Abstract
Purpose
To determine the inter-reader agreement of abbreviated magnetic resonance imaging (AMRI) for the detection of hepatocellular carcinoma (HCC) and explore the causes of heterogeneity between the reported results.
Methods
Original studies reporting the inter-reader agreement of AMRI for detecting HCC were identified in MEDLINE, EMBASE, and Cochrane databases. The pooled kappa coefficient (κ) was calculated using the DerSimonian–Laird random-effects model. Subgroup analyses were performed according to the AMRI protocol (non-contrast [NC]-AMRI, dynamic contrast-enhanced [DCE]-AMRI, and hepatobiliary phase [HBP]-AMRI). Meta-regression analyses were performed to further explore study heterogeneity.
Results
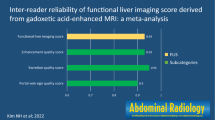
In the eight included studies (1182 patients), the overall pooled κ was 0.76 (95% confidence interval [CI], 0.70–0.82; I2 = 74.4%). The κ of NC-AMRI, DCE-AMRI, and HBP-AMRI were 0.72 (95% CI, 0.62–0.82), 0.80 (95% CI, 0.78–0.82), and 0.98 (95% CI, 0.95–1.00), respectively. In the NC-AMRI, the pooled κ of NC-AMRI using only diffusion-weighted imaging (DWI) was 0.64, which was lower than the values using two or more imaging sequences (κ = 0.74–0.77). In subgroup analysis, no study heterogeneity was noted in studies using DCE-AMRI (I2 = 0%), whereas high heterogeneity was noted with NC-AMRI (I2 = 80.5%). Especially, NC-AMRI including more than two imaging sequences showed high residual heterogeneity (I2 = 87.6%). Meta-regression analysis found that difference in reader experience was significantly associated with study heterogeneity (p = .02).
Conclusion
AMRI for detecting HCC showed substantial inter-reader agreement across all examined protocols. NC-AMRI, notably NC-AMRI using only DWI, had relatively low inter-reader agreement. Therefore, DCE-AMRI or HBP-AMRI may be more reliable than NC-AMRI using only DWI.
Graphic abstract



Similar content being viewed by others
Abbreviations
- AMRI:
-
Abbreviated magnetic resonance imaging
- CI:
-
Confidence interval
- CT:
-
Computed tomography
- DCE:
-
Dynamic contrast-enhanced
- DWI:
-
Diffusion-weighed imaging
- HBP:
-
Hepatobiliary phase
- HCC:
-
Hepatocellular carcinoma
- NC:
-
Non-contrast
- MRI:
-
Magnetic resonance imaging
- T2WI:
-
T2-weighted imaging
- US:
-
Ultrasound
References
Zhang BH, Yang BH, Tang ZY (2004) Randomized controlled trial of screening for hepatocellular carcinoma. J Cancer Res Clin Oncol 130:417-422. doi: https://doi.org/10.1007/s00432-004-0552-0.
Singal AG, Pillai A, Tiro J (2014) Early detection, curative treatment, and survival rates for hepatocellular carcinoma surveillance in patients with cirrhosis: a meta-analysis. PLoS Med 11:e1001624. doi: https://doi.org/10.1371/journal.pmed.1001624.
Marrero JA, Kulik LM, Sirlin CB et al (2018) Diagnosis, Staging, and Management of Hepatocellular Carcinoma: 2018 Practice Guidance by the American Association for the Study of Liver Diseases. Hepatology 68:723-750. doi: https://doi.org/10.1002/hep.29913.
European Association for the Study of the Liver (2018) EASL Clinical Practice Guidelines: Management of hepatocellular carcinoma. J Hepatol 69:182-236. doi: https://doi.org/10.1016/j.jhep.2018.03.019.
Omata M, Cheng AL, Kokudo N et al (2017) Asia-Pacific clinical practice guidelines on the management of hepatocellular carcinoma: a 2017 update. Hepatol Int 11:317-370. doi: https://doi.org/10.1007/s12072-017-9799-9.
Tzartzeva K, Obi J, Rich NE et al (2018) Surveillance Imaging and Alpha Fetoprotein for Early Detection of Hepatocellular Carcinoma in Patients With Cirrhosis: A Meta-analysis. Gastroenterology 154:1706-1718.e1. doi: https://doi.org/10.1053/j.gastro.2018.01.064.
