Abstract
Objective
The objective of this study was to explore whether volumetric apparent diffusion coefficient (ADC) histogram analysis can provide additional value to Vesical Imaging Reporting and Data System (VI-RADS) in differentiating muscle-invasive bladder cancer (MIBC) from non-muscle-invasive bladder cancer (NMIBC).
Materials and methods
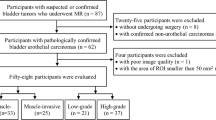
80 patients were retrospectively reviewed with pathologically proven NMIBC (n = 53) or MIBC (n = 27). All patients underwent MRI including diffusion-weighted imaging (DWI) (b = 0, 800 s/mm2), and the VI-RADS score was evaluated based on DWI. Volumetric ADC histogram parameters were calculated from the volumetric of interest (VOI) on DWI, including the min ADC, mean ADC, median ADC, max ADC, 10th, 25th, 75th, 90th percentiles ADC, skewness, kurtosis, and entropy. The Mann–Whitney U-test was used to compare histogram parameters between NMIBC and MIBC. Receiver operating characteristic analysis was used to evaluate the diagnostic value of each significant parameter.
Results
Among all parameters, the VI-RADS yield the highest Area Under the Curve (AUC, 0.88; sensitivity, 88.89%; specificity, 83.61%). MIBC had significantly lower min ADC, mean ADC, median ADC, 10th, 25th, 75th, and 90th percentiles ADC than NMIBC (p = 0.002, p < 0.001, p < 0.001, p = 0.003, p = 0.004, p < 0.001, p < 0.001). Skewness and kurtosis of MIBC were significantly higher than those of NMIBC (p < 0.001, p < 0.001). The combination of VI-RADS and skewness showed significantly higher AUC (AUC 0.923; 95% CI 0.847–0.969) than only with VI-RADS (AUC 0.880; 95% CI 0.793–0.940).
Conclusion
Volumetric ADC histogram analysis and VI-RADS are both useful methods in differentiating MIBC from NMIBC, and the volumetric ADC histogram analysis can provide additional value to VI-RADS.






Similar content being viewed by others
References
Antoni S, et al. Bladder Cancer Incidence and Mortality: A Global Overview and Recent Trends. Eur Urol, 2017. 71(1): p. 96-108.
Cumberbatch MGK, et al. Epidemiology of Bladder Cancer: A Systematic Review and Contemporary Update of Risk Factors in 2018. Eur Urol, 2018. 74(6): p. 784-795.
Gregg JR, et al. Guideline-based management of non-muscle invasive bladder cancer. Indian journal of urology : IJU : journal of the Urological Society of India, 2015. 31(4): p. 320-326.
Jordan B, Meeks JJ. T1 bladder cancer: current considerations for diagnosis and management. Nat Rev Urol, 2019. 16(1): p. 23-34.
Babjuk M, et al. EAU Guidelines on Non-Muscle-invasive Urothelial Carcinoma of the Bladder: Update 2016. Eur Urol, 2017. 71(3): p. 447-461.
Babjuk M, et al. European Association of Urology Guidelines on Non-muscle-invasive Bladder Cancer (TaT1 and Carcinoma In Situ) - 2019 Update. Eur Urol, 2019. 76(5): p. 639-657.
Wang H, et al. Radiomics analysis of multiparametric MRI for the preoperative evaluation of pathological grade in bladder cancer tumors. European radiology, 2019. 29(11): p. 6182-6190.
Yoshida S, et al. DWI as an Imaging Biomarker for Bladder Cancer. AJR American journal of roentgenology, 2017. 208(6): p. 1218-1228.
Takeuchi M, et al. MR imaging of urinary bladder cancer for T-staging: a review and a pictorial essay of diffusion-weighted imaging. Journal of magnetic resonance imaging : JMRI, 2013. 38(6): p. 1299-1309.
Zhao Q, et al. Differentiation between idiopathic granulomatous mastitis and invasive breast carcinoma, both presenting with non-mass enhancement without rim-enhanced masses: The value of whole-lesion histogram and texture analysis using apparent diffusion coefficient. European journal of radiology, 2020. 123: p. 108782.
Zou X, et al. Volumetric Apparent Diffusion Coefficient Histogram Analysis in Differentiating Intrahepatic Mass-Forming Cholangiocarcinoma From Hepatocellular Carcinoma. Journal of magnetic resonance imaging : JMRI, 2019. 49(4): p. 975-983.
Kondo M, Uchiyama Y. Apparent diffusion coefficient histogram analysis for prediction of prognosis in glioblastoma. Journal of neuroradiology = Journal de neuroradiologie, 2018. 45(4): p. 236–241.
Lim CS, et al. Use of Quantitative T2-Weighted and Apparent Diffusion Coefficient Texture Features of Bladder Cancer and Extravesical Fat for Local Tumor Staging After Transurethral Resection. AJR American journal of roentgenology, 2019. p. 1–10.
Panebianco V, et al. An evaluation of morphological and functional multi-parametric MRI sequences in classifying non-muscle and muscle invasive bladder cancer. European radiology, 2017. 27(9): p. 3759-3766.
