Abstract
Purpose
To determine the feasibility of using blood oxygenation level-dependent (BOLD) MRI in the characterization of hepatic tumors.
Methods
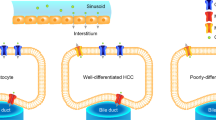
A total of 100 patients with 43 hepatocellular carcinomas (HCCs), 36 metastases, 17 cholangiocarcinomas, and 23 hemangiomas underwent gadoxetic acid-enhanced and BOLD MRI at 3T. BOLD MRI was performed using a multiple fast-field echo sequence (TR/TE, 290/10–28; slice thickness 5 mm) to generate 20 T2*-weighted images. The T2* value of each tumor were calculated. On a color-coded T2* map, tumors were classified into five categories of high signal intensity (strong, moderate, rim, mild) and iso-intensity, which was correlated with the enhancement pattern on dynamic phases by two observers.
Results
The mean T2* value (ms) of hemangiomas (97.3 ± 20.2) was the highest, followed by HCCs (48.4 ± 12.7), metastases (37.1 ± 10.5), and cholangiocarcinomas (36.6 ± 11.1). These values were significantly different (hemangioma vs. others tumors and HCC vs. metastasis or cholangiocarcinoma) (P ≤ 0.001). The agreement between the T2* color map and dynamic images was moderate for all tumors (k = 0.544), good for tumors >2.0 cm (k = 0.666), and fair for tumors ≤2.0 cm (k = 0.334). With the gadoxetic acid-enhanced MRI used as a reference, the sensitivities of BOLD MRI (T2* color map) for displaying hypervascularity of HCC (categories of 1–3) were 81.0 % (n = 34/42) and 78.6 % (n = 33/42) for both observers.
Conclusion
Liver BOLD MRI has a potential to predict the vascular pattern of hepatic tumors.





Similar content being viewed by others
References
Bruix J, Sherman A, American Association for the Study of Liver Disease (2011) Management of hepatocellular carcinoma: an update. Hepatology 53:1020–1021
Tan CH, Low SC, Thng CH (2011) APASL and AASLD Consensus Guidelines on imaging diagnosis of hepatocellular carcinoma: a review. Int J Hepatol 2011:519783
Sadowski EA, Bennett LK, Chan MR, et al. (2007) Nephrogenic systemic fibrosis: risk factors and incidence estimation. Radiology 243:148–157
Fazel R, Krumholz HM, Wang Y, et al. (2009) Exposure to low-dose ionizing radiation from medical imaging procedures. N Engl J Med 361:849–857
Gleeson TG, Bulugahapitiya S (2004) Contrast-induced nephropathy. AJR 183:1673–1689
Vivier PH, Storey P, Chandarana H, et al. (2013) Renal blood oxygenation level-dependent imaging: contribution of r2 to r2* values. Invest Radiol 48:501–508
Rakow-Penner R, Daniel B, Glover GH (2010) Detecting blood oxygen level dependent (BOLD) contrast in the breast. J Magn Reson Imaging 32:120–129
McPhail LD, Robinson SP (2010) Intrinsic susceptibility MR imaging of chemically induced rat mammary tumors: relationship to histological assessment of hypoxia and fibrosis. Radiology 254:110–118
Sadowski EA, Fain SB, Alford SK, et al. (2005) Assessment of acute renal transplant rejection with blood oxygen level-dependent MR imaging: initial experience. Radiology 236:911–919
Prielmeier F, Nagatomo Y, Frahm J (1994) Cerebral blood oxygenation in rat brain during hypoxic hypoxia: quantitative MRI of effective transverse relaxation rates. Magn Reson Med 31:678–681
Boxerman JL, Hamberg LM, Rosen BR, Weisskoff RM (1995) MR contrast due to intravascular magnetic susceptibility perturbations. Magn Reson Med 34:555–566
Dennie J, Mandeville JB, Boxerman JL, et al. (1998) NMR imaging of changes in vascular morphology due to tumor angiogenesis. Magn Reson Med 40:793–799
Hoskin PJ, Carnell DM, Taylor NJ, et al. (2007) Hypoxia in prostate cancer: correlation of BOLD MRI with pimonidazole immunohistochemistry—initial observations. Int J Radiat Oncol Biol Phys 68:1065–1071
Padhani AR, Krohn KA, Lewis JS, Alber M (2007) Imaging oxygenation of human tumours. Eur Radiol 17:861–872
Christen T, Lemasson B, Pannetier N, et al. (2012) Is T2* enough to assess oxygenation? Quantitative blood oxygen level-dependent analysis in brain tumor. Radiology 262:495–502
O’Connor JP, Naish JH, Parker GJ, et al. (2009) Preliminary study of oxygen-enhanced longitudinal relaxation in MRI: a potential novel biomarker of oxygenation changes in solid tumors. Int J Radiat Oncol Biol Phys 75:1209–1215
Rhee TK, Larson AC, Prasad PV, et al. (2005) Feasibility of blood oxygenation level-dependent MR imaging to monitor hepatic transcatheter arterial embolization in rabbits. J Vasc Interv Radiol 16:1523–1528
Min JH, Kim CK, Park BK, Kim E, Kim B (2011) Assessment of renal lesions with blood oxygenation level-dependent MRI at 3T: preliminary experience. AJR 197:489–494
Fleiss JL, Levin B, Paik MC (2003) Statistical methods for rates and proportions, 3rd edn. New York: Wiley, pp 222–225
Padhani A (2010) Science to practice: what does MR oxygenation imaging tell us about human breast cancer hypoxia? Radiology 254:1–3
Howe FA, Robinson SP, McINtyre DJ, et al. (2001) Issues in flow and oxygenation dependent contrast (FLOOD) imaging of tumours. NMR Biomed 14:497–506
Alonzi R, Padhani AR, Maxwell RJ, et al. (2009) Carbogen breathing increases prostate cancer oxygenation: a translational MRI study in murine xenografts and humans. Br J Cancer 100:644–648
Jhaveri KS, Cleary SP, Fischer S, et al. (2013) Blood oxygen level-dependent liver MRI: can it predict microvascular invasion in HCC? J Magn Reson Imaging 37:692–699
Park MS, Kim S, Patel J, et al. (2012) Hepatocellular carcinoma: detection with diffusion-weighted versus contrast-enhanced magnetic resonance imaging in pretransplant patients. Hepatology 56:140–148
Park MJ, Kim YK, Lee MW, et al. (2012) Small hepatocellular carcinomas: improved sensitivity by combining gadoxetic acid-enhanced and diffusion-weighted MR imaging patterns. Radiology 264:761–770
Chong YS, Kim YK, Lee MW, et al. (2012) Differentiating mass-forming intrahepatic cholangiocarcinoma from atypical hepatocellular carcinoma using gadoxetic acid-enhanced MRI. Clin Radiol 67:766–773
Kido A, Koyama T, Kataoka M, et al. (2007) Physiological changes of the human uterine myometrium during menstrual cycle: preliminary evaluation using BOLD MR imaging. J Magn Reson Imaging 26:695–700
Conflict of interest
All the authors had no relevant financial interests or personal affiliations in connection with the content of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Park, H.J., Kim, Y.K., Min, J.H. et al. Feasibility of blood oxygenation level-dependent MRI at 3T in the characterization of hepatic tumors. Abdom Imaging 39, 142–152 (2014). https://doi.org/10.1007/s00261-013-0044-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00261-013-0044-2