Kim SY, An J, Lim YS et al (2017) MRI With Liver-Specific Contrast for Surveillance of Patients With Cirrhosis at High Risk of Hepatocellular Carcinoma. JAMA Oncol 3:456-463. doi: https://doi.org/10.1001/jamaoncol.2016.3147.
An JY, Pena MA, Cunha GM et al (2020) Abbreviated MRI for Hepatocellular Carcinoma Screening and Surveillance. Radiographics 40:1916-1931. doi: https://doi.org/10.1148/rg.2020200104.
Gupta P, Soundararajan R, Patel A, Kumar MP, Sharma V, Kalra N (2021) Abbreviated MRI for hepatocellular carcinoma screening: A systematic review and meta-analysis. J Hepatol 75:108-119. doi: https://doi.org/10.1016/j.jhep.2021.01.041.
Han S, Choi J-I, Park MY, Choi MH, Rha SE, Lee YJ (2018) The Diagnostic Performance of Liver MRI without Intravenous Contrast for Detecting Hepatocellular Carcinoma: A Case-Controlled Feasibility Study. Korean J Radiol 19:568-577. doi: https://doi.org/10.3348/kjr.2018.19.4.568.
Chan MV, McDonald SJ, Ong Y-Y et al (2019) HCC screening: assessment of an abbreviated non-contrast MRI protocol. Eur Radiol Exp 3:49. doi: https://doi.org/10.1186/s41747-019-0126-1.
Khatri G, Pedrosa I, Ananthakrishnan L et al (2019) Abbreviated‐protocol screening MRI vs. complete‐protocol diagnostic MRI for detection of hepatocellular carcinoma in patients with cirrhosis: An equivalence study using LI‐RADS v2018. J Magn Reson Imaging 51:415-425. doi: https://doi.org/10.1002/jmri.26835.
Kim JS, Lee JK, Baek SY, Yun HI (2019) Diagnostic performance of a minimized protocol of non-contrast MRI for hepatocellular carcinoma surveillance. Abdom Radiol (NY) 45:211-219. doi: https://doi.org/10.1007/s00261-019-02277-9.
Whang S, Choi MH, Choi J-I, Youn SY, Kim DH, Rha SE (2020) Comparison of diagnostic performance of non-contrast MRI and abbreviated MRI using gadoxetic acid in initially diagnosed hepatocellular carcinoma patients: a simulation study of surveillance for hepatocellular carcinomas. Eur Radiol 30:4150-4163. doi: https://doi.org/10.1007/s00330-020-06754-4.
Stroup DF, Berlin JA, Morton SC et al (2000) Meta-analysis of observational studies in epidemiology: a proposal for reporting. Meta-analysis Of Observational Studies in Epidemiology (MOOSE) group. JAMA 283:2008-2012. doi: https://doi.org/10.1001/jama.283.15.2008.
Liberati A, Altman DG, Tetzlaff J et al (2009) The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate healthcare interventions: explanation and elaboration. BMJ 339:b2700. doi: https://doi.org/10.1136/bmj.b2700.
Kottner J, Audigé L, Brorson S et al (2011) Guidelines for Reporting Reliability and Agreement Studies (GRRAS) were proposed. J Clin Epidemiol 64:96-106. doi: https://doi.org/10.1016/j.jclinepi.2010.03.002.
IntHout J, Ioannidis JPA, Borm GF (2014) The Hartung-Knapp-Sidik-Jonkman method for random effects meta-analysis is straightforward and considerably outperforms the standard DerSimonian-Laird method. BMC Med Res Methodol 14:25. doi: https://doi.org/10.1186/1471-2288-14-25.
Landis JR, Koch GG (1977) The measurement of observer agreement for categorical data. Biometrics 33:159-174.
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327:557-560. doi: https://doi.org/10.1136/bmj.327.7414.557.
Hardie AD, Kizziah MK, Boulter DJ (2011) Diagnostic accuracy of diffusion-weighted MRI for identifying hepatocellular carcinoma with liver explant correlation. J Med Imaging Radiat Oncol 55:362-367. doi: https://doi.org/10.1111/j.1754-9485.2011.02286.x.
Hardie AD, Kizziah MK, Rissing MS (2011) Can the Patient With Cirrhosis Be Imaged for Hepatocellular Carcinoma Without Gadolinium? J Comput Assist Tomogr 35:711-715. doi: https://doi.org/10.1097/RCT.0b013e31823421ac.
