Abstract
Purpose
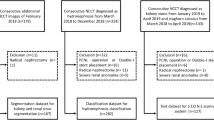
Hydronephrosis is the dilation of the pelvicalyceal system due to the urine flow obstruction in one or both kidneys. Conventionally, renal pelvis anterior–posterior diameter (APD) was used for quantifying hydronephrosis in medical images (e.g., ultrasound, CT, and functional MRI). Our study aimed to automatically detect and quantify the fluid and kidney areas on ultrasonography, using a deep learning approach.
Methods
An attention-Unet was used to segment the kidney and the dilated pelvicalyceal system with fluid. The gold standard for diagnosing hydronephrosis was the APD > 1.0 cm. For semi-quantification, we proposed a fluid-to-kidney-area ratio measurement, i.e., \(\frac{{{\text{renal~pelvicalyceal~area~with~fluid}}}}{{{\text{~kidney~area}}}}\), as a deep learning-derived biomarker. Dice coefficient, confusion matrix, ROC curve, and Z-test were used to evaluate the model performance. Linear regression was applied to obtain the fluid-to-kidney-area ratio cutoff for detecting hydronephrosis.
Results
For regional kidney segmentation, the Dice coefficients were 0.92 and 0.83 for the kidney and dilated pelvicalyceal system, respectively. The sensitivity and specificity of detecting dilated pelvicalyceal system were 0.99 and 0.83, respectively. The linear equation was fluid-to-kidney-area ratio = (0.213 ± 0.004) × APD (in cm) for 95% confidence interval on the slope with R2 = 0.87. The fluid-to-kidney-area ratio cutoff for detecting hydronephrosis was 0.213. The sensitivity and specificity for detecting hydronephrosis were 0.90 and 0.80, respectively.
Conclusion
Our study confirmed the feasibility of deep learning characterization of the kidney and fluid, showing an automatic pediatric hydronephrosis detection.








Similar content being viewed by others
Abbreviations
- APD:
-
Anterior–posterior diameter
- UTI:
-
Urinary tract infection
- CAP:
-
Continuous antibiotic prophylaxis
- CKD:
-
Chronic kidney disease
- SFU:
-
Society for fetal urology
- UTD:
-
Urinary tract dilation
- SAM:
-
Active shape models
- AUC:
-
Area under the curve
References
Riccabona, M., Assessment and management of newborn hydronephrosis. World J Urol, 2004. 22(2): p. 73-78
Skoog, S.J., et al., Pediatric vesicoureteral reflux guidelines panel summary report: clinical practice guidelines for screening siblings of children with vesicoureteral reflux and neonates/infants with prenatal hydronephrosis. J Urol, 2010. 184(3): p. 1145-1151.
Walsh, T.J., et al., Antenatal Hydronephrosis and the Risk of Pyelonephritis Hospitalization During the First Year of Life. Urology, 2007. 69(5): p. 970-974.
Silay, M.S., et al., Role of antibiotic prophylaxis in antenatal hydronephrosis: A systematic review from the European Association of Urology/European Society for Paediatric Urology Guidelines Panel. J Pediatr Urol, 2017. 13(3): p. 306-315.
Sencan, A., et al., Urinary tract infection and vesicoureteral reflux in children with mild antenatal hydronephrosis. J Pediatr Urol, 2014. 10(6):1008-1013.
Braga, L.H., et al., Antibiotic prophylaxis for urinary tract infections in antenatal hydronephrosis. Pediatrics, 2013. 131(1): p. e251-e261.
Chevalier, R.L., et al., Mechanisms of renal injury and progression of renal disease in congenital obstructive nephropathy. Pediatr Nephrol, 2010. 25(4): p. 687-697.
Liu, S., et al., Deep Learning in Medical Ultrasound Analysis: A Review. Engineering, 2019. 5(2): p. 261-275.
Fernbach, S.K., M. Maizels, and J.J. Conway, Ultrasound grading of hydronephrosis: Introduction to the system used by the society for fetal urology. Pediatr Radiol, 1993. 23(6): p. 478-480.
Sidhu, G., J. Beyene, and N.D. Rosenblum, Outcome of isolated antenatal hydronephrosis: a systematic review and meta-analysis. Pediatr Nephrol, 2005. 21(2): p. 218-224.
Cheng, A.M., et al., Outcome of Isolated Antenatal Hydronephrosis. Arch Pediatr Adolesc Med, 2004. 158(1): p. 38-40.
