Abstract
Purpose
In our study, our aim was to investigate the role of [68 Ga]Ga-PSMA-11 PET /CT imaging in the diagnosis of clinically significant prostate cancer (csPCa) (ISUP GG 2 and higher) in patients initially diagnosed with ISUP GG 1 and 2 after prostate biopsy.
Materials and methods
We retrospectively reviewed 147 patient records in whom [68 Ga]Ga-PSMA-11 PET/CT imaging was performed preoperatively. All patients were initially diagnosed with ISUP GG 1 and 2 PCa by biopsy. Final pathology reports were obtained after radical prostatectomy. The [68 Ga]Ga-PSMA-11 PET/CT images were evaluated to determine the PRIMARY score. Patients’ mpMRI-PIRADS scores were also recorded when available and analyzed in correlation with the pathology results.
Results
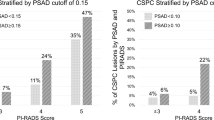
For the 114 patients scored using PRIMARY, 19 out of 37 patients with scores of 1 and 2 (51%) were diagnosed with csPCa. Of the 77 patients with PRIMARY scores between 3 and 5, 64 (83%) had csPCa. Notably, every patient with a PRIMARY score of 5 had csPCa. PRIMARY scoring had a sensitivity of 77% and specificity of 58%, with a positive predictive value of 83%. A moderate correlation was observed between PRIMARY scores and ISUP GG (Rho = 0.54, p < 0.001). In contrast, the PIRADS score displayed a sensitivity and specificity of 86% and 25% respectively, with a positive predictive value of 68%. No substantial correlation was found between PIRADS and ISUP GG. Statistical analysis revealed a significant correlation between PRIMARY and ISUP GG (p < 0.001), but not between PIRADS and ISUP GG (p = 0.281). Comparatively, PRIMARY scoring was significantly more reliable than PIRADS scoring in identifying csPCa.
Conclusion
[68 Ga]Ga-PSMA-11 PET/CT imaging is promising for distinguishing high-risk prostate cancer patients from those apt for active surveillance, potentially aiding in the identification of csPCa.

Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Hugosson J, Roobol MJ, Mansson M, Tammela TLJ, Zappa M, Nelen V, et al. A 16-yr follow-up of the European randomized study of screening for prostate cancer. Eur Urol. 2019;76(1):43–51.
Mottet N, van den Bergh RCN, Briers E, Van Den Broeck T, Cumberbatch MG, De Santis M, et al. EAU-EANM-ESTRO-ESUR-SIOG Guidelines on prostate cancer-2020 update Part 1: screening, diagnosis, and local treatment with curative intent. Eur Urol. 2021;79(2):243–62.
van Leenders G, van der Kwast TH, Grignon DJ, Evans AJ, Kristiansen G, Kweldam CF, et al. The 2019 International Society of Urological Pathology (ISUP GG) Consensus Conference on Grading of Prostatic Carcinoma. Am J Surg Pathol. 2020;44(8):e87–99.
Suardi N, Briganti A, Gallina A, et al. Testing the most stringent criteria for selection of candidates for active surveillance in patients with low-risk prostate cancer. BJU Int. 2010;105(11):1548–52.
Djavan B, Kadesky K, Klopukh B, Marberger M, Roehrborn CG. Gleason scores from prostate biopsies obtained with 18-gauge biopsy needles poorly predict Gleason scores of radical prostatectomy specimens. Eur Urol. 1998;33(3):261–70.
Demirci E, Kabasakal L, Şahin OE, Akgün E, Güntekin MH, Doğancaa T, et al. Can SUVmax values of Ga-68-PSMA PET/CT scan predict the clinically significant prostate cancer? Nucl Med Commun. 2019;40(1):86–91.
Ahmed HU, Bosaily AES, Brown LC, et al. Diagnostic accuracy of multi-parametric MRI and TRUS biopsy in prostate cancer (PROMIS): a paired validating confirmatory study. Lancet. 2017;389(10071):815–22.
