Abstract
Purpose
The recent introduction of integrated PET-MRI systems into practice seems promising in oncologic imaging, and efforts are made to specify their added values. The current study evaluates the added values of PET-MRI over PET-CT in detecting active malignant hepatic lesions.
Methods
As part of an ongoing prospective study in our institution that assesses the added values of PET-MRI, subjects undergo PET-CT and subsequent PET-MRI after single radiotracer injection. The current study included 97 pairs of whole-body PET-CT and liver PET-MRI scans, of 61 patients (19/61 had ≥ 2 paired scans), all performed with [18F]FDG and interpreted as showing active malignant hepatic involvement. Primary malignancies were of colorectal/biliary/pancreatic/breast/other origins in 19/9/9/7/17 patients. Monitoring response to therapy was the indication in 86/97 cases. When PET-MRI detected additional malignant lesions over PET-CT, lesions size, their characteristics on PET-MRI, and the influence on the final report were recorded.
Results
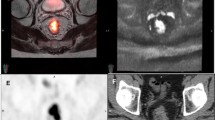
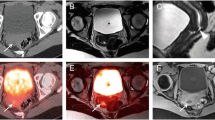
In 37/97 (38.1%) cases, a total of 78 malignant lesions were identified on PET-MRI but not on PET-CT: 19 lesions (11 cases) were identified on PET of PET-MRI but not on PET of PET-CT; 37 lesions (14 cases) were small (≤ 0.8 cm) and identified on MRI only; 22 lesions (12 cases) were > 0.8 cm, had low/no [18F]FDG uptake, but were categorized as viable based on MRI. These 78 lesions caused major effect on final reports in 11/97 (11.3%) cases, changing reported response assessment category (10/86 cases) or defining malignant hepatic disease on staging/restaging scans (1/11 cases).
Conclusion
PET-MRI offers several advantages over PET-CT in assessing the extent and response to therapy of malignant hepatic involvement. Additional malignant lesions detected on PET-MRI are attributed to superior PET performance (compared with PET of PET-CT), greater spatial resolution provided by MRI, and improved multi-parametric viability assessment. In around one-tenth of cases, findings identified on PET-MRI but not on PET-CT significantly change the final report’s conclusion.




Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- [18F]FDG:
-
18F-fluorodeoxyglucose
- PET-MRI:
-
Positron emission tomography-magnetic resonance imaging
- PET-CT:
-
Positron emission tomography-computed tomography
- DWI:
-
Diffusion-weighted imaging
- ADC:
-
Apparent diffusion coefficient
- DCE:
-
Dynamic contrast enhanced
- TOF:
-
Time of flight
- Pv:
-
P-Value
- IQR:
-
Interquartile range
References
Hectors SJ, Wagner M, Besa C, Huang W, Taouli B. Multiparametric FDG-PET/MRI of hepatocellular carcinoma: initial experience. Contrast Media Mol Imaging. 2018;3:2018.
Bipat S, van Leeuwen MS, Comans EF, Pijl ME, Bossuyt PM, Zwinderman AH, Stoker J. Colorectal liver metastases: CT, MR imaging, and PET for diagnosis. Meta-analysis. In Database of abstracts of reviews of effects (DARE): quality-assessed reviews [Internet] 2005. Centre for Reviews and Dissemination (UK).
Niekel MC, Bipat S, Stoker J. Diagnostic imaging of colorectal liver metastases with CT, MR imaging, FDG PET, and/or FDG PET/CT: a meta-analysis of prospective studies including patients who have not previously undergone treatment. In Database of abstracts of reviews of effects (DARE): quality-assessed reviews [Internet] 2010. Centre for Reviews and Dissemination (UK).
Floriani I, Torri V, Rulli E, Garavaglia D, Compagnoni A, Salvolini L, Giovagnoni A. Performance of imaging modalities in diagnosis of liver metastases from colorectal cancer: a systematic review and meta-analysis. J Magn Reson Imaging. 2010;31(1):19–31.
Vilgrain V, Esvan M, Ronot M, Caumont-Prim A, Aubé C, Chatellier G. A meta-analysis of diffusion-weighted and gadoxetic acid-enhanced MR imaging for the detection of liver metastases. Eur Radiol. 2016;26(12):4595–615.