Nguyen, H.T., et al., Multidisciplinary consensus on the classification of prenatal and postnatal urinary tract dilation (UTD classification system). J Pediatr Urol, 2014. 10(6): p. 982-998.
P H Arger, B.G.C., M C Mintz, H P Snyder, T Camardese, R L Arenson, S G Gabbe, L Aquino, Routine fetal genitourinary tract screening. Radiology, 1985 156.
Onen, A., Grading of Hydronephrosis: An Ongoing Challenge. Frontiers in Pediatrics, 2020. 8(458).
Onen, A., An alternative grading system to refine the criteria for severity of hydronephrosis and optimal treatment guidelines in neonates with primary UPJ-type hydronephrosis. Journal of Pediatric Urology, 2007(3): p. 200-205.
Vemulakonda, V.M., et al., Inter-rater reliability of postnatal ultrasound interpretation in infants with congenital hydronephrosis. Int Urol Nephrol, 2015. 47(9): p. 1457-1461.
Longpre, M., et al., Prediction of the outcome of antenatally diagnosed hydronephrosis: A multivariable analysis. J Pediatr Urol, 2012. 8(2): p. 135-139.
Bouzada, M.C.F., et al., Diagnostic accuracy of fetal renal pelvis anteroposterior diameter as a predictor of uropathy: a prospective study: Diagnostic accuracy of fetal renal pelvis diameter. Ultrasound in obstetrics & gynecology, 2004. 24(7): p. 745-749.
Arora, S., et al., Predictors for the need of surgery in antenatally detected hydronephrosis due to UPJ obstruction – A prospective multivariate analysis. J Pediatr Urol, 2015. 11(5): p. 248.e1-248.e5.
Dos Santos, J., et al., A New Grading System for the Management of Antenatal Hydronephrosis. Clin J Am Soc Nephrol, 2015. 10(10): p. 1783-1790.
Sadeghi-Bojd, S., et al., Postnatal Evaluation and Outcome of Prenatal Hydronephrosis. Iran J Pediatr, 2016. 26(2): p. e3667-e3667.
Conway, J.J. and M. Maizels, The "well tempered" diuretic renogram: a standard method to examine the asymptomatic neonate with hydronephrosis or hydroureteronephrosis. A report from combined meetings of The Society for Fetal Urology and members of The Pediatric Nuclear Medicine Council--The Society of Nuclear Medicine. J Nucl Med, 1992. 33(11): p. 2047-2051.
Blachar, A., et al., Clinical outcome and follow-up of prenatal hydronephrosis. Pediatr Nephrol, 1994. 8(1): p. 30-35.
Grignon, A., et al., Urinary tract dilatation in utero: classification and clinical applications. Radiology, 1986. 160(3): p. 645-7.
Andrzej Paweł Wieczorek, M.M.W.a.J.F.T., Errors in the ultrasound diagnosis of the kidneys, ureters and urinary bladder. Journal of Ultrasound, 2013. 13: p. 308-318.
Hermsen, M., et al., Deep Learning–Based Histopathologic Assessment of Kidney Tissue. J Am Soc Nephrol, 2019. 30(10): p. 1968-1979.
Sabanayagam, C., et al., A deep learning algorithm to detect chronic kidney disease from retinal photographs in community-based populations. Lancet Digit Health, 2020. 2(6): p. e295-e302.
Ginley, B., et al., Computational Segmentation and Classification of Diabetic Glomerulosclerosis. J Am Soc Nephrol, 2019. 30(10): p. 1953-1967.
Makino, M., et al., Artificial intelligence predicts the progression of diabetic kidney disease using big data machine learning. Sci Rep, 2019. 9(1): p. 11862-9.
Khamparia, A., et al., KDSAE: Chronic kidney disease classification with multimedia data learning using deep stacked autoencoder network. Multimedia tools and applications, 2019: p. 1-16.
Chin-Chi Kuo, C.-M.C., Kuan-Ting Liu, Wei-Kai Lin, Hsiu-Yin Chiang, Chih-Wei Chung, Meng-Ru Ho, Pei-Ran Sun, Rong-Lin Yang & Kuan-Ta Chen Automation of the kidney function prediction and classification through ultrasound-based kidney imaging using deep learning. npj Digital Medicine, 2019.
Almarashi, A., M. Alghamdi, and I. Mechai, A new mathematical model for diagnosing chronic diseases (kidney failure) using ANN. Cogent Mathematics & Statistics, 2018. 5(1): p. 1559457.