Borofsky S, George AK, Gaur S, Bernardo M, Greer MD, Mertan FV, et al. What are we missing? False-negative cancers at multiparametric MR imaging of the prostate. Radiology. 2018;286(1):186–95.
Drost FH, Osses DF, Nieboer D, Steyerberg EW, Bangma CH, Roobol MJ, Schoots IG. Prostate MRI, with or without MRI-targeted biopsy, and systematic biopsy for detecting prostate cancer. Cochrane Database Syst Rev. 2019;4(4):CDO12663.
Gultekin MH, Demirci E, Turegun FA, et al. The role of 68GA-PSMA PET/CT scan in patients with prostate adenocarcinoma who underwent radical prostatectomy. Urol J. 2020;18(1):58–65.
Kabasakal L, Demirci E, Nematyazar J, Akyel R, Razavi B, Ocak M. The role of PSMA PET/CT imaging in restaging of prostate cancer patients with low prostate-specific antigen. Nucl Med Commun. 2017;38(2):149–55.
Fendler WP, Eiber M, Behesthi M, Bomanji J, Calais J, Ceci F, et al. PSMA PET/CT: Joint EANM procedure guideline/SNMMI procedure standard for prostate cancer imaging: version 2.0. Eur J Nucl Med Mol Imaging. 2023;50(5):1456–86.
Hupe MC, Philippi C, Roth D, Kümpers C, Ribbat-İdel J, Becker F, et al. Expression of prostate-specific membrane antigen (PSMA) on biopsies is an independent risk stratifier of prostate cancer patients at time of initial diagnosis. Front Oncol. 2018;20(8):623.
Uprimny C, Kroiss AS, Decristoforo C, et al. (68)Ga-PSMA-11 PET/CT in primary staging of prostate cancer: PSA and Gleason score predict the intensity of tracer accumulation in the primary tumour. Eur J Nucl Med Mol Imaging. 2017;44(6):941–9.
Raveenthiran S, Yaxley WJ, Franklin T, Coughlin G, Roberts M, Gianduzzo T, et al. Findings in 1,123 men with preoperative (68)Ga-prostate-specific membrane antigen positron emission tomography/computerized tomography and multiparametric magnetic resonance imaging compared to totally embedded radical prostatectomy histopathology: implications for the diagnosis and management of prostate cancer. J Urol. 2022;207(3):573–80.
Emmett L, Papa N, Buteau J, Ho B, Liu V, Roberts M, et al. The PRIMARY score: using intraprostatic 68Ga-PSMA PET/CT patterns to optimize prostate cancer diagnosis. J Nucl Med. 2022;63(11):1644–50.
Koole M, Armstrong I, Krizsan AK, Stromvall A, Visvikis D, Sattler B, et al. EANM guidelines for PET-CT and PET-MR routine quality control. Z Med Phys. 2023;33(1):103–13.
Kalapara AA, Ballok ZE, Ramdave S, O’Sullivan R, Ryan A, Konety B, et al. Combined utility of 68Ga-prostate-specific membrane antigen positron emission tomography/computed tomography and multiparametric magnetic resonance imaging in predicting prostate biopsy pathology. Eur Urol Oncol. 2022;5(3):314–20.
Cheng C, Liu J, Yi X, Yin H, Qiu D, Zhang J, et al. Prediction of clinically significant prostate cancer using a novel 68Ga-PSMA PET-CT and multiparametric MRI-based model. Transl Androl Urol. 2023;12(7):1115–26.
Pepe P, Roscigno M, Pepe L, Panella P, Tamburo M, Marletta G, et al. (2022) Could 68Ga-PSMA PET/CT evaluation reduce the number of scheduled prostate biopsies in men enrolled in active surveillance protocols? J Clin Med 11(12):3473.
