Abstract
Purpose
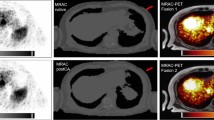
Recent technical advancements in PET imaging have improved sensitivity and spatial resolution. Consequently, clinical nuclear medicine will be confronted with PET images on a previously unfamiliar resolution. To better understand [18F]FDG distribution at submillimetric scale, a direct correlation of radionuclide-imaging and histopathology is required.
Methods
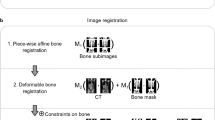
A total of five patients diagnosed with a malignancy of the head and neck were injected with a clinical activity of [18F]FDG before undergoing surgical resection. The resected specimen was imaged using a preclinical high-resolution PET/CT, followed by slicing of the specimen. Multiple slices were rescanned using a micro-PET/CT device, and one of the slices was snap-frozen for frozen sections. Frozen sections were placed on an autoradiographic film, followed by haematoxylin and eosin staining to prepare them for histopathological assessment. The results from both autoradiography and histopathology were co-registered using an iterative co-registration algorithm, and regions of interest were identified to study radiotracer uptake.
Results
The co-registration between the autoradiographs and their corresponding histopathology was successful in all specimens. The use of this novel methodology allowed direct comparison of autoradiography and histopathology and enabled the visualisation of uncharted heterogeneity in [18F]FDG uptake in both benign and malignant tissue.
Conclusion
We here describe a novel methodology enabling the direct co-registration of [18F]FDG autoradiography with the gold standard of histopathology in human malignant tissue. The future use of the current methodology could further increase our understanding of the distribution of radionuclides in surgically excised malignancies and hence, improve the integration of pathology and molecular imaging in a multiscale perspective.
Trial registration
ClinicalTrials.gov Identifier: NCT05068687







Similar content being viewed by others
References
Hess S, Blomberg BA, Zhu HJ, Høilund-Carlsen PF, Alavi A. The pivotal role of FDG-PET/CT in modern medicine. Acad Radiol. 2014;21:232–49. https://doi.org/10.1016/j.acra.2013.11.002.
Zhang X, Xie Z, Berg E, Judenhofer MS, Liu W, Xu T, et al. Total-body dynamic reconstruction and parametric imaging on the uexplorer. J Nucl Med. 2020;61:285–91. https://doi.org/10.2967/jnumed.119.230565.
Soriano A, González A, Orero A, Moliner L, Carles M, Sánchez F, et al. Attenuation correction without transmission scan for the MAMMI breast PET. Nucl Instruments Methods Phys Res Sect A Accel Spectrometers, Detect Assoc Equip. 2011;648:S75–8. https://doi.org/10.1016/j.nima.2010.12.138.
Krishnamoorthy S, Blankemeyer E, Mollet P, Surti S, Van Holen R, Karp JS. Performance evaluation of the MOLECUBES β-CUBE - A high spatial resolution and high sensitivity small animal PET scanner utilising monolithic LYSO scintillation detectors. Phys Med Biol. 2018;63:155013. https://doi.org/10.1088/1361-6560/aacec3.
Göker M, Marcinkowski R, Van Bockstal M, Keereman V, Van Holen R, Van Dorpe J, et al. 18F-FDG micro-PET/CT for intra-operative margin assessment during breast-conserving surgery. Acta Chir Belg. 2020;120:366–74. https://doi.org/10.1080/00015458.2020.1774163.
Debacker JM, Schelfhout V, Brochez L, Creytens D, D’asseler Y, Deron P, et al. High-resolution18 f-fdg pet/ct for assessing three-dimensional intraoperative margins status in malignancies of the head and neck, a proof-of-concept. J Clin Med. 2021;10:3737. https://doi.org/10.3390/jcm10163737.
Higashi K, Ueda Y, Sakurai A, MingWang X, Xu L, Murakami M, et al. Correlation of Glut-1 glucose transporter expression with [18F]FDG uptake in non-small cell lung cancer. Eur J Nucl Med. 2000;27:1778–85. https://doi.org/10.1007/s002590000367.
Brown RS, Leung JY, Fisher SJ, Frey KA, Ethier SP, Wahl RL. Intratumoral Distribution of tritiated-fdg in breast carcinoma: correlation between Glut-1 expression and FDG uptake. J Nucl Med. 1996;37:1042–7.
Meyer HJ, Wienke A, Surov A. Associations between GLUT expression and SUV values derived from FDG-PET in different tumors—a systematic review and meta analysis. PLoS One. 2019;14:e0217781. https://doi.org/10.1371/journal.pone.0217781.
Szablewski L. Expression of glucose transporters in cancers. Biochim Biophys Acta - Rev Cancer. 2013;1835:164–9. https://doi.org/10.1016/j.bbcan.2012.12.004.
Marstal K, Berendsen F, Staring M, Klein S. SimpleElastix: a user-friendly, multi-lingual library for medical image registration. 2016 IEEE conference on computer vision and pattern recognition workshops (CVPRW). 2016;574–82. https://doi.org/10.1109/CVPRW.2016.78.
Schneider CA, Rasband WS, Eliceiri KW. NIH Image to ImageJ: 25 years of image analysis. Nat Methods. 2012;9:671–5. https://doi.org/10.1038/nmeth.2089.
Bankhead P, Loughrey MB, Fernández JA, Dombrowski Y, McArt DG, Dunne PD, et al. QuPath: Open source software for digital pathology image analysis. Sci Rep. 2017;7:16878. https://doi.org/10.1038/s41598-017-17204-5.