Ronneberger, O., Fischer, P., Brox, T., U-Net: Convolutional Networks for Biomedical Image Segmentation. Medical Image Computing and Computer-Assisted Intervention, 2015. pp. 234–241. https://doi.org/10.1007/978-3-319-24574-4_28.
O Oktay, J. Schlemper, LL Folgoc, M Lee, M Heinrich, K Misawa, K Mori, S McDonagh, NY Hammerla, B Kainz, B Glocker, D Rueckert, Attention U-Net: Learning Where to Look for the Pancreas. 2018, arXiv:1804.03999.
Rathnayaka, P., et al., Kidney Tumor Detection using Attention based U-Net. 2019, University of Minnesota Libraries Publishing. https://doi.org/10.24926/548719.079.
Yin, S., et al., Automatic kidney segmentation in ultrasound images using subsequent boundary distance regression and pixelwise classification networks. Med Image Anal, 2020. 60: p. 101602-101602.
Jokar, E., Pourghassem, H., Kidney Segmentation in Ultrasound Images Using Curvelet Transform and Shape Prior. International Conference on Communication Systems and Network Technologies, 2013, pp. 180–185. https://doi.org/10.1109/CSNT.2013.47.
Mendoza, C.S., et al. Automatic Analysis of Pediatric Renal Ultrasound Using Shape, Anatomical and Image Acquisition Priors. 2013. Berlin, Heidelberg: Springer Berlin Heidelberg.
Mendoza, C.S., et al., Kidney segmentation in ultrasound via genetic initialization and Active Shape Models with rotation correction. IEEE 10th International Symposium on Biomedical Imaging, 2013. pp. 69–72. https://doi.org/10.1109/ISBI.2013.6556414
Lauren C. Smail, K.D., Luis H. Braga, Suzanna Becker, and R.R. Sonnadara, Using Deep Learning Algorithms to Grade Hydronephrosis Severity: Toward a Clinical Adjunct. Frontiers in pediatrics, 2020.
Dhindsa, K., Smail, L.C., McGrath, M., Braga, L.H., Becker, S., Sonnadara, R.R., Grading Prenatal Hydronephrosis from Ultrasound Imaging Using Deep Convolutional Neural Networks. 2018 15th Conference on Computer and Robot Vision (CRV), 2018. pp. 80–87. https://doi.org/10.1109/CRV.2018.00021
Erdman, L., et al. Predicting Obstructive Hydronephrosis Based on Ultrasound Alone. 2020. Cham: Springer International Publishing.
Holmes N, H.M., Baskin LS,, Fetal surgery for posterior urethral valves: long-term postnatal outcomes. Pediatrics, 2001. 108(1).
Alconcher LF, T.M., Natural history of bilateral mild isolated antenatal hydronephrosis conservatively managed. Pediatr Nephrol, 2021. 27(7): p. 19-23.
Sinha, A., et al., Revised guidelines on management of antenatal hydronephrosis. Indian Pediatr, 2013. 50(2): p. 215-231.
Lei, T., et al., Medical Image Segmentation Using Deep Learning: A Survey. 2020. arXiv:2009.13120.
Milletari, F., Navab, N., Ahmadi, S.-A., V-Net: Fully Convolutional Neural Networks for Volumetric Medical Image Segmentation. 2016 Fourth International Conference on 3D Vision (3DV). 2016. pp. 565–571. https://doi.org/10.1109/3DV.2016.79
Kingma, D.P. and J. Ba, Adam: A Method for Stochastic Optimization. 2014, arXiv:1412.6980
Bahareh Behboodi, H.R., Ultrasound segmentation using U-Net: learning from simulated data and testing on real data. 2019, arXiv:1904.11031.
Chen, Y.-Q., et al., Three-dimensional ultrasound virtual organ computer-aided analysis technique in the assessment of function of diseased kidney in children with hydronephrosis. Chinese Journal of Medical Imaging Technology, 2013. 29(9): p. 1505-1508.
Otero, H.J., et al., Feasibility and Quality Determinants of 3D Sonography in Children With Hydronephrosis. Journal of diagnostic medical sonography, 2018. 34(1): p. 31-36.
Scanlan, K.A., Sonographic artifacts and their origins. AJR Am J Roentgenol, 1991. 156(6): p. 1267-1272.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lin, Y., Khong, PL., Zou, Z. et al. Evaluation of pediatric hydronephrosis using deep learning quantification of fluid-to-kidney-area ratio by ultrasonography. Abdom Radiol 46, 5229–5239 (2021). https://doi.org/10.1007/s00261-021-03201-w
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-021-03201-w