Abstract
Purpose
Actinium-225-labeled prostate-specific membrane antigen ([225Ac]Ac-PSMA-617) is safe and effective in the treatment of metastatic castration-resistant prostate cancer (mCRPC). No study has specifically assessed its safety in patients with extensive skeletal metastases of mCRPC. We aimed to investigate the hematologic toxicity and efficacy of [225Ac]Ac-PSMA-617 therapy in patients with extensive skeletal metastases of mCRPC.
Methods
We retrospectively reviewed the medical record of patients treated with [225Ac]Ac-PSMA-617 for mCRPC. We included patients with a superscan pattern of skeletal metastases and those with 20 or more multifocal sites of skeletal metastases on baseline [68 Ga]Ga-PSMA-11 PET/CT. We reviewed the levels of hemoglobin, white blood cell (WBC), and platelet prior to each cycle of treatment and determined the presence of impaired bone marrow function at baseline and the grade of toxicity in the hematologic parameters induced by treatment. We evaluated the predictors of hematologic toxicity using binary logistic regression analysis. We also determined the presence of renal dysfunction before or during treatment. We assessed response to treatment using prostate-specific antigen response and the progression-free survival (PFS) and overall survival (OS).
Results
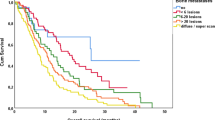
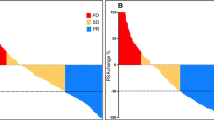
A total of 106 patients were included. Skeletal metastasis was in the superscan pattern in 34 patients (32.1%) and multifocal in 72 patients (67.9%). The median treatment cycle was 4 (range = 1–9). Ninety-eight patients (92.5%) had abnormal baseline hematologic parameters. One patient had grade 4 thrombocytopenia. Grade 3 anemia, leukopenia, and thrombocytopenia were seen in 1 (0.9%), 3 (2.8%), and 2 (1.9%) patients, respectively. Age, the number of treatment cycles, and the presence of renal dysfunction were significant predictors of hematologic toxicity. Eighty-five patients (80.2%) achieved PSA response. The median PFS and OS of the study population were 14:00 (95%CI: 8.15–19.86) months and 15.0 (95%CI: 12.8–17.2) months, respectively.
Conclusions
[225Ac]Ac-PSMA-617 induces a good anti-tumor effect in about 80% of patients with extensive skeletal metastases of mCRPC with a rare incidence of severe hematologic toxicity. Age, number of treatment cycles, and the presence of renal dysfunction were significant risk factors for hematologic toxicity of [225Ac]Ac-PSMA-617 therapy.






Similar content being viewed by others
Data availability
All data collected during the conduct of this study are included in this report.
References
Pomykala KL, Czernin J, Grogan TR, Armstrong WR, Williams J, Calais J. Total-body 68Ga-PSMA-11 PET/CT for bone metastases detection in prostate cancer patients: potential impact on bone scan guidelines. J Nucl Med. 2020;61:405–11.
James ND, Spears M, Clarke NW, Dearnaley DP, De Bono JS, Gale J, et al. Survival with newly diagnosed metastatic prostate cancer in the “docetaxel era”: data from 917 patients in the control arm of the STAMPEDE trial (MRC PR08, CRUK/06/019). Eur Urol. 2015;67:1028–38.
Kesler M, Kerzhner K, Druckmann I, Kuten J, Levine C, Sarid D, et al. Staging 68Ga-PSMA PET/CT in 963 consecutive patients with newly diagnosed prostate cancer: incidence and characterization of skeletal involvement. Eur J Nucl Med Mol Imaging. Epub ahead of print on 27 December 2021.
Broder MS, Gutierrez B, Cherepanov D, Linhares Y. Burden of skeletal-related events in prostate cancer: unmet need in pain improvement. Support Care Cancer. 2015;23:237–47.
Tannock IF, de Wit R, Berry WR, Horti J, Pluzanska A, Chi KN, et al. Docetaxel plus prednisone or mitoxantrone plus prednisone for advanced prostate cancer. N Engl J Med. 2004;351:1502–12.
de Bono JS, Oudard S, Ozguroglu M, Hansen S, Machiels JP, Kocak I, et al. Prednisone plus cabazitaxel or mitoxantrone for metastatic castration-resistant prostate cancer progression after docetaxel treatment: a randomized open-label trial. Lancet. 2010;376:1147–54.