Renzulli M, Clemente A, Ierardi AM, Pettinari I, Tovoli F, Brocchi S, Peta G, Cappabianca S, Carrafiello G, Golfieri R. Imaging of colorectal liver metastases: new developments and pending issues. Cancers. 2020;12(1):151.
Tahtabasi M, Erturk SM, Basak M. Comparison of MRI and 18F-FDG PET/CT in the liver metastases of gastrointestinal and pancreaticobiliary tumors. Med Bull Sisli Etfal Hosp. 2021;55(1):12.
Yong TW, Yuan ZZ, Jun Z, Lin Z, He WZ, Juanqi Z. Sensitivity of PET/MR images in liver metastases from colorectal carcinoma. Hell J Nucl Med. 2011;14(3):264–8.
Donati OF, Hany TF, Reiner CS, von Schulthess GK, Marincek B, Seifert B, Weishaupt D. Value of retrospective fusion of PET and MR images in detection of hepatic metastases: comparison with 18F-FDG PET/CT and Gd-EOB-DTPA–enhanced MRI. J Nucl Med. 2010;51(5):692–9.
Brendle C, Schwenzer NF, Rempp H, Schmidt H, Pfannenberg C, La Fougère C, Nikolaou K, Schraml C. Assessment of metastatic colorectal cancer with hybrid imaging: comparison of reading performance using different combinations of anatomical and functional imaging techniques in PET/MRI and PET/CT in a short case series. Eur J Nucl Med Mol Imaging. 2016;43(1):123–32.
Zhou N, Guo X, Sun H, Yu B, Zhu H, Li N, Yang Z. The value of 18F-FDG PET/CT and abdominal PET/MRI as a one-stop protocol in patients with potentially resectable colorectal liver metastases. Front Oncol. 2021;11:714948. https://doi.org/10.3389/fonc.2021.714948.
Zhou N, Meng X, Zhang Y, Yu B, Yuan J, Yu J, Zhu H, Yang Z. Diagnostic value of delayed PET/MR in liver metastasis in comparison with PET/CT. Front Oncol. 2021;11:717687. https://doi.org/10.3389/fonc.2021.717687
Beiderwellen K, Geraldo L, Ruhlmann V, Heusch P, Gomez B, Nensa F, Umutlu L, Lauenstein TC. Accuracy of [18F] FDG PET/MRI for the detection of liver metastases. PLoS One. 2015;10(9): e0137285.
Yoshino T, Arnold D, Taniguchi H, Pentheroudakis G, Yamazaki K, Xu RH, Kim TW, Ismail F, Tan IB, Yeh KH, Grothey A. Pan-Asian adapted ESMO consensus guidelines for the management of patients with metastatic colorectal cancer: a JSMO–ESMO initiative endorsed by CSCO, KACO, MOS SSO and TOS. Annals Oncol. 2018;29(1):44–70.
National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology colon cancer version 1. 2020. Available online: https://www.nccn.org/professionals/physician_gls/pdf/colon.pdf. Accessed 13 Nov 2022.
Martin O, Schaarschmidt BM, Kirchner J, Suntharalingam S, Grueneisen J, Demircioglu A, et al. PET/MRI versus PET/CT in whole-body staging: results from a unicenter observational study in 1003 subsequent examinations. J Nucl Med. 2020;61(8):1131–6.
Mayerhoefer ME, Prosch H, Beer L, Tamandl D, Beyer T, Hoeller C, Berzaczy D, Raderer M, Preusser M, Hochmair M, Kiesewetter B. PET/MRI versus PET/CT in oncology: a prospective single-center study of 330 examinations focusing on implications for patient management and cost considerations. Eur J Nucl Med Mol Imaging. 2020;47(1):51–60.
Musafargani S, Ghosh KK, Mishra S, Mahalakshmi P, Padmanabhan P, Gulyás B. PET/MRI: a frontier in era of complementary hybrid imaging. Eur J Hybrid Imaging. 2018;2(1):1–28.
Torrado-Carvajal A, Catana C. PET/MRI: technical and methodological aspects. Clinical PET/MRI. 2023;1:1–33.