Woo S, et al. Diagnostic Performance of Vesical Imaging Reporting and Data System for the Prediction of Muscle-invasive Bladder Cancer: A Systematic Review and Meta-analysis. European urology oncology, 2020. 3(3): p. 306-315.
Del Giudice F, et al. Systematic Review and Meta-Analysis of Vesical Imaging-Reporting and Data System (VI-RADS) Inter-Observer Reliability: An Added Value for Muscle Invasive Bladder Cancer Detection. Cancers, 2020. 12(10): p.
Takeuchi M, et al. Urinary bladder cancer: diffusion-weighted MR imaging--accuracy for diagnosing T stage and estimating histologic grade. Radiology, 2009. 251(1): p. 112-121.
Ueno Y, et al. Diagnostic Accuracy and Interobserver Agreement for the Vesical Imaging-Reporting and Data System for Muscle-invasive Bladder Cancer: A Multireader Validation Study. Eur Urol, 2019. 76(1): p. 54-56.
Wang H, et al. Multiparametric MRI for Bladder Cancer: Validation of VI-RADS for the Detection of Detrusor Muscle Invasion. Radiology, 2019. 291(3): p. 668-674.
Panebianco V, et al. Multiparametric Magnetic Resonance Imaging for Bladder Cancer: Development of VI-RADS (Vesical Imaging-Reporting And Data System). Eur Urol, 2018. 74(3): p. 294-306.
Yajima S, et al. Usefulness of the inchworm sign on DWI for predicting pT1 bladder cancer progression. European radiology, 2019. 29(7): p. 3881-3888.
Arévalo N, et al. "Inchworm sign" in urinary bladder cancer. Abdominal radiology (New York), 2018. 43(12): p. 3509-3510.
Li H, et al. Quantitative Assessment of Bladder Cancer Reflects Grade and Recurrence: Comparing of Three Methods of Positioning Region of Interest for ADC Measurements at Diffusion-weighted MR Imaging. Academic radiology, 2019. 26(9): p. 1148-1153.
Rosenkrantz AB, et al. Whole-lesion diffusion metrics for assessment of bladder cancer aggressiveness. Abdominal imaging, 2015. 40(2): p. 327-332.
Kobayashi S, et al. Diagnostic performance of diffusion-weighted magnetic resonance imaging in bladder cancer: potential utility of apparent diffusion coefficient values as a biomarker to predict clinical aggressiveness. European radiology, 2011. 21(10): p. 2178-2186.
Kobayashi S, et al. Apparent diffusion coefficient value reflects invasive and proliferative potential of bladder cancer. Journal of magnetic resonance imaging : JMRI, 2014. 39(1): p. 172-178.
Tang L, Zhou XJ. Diffusion MRI of cancer: From low to high b-values. Journal of magnetic resonance imaging : JMRI, 2019. 49(1): p. 23-40.
Suo ST, et al. Histogram analysis of apparent diffusion coefficient at 3.0 T in urinary bladder lesions: correlation with pathologic findings. Academic radiology, 2014. 21(8): p. 1027–1034.
Baek HJ, et al. Percent change of perfusion skewness and kurtosis: a potential imaging biomarker for early treatment response in patients with newly diagnosed glioblastomas. Radiology, 2012. 264(3): p. 834-843.
O'Brien T, et al. Different angiogenic pathways characterize superficial and invasive bladder cancer. Cancer research, 1995. 55(3): p. 510-513.
Huang L, et al. The Diagnostic Value of MR Imaging in Differentiating T Staging of Bladder Cancer: A Meta-Analysis. Radiology, 2018. 286(2): p. 502-511.
Zhang X, et al. Radiomics assessment of bladder cancer grade using texture features from diffusion-weighted imaging. Journal of magnetic resonance imaging : JMRI, 2017. 46(5): p. 1281-1288.
Xu S, et al. Combining DWI radiomics features with transurethral resection promotes the differentiation between muscle-invasive bladder cancer and non-muscle-invasive bladder cancer. European radiology, 2020. 30(3): p. 1804-1812.
Wang F, et al. Diffusion kurtosis imaging to assess correlations with clinicopathologic factors for bladder cancer: a comparison between the multi-b value method and the tensor method. European radiology, 2019. 29(8): p. 4447-4455.
Wang Y, et al. Comparison of the Diagnostic Value of Monoexponential, Biexponential, and Stretched Exponential Diffusion-weighted MRI in Differentiating Tumor Stage and Histological Grade of Bladder Cancer. Academic radiology, 2019. 26(2): p. 239-246.
Acknowledgements
This study is supported by the grants from National Natural Science Foundation of China (NSFC) No. 81771801, 82071889, 81801695, 81701657, and 82071890.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, S., Liang, P., Wang, Y. et al. Combining volumetric apparent diffusion coefficient histogram analysis with vesical imaging reporting and data system to predict the muscle invasion of bladder cancer. Abdom Radiol 46, 4301–4310 (2021). https://doi.org/10.1007/s00261-021-03091-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-021-03091-y