIST, and RECIST1.1 in patients with advanced HCC clinically treated with nivolumab.





Similar content being viewed by others
Data availability
Upon request, the authors are prepared to send relevant documentation or data in order to verify the validity of the results presented. Code availability (software application or custom code): N/A.
Code availability
N/A.
Abbreviations
- ALT:
-
Alanine aminotransferase
- AST:
-
Aspartate aminotransferase
- CP:
-
Child–Pugh
- CR:
-
Complete response
- CT:
-
Computed tomography
- HBV:
-
Hepatitis B virus
- HCC:
-
Hepatocellular carcinoma
- HCV:
-
Hepatitis C virus
- INR:
-
International normalized ratio
- iCPD:
-
Confirmed progressive disease
- iRECIST:
-
Immune Response Evaluation Criteria in Solid Tumors
- iUPD:
-
Unconfirmed progressive disease
- LRT:
-
Locoregional therapy
- MELD:
-
Model for end-stage liver disease
- mRECIST:
-
Modified Response Evaluation Criteria in Solid Tumors
- MRI:
-
Magnetic resonance imaging
- ORR:
-
Objective response rate
- OS:
-
Overall survival
- PD-1:
-
Programmed death-1
- PD:
-
Progressive disease
- PR:
-
Partial response
- RECIST1.1:
-
Response Evaluation Criteria in Solid Tumors
- SD:
-
Stable disease
- TL:
-
Target lesion
- TARE:
-
Transarterial radioembolization
- TTP:
-
Time to progression
References
Singal, A.G., P. Lampertico, and P. Nahon, Epidemiology and surveillance for hepatocellular carcinoma: New trends. J Hepatol, 2020. 72(2): p. 250-261.
Forner, A., et al., Current strategy for staging and treatment: the BCLC update and future prospects. Semin Liver Dis, 2010. 30(1): p. 61-74.
Llovet, J.M., et al., Sorafenib in advanced hepatocellular carcinoma. N Engl J Med, 2008. 359(4): p. 378-90.
Llovet, J.M., et al., Molecular therapies and precision medicine for hepatocellular carcinoma. Nat Rev Clin Oncol, 2018. 15(10): p. 599-616.
Cheng, A.L., et al., Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol, 2009. 10(1): p. 25-34.
Meyer, T., Treatment of advanced hepatocellular carcinoma: beyond sorafenib. Lancet Gastroenterol Hepatol, 2018. 3(4): p. 218-220.
El-Khoueiry, A.B., et al., Nivolumab in patientss with advanced hepatocellular carcinoma (CheckMate 040): an open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet, 2017. 389(10088): p. 2492-2502.
Cheng, H., et al., Trends in the treatment of advanced hepatocellular carcinoma: immune checkpoint blockade immunotherapy and related combination therapies. Am J Cancer Res, 2019. 9(8): p. 1536-1545.
Fessas, P., et al., Post-registration experience of nivolumab in advanced hepatocellular carcinoma: an international study. J Immunother Cancer, 2020. 8(2).
Llovet, J.M. and R. Lencioni, mRECIST for HCC: Performance and novel refinements. J Hepatol, 2020. 72(2): p. 288-306.
Ma, Y., et al., How to differentiate pseudoprogression from true progression in cancer patients treated with immunotherapy. Am J Cancer Res, 2019. 9(8): p. 1546-1553.
Seymour, L., et al., iRECIST: guidelines for response criteria for use in trials testing immunotherapeutics. Lancet Oncol, 2017. 18(3): p. e143-e152.
(ACR), A.C.o.R. CT/MRI LI-RADS® v2018. 2018.
Eisenhauer, E.A., et al., New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer, 2009. 45(2): p. 228–47.
Lencioni, R. and J.M. Llovet, Modified RECIST (mRECIST) assessment for hepatocellular carcinoma. Semin Liver Dis, 2010. 30(1): p. 52-60.
T, Z.H.a.H., Regularization and variable selection via the Elastic Net. Journal of the Royal Statistical Society, Series B, 2005. 67: p. 301–320.
Cox, D.R.a.O., D, Analysis of Survival Data. 1984, New York: Chapman & Hall.
Friedman, J., T. Hastie, and R. Tibshirani, Regularization Paths for Generalized Linear Models via Coordinate Descent. J Stat Softw, 2010. 33(1): p. 1-22.