GE Healthcare. SIGNA™ PET/MR technical data. http://promed-sa.com/wp-content/uploads/2020/08/PET-MR_Datasheet_DOC1545629.pdf. Accessed 13 Nov 2022.
Hamberg LM, Hunter GJ, Alpert NM, Choi NC, Babich JW, Fischman AJ. The dose uptake ratio as an index of glucose metabolism: useful parameter or oversimplification? J Nucl Med. 1994;35(8):1308–12.
Thie JA, Hubner KF, Smith GT. Optimizing imaging time for improved performance in oncology PET studies. Mol Imag Biol. 2002;4(3):238–44.
Boellaard R, Delgado-Bolton R, Oyen WJ, Giammarile F, Tatsch K, Eschner W, Verzijlbergen FJ, Barrington SF, Pike LC, Weber WA, Stroobants S. FDG PET/CT: EANM procedure guidelines for tumour imaging: version 2.0. Eur J Nucl Med Mol Imaging. 2015;42(2):328–54.
Lasnon C, Coudrais N, Houdu B, Nganoa C, Salomon T, Enilorac B, Aide N. How fast can we scan patients with modern (digital) PET/CT systems? Eur J Radiol. 2020;1(129): 109144.
Fragoso Costa P, Jentzen W, Brahmer A, Mavroeidi IA, Zarrad F, Umutlu L, Fendler WP, Rischpler C, Herrmann K, Conti M, Seifert R. Phantom-based acquisition time and image reconstruction parameter optimisation for oncologic FDG PET/CT examinations using a digital system. BMC Cancer. 2022;22(1):1–8.
Pan T, Einstein SA, Kappadath SC, Grogg KS, Lois Gomez C, Alessio AM, Hunter WC, El Fakhri G, Kinahan PE, Mawlawi OR. Performance evaluation of the 5-ring GE discovery MI PET/CT system using the national electrical manufacturers association NU 2–2012 standard. Med Phys. 2019;46(7):3025–33.
Levin C, Peterson W, Ross S, Stearns C, Uribe J. PET performance as a function of axial field of view for a new silicon photomultiplier-based whole body TOF PET/CT system. J Nucl Med. 2016;57(suppl 2):200.
Wagenknecht G, Kaiser HJ, Mottaghy FM, Herzog H. MRI for attenuation correction in PET: methods and challenges. Magn Reson Mater Phys, Biol Med. 2013;26(1):99–113.
Kostakoglu L, Goldsmith SJ. 18F-FDG PET evaluation of the response to therapy for lymphoma and for breast, lung, and colorectal carcinoma. J Nucl Med. 2003;44(2):224–39.
Glazer ES, Beaty K, Abdalla EK, Vauthey JN, Curley SA. Effectiveness of positron emission tomography for predicting chemotherapy response in colorectal cancer liver metastases. Arch Surg. 2010;145(4):340–5.
Akhurst T, Kates TJ, Mazumdar M, Yeung H, Riedel ER, Burt BM, Blumgart L, Jarnagin W, Larson SM, Fong Y. Recent chemotherapy reduces the sensitivity of [18F] fluorodeoxyglucose positron emission tomography in the detection of colorectal metastases. J Clin Oncol. 2005;23(34):8713–6.
Acknowledgements
We thank our department’s research assistants Irina Dana and Liron Maor for their assistance in conducting this study, and the anonymous reviewers of the European Journal of Nuclear Medicine and Molecular Imaging for their insightful feedbacks.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. All patients were enrolled as part of an ongoing prospective study to evaluate the added values of PET-MRI over PET-CT in the imaging of various malignancies (ClinicalTrials.gov identifier: NCT04158414, registered November 8, 2019). This study was approved by the institutional review board (Reference ID 0568–19-TLV), and all participating patients gave written informed consent.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Oncology—General.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cohen, D., Kesler, M., Muchnik Kurash, M. et al. A lesson in humility: the added values of PET-MRI over PET-CT in detecting malignant hepatic lesions. Eur J Nucl Med Mol Imaging 50, 1423–1433 (2023). https://doi.org/10.1007/s00259-022-06099-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-022-06099-8