Van de Sande L, Menekse G. Peri-operative 18F-FDG-PET-CT specimen imaging for margin assessment in breast malignancies: a proof-of-concept study. Eur J Surg Oncol. 2022;48:e87. https://doi.org/10.1016/j.ejso.2021.12.116.
Fanchon LM, Dogan S, Moreira AL, Carlin SA, Schmidtlein CR, Yorke E, et al. Feasibility of in situ, high-resolution correlation of tracer uptake with histopathology by quantitative autoradiography of biopsy specimens obtained under18F-FDG PET/CT guidance. J Nucl Med. 2015;56:538–44. https://doi.org/10.2967/jnumed.114.148668.
Gray BR, Koontz NA. Normal patterns and pitfalls of FDG Uptake in the head and neck. semin ultrasound. CT MRI. 2019;40:367–75. https://doi.org/10.1053/j.sult.2019.07.001.
Tian M, He X, Jin C, He X, Wu S, Zhou R, et al. Transpathology: molecular imaging-based pathology. Eur J Nucl Med Mol Imaging. 2021;48:2338–50. https://doi.org/10.1007/s00259-021-05234-1.
Kwee TC, Basu S, Saboury B, Ambrosini V, Torigian DA, Alavi A. A new dimension of FDG-PET interpretation: assessment of tumor biology. Eur J Nucl Med Mol Imaging. 2011;38:1158–70. https://doi.org/10.1007/s00259-010-1713-9.
Adams HJA, Kwee TC. Proportion of false-positive lesions at interim and end-of-treatment FDG-PET in lymphoma as determined by histology: systematic review and meta-analysis. Eur J Radiol. 2016;85:1963–70. https://doi.org/10.1016/j.ejrad.2016.08.011.
Jones M, Hruby G, Solomon M, Rutherford N, Martin J. The role of FDG-PET in the initial staging and response assessment of anal cancer: a systematic review and meta-analysis. Ann Surg Oncol. 2015;22:3574–81. https://doi.org/10.1245/s10434-015-4391-9.
Lamarca A, Barriuso J, Chander A, McNamara MG, Hubner RA, ÓReilly D, et al. 18F-fluorodeoxyglucose positron emission tomography (18FDG-PET) for patients with biliary tract cancer: Systematic review and meta-analysis. J Hepatol. 2019;71:115–29. https://doi.org/10.1016/j.jhep.2019.01.038.
Vandenberghe S, Moskal P, Karp JS. State of the art in total body PET. EJNMMI Phys. 2020;7:35. https://doi.org/10.1186/s40658-020-00290-2.
Stockhoff M, Decuyper M, Van Holen R, Vandenberghe S. High-resolution monolithic LYSO detector with 6-layer depth-ofinteraction for clinical PET. Phys Med Biol. 2021;66:155014. https://doi.org/10.1088/1361-6560/ac1459.
Grootendorst MR, Cariati M, Pinder SE, Kothari A, Douek M, Kovacs T, et al. Intraoperative assessment of tumor resection margins in breast-conserving surgery using 18F-FDG cerenkov luminescence imaging: A first-in-human feasibility study. J Nucl Med. 2017;58:891–8. https://doi.org/10.2967/jnumed.116.181032.
The Council of the European Union, Council Directive. 59/Euratom of 5 December 2013 laying down basic safety standards for protection against the dangers arising from exposure to ionising radiation, and repealing Directives 89/618/Euratom, 90/641/Euratom, 96/29/Euratom, 97/43/Euratom and 2003/122/Euratom. Off J Eur Union. 2013;2014:57. https://doi.org/10.3000/19770677.L_2014.013.eng.
de Boer LL, Kho E, Nijkamp J, Van de Vijver KK, Sterenborg HJCM, ter Beek LC, et al. Method for coregistration of optical measurements of breast tissue with histopathology: the importance of accounting for tissue deformations. J Biomed Opt. 2019;24:1. https://doi.org/10.1117/1.jbo.24.7.075002.
Acknowledgements
The authors would like to thank ir. Eva Kint for ensuring radioprotection during the course of the study, and dr. apr. Jeroen Verhoeven, PhD, and Dr. ir. Jens Mincke, PhD, for their assistance in performing the autoradiography of the samples.
Author information
Authors and Affiliations
Contributions
JD, VK, CV, & WH conceptualised the study; JD, LM, VK, DC, KVdV, & BD designed the study; JD, WH, & PhD screened patients; JD, WH, & PhD enrolled patients; JD collected the clinical data; JD, LM, FC, & CV performed image analysis; FC, DC, KVdV, & SL performed pathological analysis; JD & LM performed data analysis; JD drafted the paper. Writing - review and editing: JD, FC, LM, DC, PhD, BD, YD, KDM, VK, SL, VS, KVdV, CV, & WH; WH coordinated and supervised the project’s activities. All the authors critically revised the paper and approved the submitted version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
The committee for medical ethics of the Ghent University Hospital approved the initial study on August 26, 2019, with local authorisation code 2019/1135. The amendment for performing additional sectioning and autoradiography was approved January 21, 2021.
Consent to participate
All subjects included in the current trial have given written informed consent to participate.
Consent for publication
All subjects included in the current trial have given written informed consent regarding the publication of the study data.
Conflict of interest
Vincent Keereman is a shareholder and board member of XEOS Medical and Luna Maris is an employee of XEOS Medical. The other authors have no financial or personal relationship with other people or organisations that could inappropriately influence or bias the currently submitted work.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Oncology - Head and Neck.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Debacker, J., Maris, L., Cordier, F. et al. Direct co-registration of [18F]FDG uptake and histopathology in surgically excised malignancies of the head and neck: a feasibility study. Eur J Nucl Med Mol Imaging 50, 2127–2139 (2023). https://doi.org/10.1007/s00259-023-06153-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-023-06153-z