Pinato, D.J., et al., Immune-based therapies for hepatocellular carcinoma. Oncogene, 2020. 39(18): p. 3620-3637.
Ziogas, I.A., et al., The Role of Immunotherapy in Hepatocellular Carcinoma: A Systematic Review and Pooled Analysis of 2,402 Patients. Oncologist, 2020.
Kudo, M., Systemic Therapy for Hepatocellular Carcinoma: Latest Advances. Cancers (Basel), 2018. 10(11).
Finn, R.S., et al., Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N Engl J Med, 2020. 382(20): p. 1894-1905.
Zhu, A.X., et al., Pembrolizumab in patientss with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): a non-randomised, open-label phase 2 trial. Lancet Oncol, 2018. 19(7): p. 940-952.
Borcoman, E., et al., Patterns of Response and Progression to Immunotherapy. Am Soc Clin Oncol Educ Book, 2018. 38: p. 169-178.
Lai, Y.C., et al., Response evaluation for immunotherapy through semi-automatic software based on RECIST 1.1, irRC, and iRECIST criteria: comparison with subjective assessment. Acta Radiol, 2020. 61(7): p. 983–991.
Kurra, V., et al., Pseudoprogression in cancer immunotherapy: Rates, time course and patient outcomes. Journal of Clinical Oncology, 2016. 34(15_suppl): p. 6580–6580.
Au, J.S. and C.T. Frenette, Management of Hepatocellular Carcinoma: Current Status and Future Directions. Gut Liver, 2015. 9(4): p. 437-48.
Tai, W.M.D., et al., A phase II open-label, single-center, nonrandomized trial of Y90-radioembolization in combination with nivolumab in Asian patients with advanced hepatocellular carcinoma: CA 209–678. Journal of Clinical Oncology, 2020. 38(15_suppl): p. 4590–4590.
Wehrenberg-Klee, E., et al., Y-90 Radioembolization Combined with a PD-1 Inhibitor for Advanced Hepatocellular Carcinoma. Cardiovasc Intervent Radiol, 2018. 41(11): p. 1799-1802.
Funding
N/A.
Author information
Authors and Affiliations
Contributions
SL: Conceptualization; data curation; formal analysis; investigation; methodology; project administration; supervision; and writing. MAC: data curation; formal analysis; and project administration. KML: data curation; formal analysis; and investigation. OB: conceptualization and methodology. SJH: conceptualization; methodology; and writing. WM: formal analysis; methodology; and software. PW: formal analysis; investigation; and supervision. DS: conceptualization; formal analysis; and methodology. DM: data curation; project administration; and writing. DJP, MS, and TM: supervision and writing. MS: data curation; supervision; and writing. BT: conceptualization; supervision; visualization; and writing.
Corresponding author
Ethics declarations
Conflict of interest
Stefanie Hectors: Employment at Regeneron Pharmaceuticals. David Pinato: Lecture fees (ViiV Healthcare, EISAI, BMS, Roche, Bayer Healthcare), travel expenses (BMS, MSD and Bayer Healthcare), consulting fees (Mina Therapeutics, EISAI, Roche, Astra Zeneca, DaVolterra) research funding to institution (MSD, BMS, Biognosys). Thomas Marron: Advisory boards and/or DSMBs (Boehringer Ingelheim, BMS, AstraZeneca, Genentech, Celldex, and Regeneron), research grant support (BMS, Regeneron and Boehringer Ingelheim). Bachir Taouli: Grant/research support (Bayer, Takeda, Regeneron, Siemens, Echosens). The remaining authors (SL, MAC, KML, OB, WM, PW, DS, DVM, MS, and MS) have no relevant disclosures.
Ethical approval
Because this work involves the use of human subjects, we ensure that all procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2008. The study was approved by the Institutions Review Board at the ISMMS.
Consent to participate
Waiver of informed consent was obtained.
Consent for publication
N/A.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lewis, S., Cedillo, M.A., Lee, K.M. et al. Comparative assessment of standard and immune response criteria for evaluation of response to PD-1 monotherapy in unresectable HCC. Abdom Radiol 47, 969–980 (2022). https://doi.org/10.1007/s00261-021-03386-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-021-03386-0