Hu Q, Hong X, Xu L, Jia R. A nomogram for accurately predicting the pathological upgrading of prostate cancer, based on 68 Ga-PSMA PET/CT. Prostate. 2022;82(11):1077–87.
Emmett L, Buteau J, Papa N, et al. The additive diagnostic value of prostate-specific membrane antigen positron emission tomography computed tomography to multiparametric magnetic resonance imaging triage in the diagnosis of prostate cancer (PRIMARY): a prospective multicentre study. Eur Urol. 2021;80(6):682–9.
Meissner VH, Rauscher I, Schwamborn K, et al. Radical prostatectomy without prior biopsy following multiparametric magnetic resonance imaging and prostate-specific membrane antigen positron emission tomography. Eur Urol. 2022;82(2):156–60.
Willemse PPM, Davis NF, Grivas N, et al. Systematic review of active surveillance for clinically localised prostate cancer to develop recommendations regarding inclusion of intermediate-risk disease, biopsy characteristics at inclusion and monitoring, and surveillance repeat biopsy strategy. Eur Urol. 2022;81(4):337–46.
Stefanova V, Buckley R, Flax S, et al. Transperineal prostate biopsies using local anesthesia: experience with 1,287 patients. prostate cancer detection rate, complications and patient tolerability. J Urol. 2019;201(6):1121–6.
Barzell WE, Melamed MR, Cathcart P, Moore CM, Ahmed HU, Emberton M. Identifying candidates for active surveillance: an evaluation of the repeat biopsy strategy for men with favorable risk prostate cancer. J Urol. 2012;188(3):762–7.
Porten SP, Whitson JM, Cowan JE, Perez N, Shinohara K, Carroll PR. Changes in cancer volume in serial biopsies of men on active surveillance for early-stage prostate cancer. J Urol. 2011;186(5):1825–9.
Bokhorst LP, Alberts AR, Rannikko A, Valdagni R, Picles T, Kakehi Y, et al. Compliance rates with the prostate cancer research international active surveillance (PRIAS) protocol and disease reclassification in noncompliers. Eur Urol. 2015;68(5):814–21.
Moldovan PC, Van den Broeck T, Sylvester R, Marconi L, Bellmunt J, van den Bergh RCN, et al. What Is the negative predictive value of multiparametric magnetic resonance imaging in excluding prostate cancer at biopsy? A systematic review and meta-analysis from the European Association of Urology Prostate Cancer Guidelines Panel. Eur Urol. 2017;72(2):250–66.
Panebianco V, Barchetti G, Simone G, Del Monte M, Ciardi A, Grompone MD, et al. Negative multiparametric magnetic resonance imaging for prostate cancer: What’s next? Eur Urol. 2018;74(1):48–54.
Mazzone E, Stabile A, Pellegrino F, Basile G, Cignoli D, Cirulli GO, Sorce G, Barletta F, Scuderi S, Bravi CA, Cucchiara V, Fossati N, Gandaglia G, Montorsi F, Briganti A. Positive predictive value of prostate imaging reporting and data system version 2 for the detection of clinically significant prostate cancer: a systematic review and meta-analysis. Eur Urol Oncol. 2021;4(45):697–713.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by KA, AK, MD, GB, FA, and LK. The first draft of the manuscript was written by LK, KA, and GB, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
The study was carried out in accordance with the declaration of Helsinki. The study was approved by the local ethical committee (approval number: 666795).
Consent to participate
Written consent was obtained from all patients.
Consent for publication
There is not any patient personal data in this manuscript.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Akcay, K., Kibar, A., Sahin, O.E. et al. Prediction of clinically significant prostate cancer by [68 Ga]Ga-PSMA-11 PET/CT: a potential tool for selecting patients for active surveillance. Eur J Nucl Med Mol Imaging 51, 1467–1475 (2024). https://doi.org/10.1007/s00259-023-06556-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-023-06556-y