Abstract
Purpose
To assess the accuracy of unenhanced magnetic resonance imaging (MRI) immediately after the percutaneous ultrasound-guided radiofrequency ablation (RFA) of liver malignancy in predicting treatment efficacy at CT follow-up.
Materials and methods
Percutaneous ablation was prospectively performed in 23 liver malignancies (20 hepatocarcinomas and 3 metastases). After the procedure in the same day all patients were studied with unenhanced MRI. The best sequence to detect the coagulative necrosis was visually established. Pre-RFA CT and post-RFA MRI were registered with non-rigid transformation algorithm. Manual segmentation of lesions and ablated areas in pre-RFA CT, post-RFA MRI, and follow-up CT were obtained. Sensitivity, specificity, positive predictive value (PPV), negative predicitve value (NPV), and accuracy of MRI in predicting the correct centering and the complete treatment of the lesion were calculated in respect to the 1-month follow-up CT.
Results
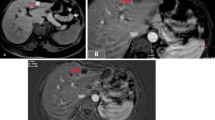
Fat-saturated T1-weighted (fs T1-w) was the sequence in which the best conspicuity of the ablated area was depicted. Coagulative necrosis was hyperintense in fs T1-w sequence in 17/23 (74%). In respect to follow-up CT, MRI predicted the correct centering of the lesions in 19/20 lesions with sensitivity, specificity, PPV, NPV, and accuracy of 100%, 75%, 95%, 100%, and 100%, respectively. MRI predicted the complete treatment of the lesions in 17/17 lesions with sensitivity, specificity, PPV, NPV, and accuracy of 100%.
Conclusion
MRI with the single fs T1-w sequence was highly accurate in predicting the treatment efficacy of percutaneous ablation of liver malignancies in comparison to follow-up CT control. Unnecessary CT in case of incomplete treatment can be therefore easily avoided.




Similar content being viewed by others
References
Giannini EG, Farinati F, Del Poggio P, et al. (2012) Ten-year outcome of radiofrequency thermal ablation for hepatocellular carcinoma: an Italian experience. Am J Gastroenterol 107:1588–1589. doi:10.1038/ajg.2012.250
Shiina S, Tateishi R, Arano T, et al. (2012) Radiofrequency ablation for hepatocellular carcinoma: 10-year outcome and prognostic factors. Am J Gastroenterol 107:569–577. doi:10.1038/ajg.2011.425
Livraghi T, Meloni F, Di Stasi M, et al. (2008) Sustained complete response and complications rates after radiofrequency ablation of very early hepatocellular carcinoma in cirrhosis: is resection still the treatment of choice? Hepatology 47:82–89. doi:10.1002/hep.21933
Livraghi T, Solbiati L, Meloni MF, et al. (2003) Treatment of focal liver tumors with percutaneous radio-frequency ablation: complications encountered in a multicenter study. Radiology 226:441–451. doi:10.1148/radiol.2262012198
Rhim H, Yoon K-H, Lee JM, et al. (2003) Major complications after radio-frequency thermal ablation of hepatic tumors: spectrum of imaging findings. Radiographics 23:123–126. doi:10.1148/rg.231025054
Ahmed M, Solbiati L, Brace CL, et al. (2014) Image-guided tumor ablation: standardization of terminology and reporting criteria—A 10-year update. Radiology 273(1):241–260. doi:10.1148/radiol.14132958
Kierans AS, Elazzazi M, Braga L, et al. (2010) Thermoablative treatments for malignant liver lesions: 10-Year experience of MRI appearances of treatment response. Am J Roentgenol 194:523–529. doi:10.2214/AJR.09.2621
Sainani NI, Gervais DA, Mueller PR, Arellano RS (2013) Imaging after percutaneous radiofrequency ablation of hepatic tumors: part 1, normal findings. Am J Roentgenol 200:184–193. doi:10.2214/AJR.12.8478