Piana G, Trinquart L, Meskine N, Barrau V, Beers BV, Vilgrain V (2011) New MR imaging criteria with a diffusion-weighted sequence for the diagnosis of hepatocellular carcinoma in chronic liver diseases. J Hepatol 55:126-132. doi: https://doi.org/10.1016/j.jhep.2010.10.023.
Chan MV, Huo YR, Trieu N et al (2021) Noncontrast MRI for Hepatocellular Carcinoma Detection: A Systematic Review and Meta-analysis – A Potential Surveillance Tool? Clin Gastroenterol Hepatol doi: https://doi.org/10.1016/j.cgh.2021.02.036.
Kim DH, Choi SH, Shim JH et al (2021) Meta-Analysis of the Accuracy of Abbreviated Magnetic Resonance Imaging for Hepatocellular Carcinoma Surveillance: Non-Contrast versus Hepatobiliary Phase-Abbreviated Magnetic Resonance Imaging. Cancers (Basel) 13:2975. doi: https://doi.org/10.3390/cancers13122975.
Vietti Violi N, Lewis S, Liao J et al (2020) Gadoxetate-enhanced abbreviated MRI is highly accurate for hepatocellular carcinoma screening. Eur Radiol 30:6003-6013. doi: https://doi.org/10.1007/s00330-020-07014-1.
Besa C, Lewis S, Pandharipande PV et al (2017) Hepatocellular carcinoma detection: diagnostic performance of a simulated abbreviated MRI protocol combining diffusion-weighted and T1-weighted imaging at the delayed phase post gadoxetic acid. Abdom Radiol (NY) 42:179-190. doi: https://doi.org/10.1007/s00261-016-0841-5.
Marks RM, Ryan A, Heba ER et al (2015) Diagnostic per-patient accuracy of an abbreviated hepatobiliary phase gadoxetic acid-enhanced MRI for hepatocellular carcinoma surveillance. AJR Am J Roentgenol 204:527-535. doi: https://doi.org/10.2214/AJR.14.12986.
Brunsing RL, Chen DH, Schlein A et al (2019) Gadoxetate-enhanced Abbreviated MRI for Hepatocellular Carcinoma Surveillance: Preliminary Experience. Radiol Imaging Cancer 1:e190010. doi: https://doi.org/10.1148/rycan.2019190010.
Lee JY, Huo EJ, Weinstein S et al (2018) Evaluation of an abbreviated screening MRI protocol for patients at risk for hepatocellular carcinoma. Abdom Radiol (NY) 43:1627-1633. doi: https://doi.org/10.1007/s00261-017-1339-5.
Brunsing RL, Fowler KJ, Yokoo T, Cunha GM, Sirlin CB, Marks RM (2020) Alternative approach of hepatocellular carcinoma surveillance: abbreviated MRI. Hepatoma Res 6:59. doi: https://doi.org/10.20517/2394-5079.2020.50.
Saito K, Moriyasu F, Sugimoto K et al (2012) Histological grade of differentiation of hepatocellular carcinoma: Comparison of the efficacy of diffusion-weighted MRI with T2-weighted imaging and angiography-assisted CT. J Med Imaging Radiat Oncol 56:261-269. doi: https://doi.org/10.1111/j.1754-9485.2012.02374.x.
Chandarana H, Taouli B (2010) Diffusion and perfusion imaging of the liver. Eur J Radiol 76:348-358. doi: https://doi.org/10.1016/j.ejrad.2010.03.016.
Deeks JJ, Higgins JPT, Altman DG (2021) Chapter 10: Analysing data and undertaking meta-analyses. Cochrane Handbook for Systematic Reviews of Interventions version 6.2 (updated February 2021). Cochrane. Available from http://www.training.cochrane.org/handbook. Accessed 8 May 2021.
Chernyak V, Fowler KJ, Kamaya A et al (2018) Liver Imaging Reporting and Data System (LI-RADS) Version 2018: Imaging of Hepatocellular Carcinoma in At-Risk Patients. Radiology 289:816-830. doi: https://doi.org/10.1148/radiol.2018181494.
Funding
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MSIT) (Grant Number: NRF-2019R1G1A1099743).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
Institutional review board approval and written informed consent is not required because this study is a meta-analysis based on previously published studies.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kim, D.H., Choi, S.H., Lee, J.S. et al. Inter-reader agreement of abbreviated magnetic resonance imaging for hepatocellular carcinoma detection: a systematic review and meta-analysis. Abdom Radiol 47, 123–132 (2022). https://doi.org/10.1007/s00261-021-03297-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-021-03297-0