Abstract
Purpose
To compare the tumor size measurements assessed by computed tomography (CT) and magnetic resonance imaging (MRI) versus measurements of resected pathologic specimens from patients with pancreatic ductal adenocarcinoma (PDAC).
Methods
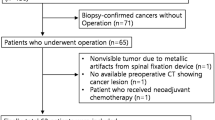
This study included 114 patients with histologically confirmed PDAC who underwent contrast-enhanced CT and MRI before surgery. The tumor sizes from CT, MRI, and pathologic specimens were compared by using Bland–Altman analyses and intraclass correlation coefficients (ICCs). The discrepancies in PDAC size between CT/MRI and pathologic specimens were calculated and contributing factors for the discrepancies, including tumor locations (pancreatic head/neck, body, or tail), T stages (T1, T2, or T3), and N stages (N0, N1, or N2), were analyzed with Pearson’s correlation coefficients and multivariable linear regression analyses.
Results
There was significant difference among the mean tumor sizes of three measurements (P < 0.001). The difference in mean tumor size between the pathologic sizes for PDAC was 4.3 mm (ICC 0.67) on CT and 5.8 mm (ICC 0.65) on MRI. Both CT and MRI showed wide ranges of limits of agreement (LOAs) between the pathologic specimens for tumor size measurements (LOAs, − 28.9 to 21.4 and − 29.4 to 17.8, respectively). The tumor size on CT or MRI was estimated to be smaller than that on pathology when the tumor was > 30 mm. The discrepancies in the tumor size estimated between CT/MRI and pathologic specimens were significantly different for tumors of different T stages (P < 0.001).
Conclusions
Both contrast-enhanced CT and MRI underestimate the mean tumor size by 4.3 mm and 5.8 mm, respectively, compared to the size of pathologic specimens. A larger tumor size indicates a greater discrepancy in the PDAC size measurements between CT/MRI and pathologic specimens.







Similar content being viewed by others
Abbreviations
- CT:
-
Computed tomography
- MRI:
-
Magnetic resonance imaging
- PDAC:
-
Pancreatic ductal adenocarcinoma
- ICC:
-
Intraclass correlation coefficient
- LOA:
-
Limits of agreement
- EUS:
-
Endoscopic ultrasonography
- PET:
-
Positron emission tomography
- MDCT:
-
Multi-detector CT
- AJCC:
-
American Joint Committee on Cancer
- TNM:
-
Tumor, node, and metastases
- RT:
-
Radiation therapy
- T2WI:
-
T2-weighted imaging
- DWI:
-
Diffusion-weighted magnetic resonance imaging
- T1WI:
-
T1-weighted imaging
- RECIST:
-
Response evaluation criteria in solid tumors
References
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2018; 68:394-424.
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2018. CA Cancer J Clin 2018; 68: 7-30.
Rahib L, Smith BD, Aizenberg R, Rosenzweig AB, Fleshman JM, Matrisian LM. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res 2014;74:2913-2921.
McGuigan A, Kelly P, Turkington RC, Jones C, Coleman HG, McCain RS. Pancreatic cancer: A review of clinical diagnosis, epidemiology, treatment and outcomes.World J Gastroenterol 2018;24:4846-4861.
Vaccaro V, Sperduti I, Milella M. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med 2011;365:768-769.
Lee ES, Lee JM. Imaging diagnosis of pancreatic cancer:A state-of-the-art review. World J Gastroenterol 2014;20:7864-7877.
Treadwell JR, Zafar HM, Mitchell MD, Tipton K, Teitelbaum U, Jue J. Imaging Tests for the Diagnosis and Staging of Pancreatic Adenocarcinoma: A Meta-Analysis. Pancreas 2016; 45: 789-795.
Amin MB, Edge SB, Greene F, Byrd DR, Brookland RK, Washington MK, Gershenwald JE, Compton CC, Hess KR, Sullivan DC, Jessup JM, Brierley JD, Gaspar LE, Schilsky RL, Balch CM, Winchester DP, Asar EA, Madera M, Gress DM, Meyer LR. AJCC Cancer Staging Manual. 8 ed. 2017, New Yor: Springer. 337-406.
Huguet F, Girard N, Guerche CS, Hennequin C, Mornex F, Azria D. Chemoradiotherapy in the management of locally advanced pancreatic carcinoma: a qualitative systematic review. J Clin Oncol 2009;27:2269-2277.
Arvold ND, Niemierko A, Mamon HJ, Fernandez-del Castillo C, Hong TS. Pancreatic cancer tumor size on CT scan versus pathologic specimen: implications for radiation treatment planning. Int J Radiat Oncol Biol Phys 2011;80:1383-1390.
Hall WA, Mikell JL, Mittal P, Colbert L, Prabhu RS, Kooby DA, Nickleach D, Hanley K, Sarmiento JM, Ali AN, Landry JC. Tumor Size on Abdominal MRI Versus Pathologic Specimen in Resected Pancreatic Adenocarcinoma: Implications for Radiation Treatment Planning. Int J Radiat Oncol Biol Phys 2013;86:102-107.
Legrand L, Duchatelle V, Molinié V, Boulay-Coletta I, Sibileau E, Zins M. Pancreatic adenocarcinoma: MRI conspicuity and pathologic correlations. Abdom Imaging 2015;40:85-94.
Kassardjian A, Stanzione N, Wang HL. Comparative Accuracy of Tumor Size Assessment and Stage Analysis by Imaging Modalities Versus Gross Examination for Pancreatic Ductal Adenocarcinoma. Pancreas 2019;48: 223-227.
Nishino M. Tumor Response Assessment for Precision Cancer Therapy: Response Evaluation Criteria in Solid Tumors and Beyond.Am Soc Clin Oncol Educ Book 2018;38:1019-1029.
Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986;1:307-310.
Cohen J. Weighted kappa: nominal scale agreement with provision for scaled disagreement or partial credit. Psychol Bull 1968;70:213-220.
Shrikhande SV, Barreto SG, Goel M, Arya S. Multimodality imaging of pancreatic ductal adenocarcinoma: a review of the literature. HPB 2012;14:658-668
Allen PJ, Kuk D, Castillo CF, Basturk O, Wolfgang CL, Cameron JL, Lillemoe KD, Ferrone CR, Morales-Oyarvide V, He J, Weiss MJ, Hruban RH, Gönen M, Klimstra DS, Mino-Kenudson M. Multi-institutional Validation Study of the American Joint Commission on Cancer (8th Edition) Changes for T and N Staging in Patients With Pancreatic Adenocarcinoma. Ann Surg 2017;265:185-191.
Kansu L, Aydın E, Akkaya H, Avcı S, Akalın N. Shrinkage of Nasal Mucosa and Cartilage During Formalin Fixation. Balkan Med J 2017;34:458-463.
Jonmarker S, Valdman A, Lindberg A, Hellström M, Egevad L. Tissue shrinkage after fixation with formalin injection of prostatectomy specimens. Virchows Arch 2006;449:297-301.
Funding
This study was funded by Grants from the Key junior college of national clinical of China, National Natural Science Foundation of China (81601468, 81701689), Project of precision medical transformation application of SMMU (2017JZ42), and Science and Technology Innovation Foundation of Shanghai (17411952200).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ma, C., Yang, P., Li, J. et al. Pancreatic adenocarcinoma: variability in measurements of tumor size among computed tomography, magnetic resonance imaging, and pathologic specimens. Abdom Radiol 45, 782–788 (2020). https://doi.org/10.1007/s00261-019-02125-w
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-019-02125-w