Guibal A, Bertin C, Egels S, et al. (2013) Contrast-enhanced ultrasound (CEUS) follow-up after radiofrequency ablation or cryoablation of focal liver lesions: Treated-area patterns and their changes over time. Eur Radiol 23:1392–1400. doi:10.1007/s00330-012-2702-0
Lekht I, Gulati M, Nayyar M, et al. (2016) Role of contrast-enhanced ultrasound (CEUS) in evaluation of thermal ablation zone. Abdom Radiol . doi:10.1007/s00261-016-0700-4
Catalano O, Izzo F, Vallone P, et al. (2015) Integrating contrast-enhanced sonography in the follow-up algorithm of hepatocellular carcinoma treated with radiofrequency ablation: single cancer center experience. Acta Radiol 56:133–142. doi:10.1177/0284185114521108
Rempp H, Unterberg J, Hoffmann R, et al. (2013) Therapy monitoring of magnetic resonance-guided radiofrequency ablation using T1- and T2-weighted sequences at 1.5 T: reliability of estimated ablation zones. Invest Radiol 48(6):429–436. doi:10.1097/RLI.0b013e318280b007
Fedorov A, Beichel R, Kalphaty-Cramer J, et al. (2012) 3D slicers as an image computing platform for thw quantitative imaging network. Magn Reson Imaging 30:1323–1341. doi:10.1016/j.mri.2012.05.001.3D
Dromain C, de Baere T, Elias D, et al. (2002) Hepatic tumors treated with percutaneous radio-frequency ablation: CT and MR imaging follow-up. Radiology 223:255–262. doi:10.1148/radiol.2231010780
Jacobs A (2015) Radiofrequency ablation for liver cancer. Radiol Technol 86:645–664
Marin D, Cappabianca S, Serra N, et al. (2015) CT appearance of hepatocellular carcinoma after locoregional treatments: a comprehensive review. Gastroenterol Res Pract 2015:1–10. doi:10.1155/2015/670965
Sainani NI, Gervais DA, Mueller PR, Arellano RS (2013) Imaging after percutaneous radiofrequency ablation of hepatic tumors: Part 2, abnormal findings. Am J Roentgenol 200:194–204. doi:10.2214/AJR.12.8479
Koda M, Tokunaga S, Okamoto T, et al. (2015) Clinical usefulness of the ablative margin assessed by magnetic resonance imaging with Gd-EOB-DTPA for radiofrequency ablation of hepatocellular carcinoma. J Hepatol 63:1360–1367. doi:10.1016/j.jhep.2015.07.023
Maruyama H, Matsutani S, Saisho H, et al. (2005) Real-time blood-pool images of contrast enhanced ultrasound with definity in the detection of tumour nodules in the liver. Br J Radiol 78:512–518. doi:10.1259/bjr/59648297
Kim TK, Khalili K, Jang H-J (2015) Local ablation therapy with contrast-enhanced ultrasonography for hepatocellular carcinoma: a practical review. Ultrasonography 34:235–245
Vogt FM, Antoch G, Veit P, et al. (2007) Morphologic and functional changes in nontumorous liver tissue after radiofrequency ablation in an in vivo model: comparison of 18F-FDG PET/CT, MRI, ultrasound, and CT. J Nucl Med 48:1836–1844. doi:10.2967/jnumed.107.042846
Burns SK, Dodd GD 3rd, McManus LM, et al. (2012) 3T magnetic resonance imaging accurately depicts radiofrequency ablation zones in a blood-perfused bovine liver model. J Vasc Interv Radiol 23(6):801–808. doi:10.1016/j.jvir.2012.01.076
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
The present study did not receive any funding support.
Conflict of interest
All the authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Research involving human participants and/or animals
This article does not contain any studies with animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
D’Onofrio, M., Cardobi, N., Ruzzenente, A. et al. Unenhanced magnetic resonance imaging immediately after radiofrequency ablation of liver malignancy: preliminary results. Abdom Radiol 43, 1379–1385 (2018). https://doi.org/10.1007/s00261-017-1299-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-017-1299-9