Fizazi K, Tran N, Fein L, Matsubara N, Rodriguez-Antolin A, Alekseev BY, et al. Abiraterone acetate plus prednisone in patients with newly diagnosed high-risk metastatic castration-sensitive prostate cancer (LATITUDE): final overall survival analysis of a randomised, double-blind, phase 3 trial. Lancet Oncol. 2019;20:686–700.
Beer TM, Armstrong AJ, Rathkopf DE, Loriot Y, Sternberg CN, Higano CS, et al. Enzalutamide in metastatic prostate cancer before chemotherapy. N Engl J Med. 2014;371:424–33.
Parker C, Nilsson S, Heinrich D, Helle SI, O’Sullivan JM, Fosså SD, et al. Alpha emitter radium-223 and survival in metastatic prostate cancer. N Engl J Med. 2013;369:213–23.
Rahbar K, Ahmadzadehfar H, Kratochwil C, Haberkorn U, Schafers M, Schmidt M, et al. German multicenter study investigating 177Lu-PSMA-617 radioligand therapy in advanced prostate cancer patients. J Nucl Med. 2017;58:85–90.
Ahmadzadehfar H, Rahbar K, Baum R, Seifert R, Kessel K, Bogemann M, et al. Prior therapies as prognostic factors of overall survival in metastatic castration-resistant prostate cancer patients treated with [177Lu]Lu-PSMA-617. A WARMTH multicentre study (the 617 trial). Eur J Nuclear Med Mol Imaging. 2021;48:113–22.
Heck MM, Tauber R, Schwaiger S, Retz M, D’Alessandria C, Maurer T, et al. Treatment outcome, toxicity, and predictive factors for radioligand therapy with 177Lu-PSMA-I&T in metastatic castration-resistant prostate cancer. Eur Urol. 2019;75:920–6.
Khreish F, Ghazal Z, Marlowe RJ, Rosar F, Sabet A, Maus S, et al. 177Lu-PSMA-617 radioligand therapy of metastatic castration-resistant prostate cancer: Initial 254-patient results from a prospective registry (REALITY Study). Eur J Nucl Med Mol Imaging. 2022;49:1075–85.
Hofman MS, Emmett L, Sandhu S, Iravani A, Joshua AM, Goh JC, et al. [177Lu]Lu-PSMA-617 versus cabazitaxel in patients with metastatic castration-resistant prostate cancer (TheraP): a randomised, open-label, phase 2 trial. Lancet. 2021;397:797–804.
Sator O, de Bono J, Chi KN, Fizazi K, Hermann K, Rahbar K, et al. Lutetium-177-PSMA-617 for metastatic castration-resistant prostate cancer. N Engl J Med. 2021;385:1091–103.
Sadaghiani MS, Sheikhbahaei S, Werner RA, Pienta KJ, Pomper MG, Solnes LB, et al. A systematic review and meta-analysis of the effectiveness and toxicities of lutetium-177-labeled prostate-specific membrane antigen-targeted radioligand therapy in metastatic castration-resistant prostate cancer. Eur Urol. 2021;80:82–94.
Morgenstern A, Apostolidis C, Kratochwil C, Sathekge M, Krolicki L, Bruchertseifer F. An overview of targeted alpha therapy with 225actnium and 213bismuth. Curr Radiopharm. 2018;11:200–8.
Kratochwil C, Bruchertseifer F, Giesel FL, Weis N, Verburg FA, Mottaghy F, et al. 225Ac-PSMA-617 for PSMA-targeted α-radiation therapy of metastatic castration-resistant prostate cancer. J Nucl Med. 2016;57:1941–4.
Kratochwil C, Bruchertseifer F, Rathke H, Bronzel M, Apostolidis C, Weichert W, et al. Targeted α-therapy of metastatic castration-resistant prostate cancer with 225Ac-PSMA-617: dosimetry estimate and empiric dose finding. J Nucl Med. 2017;58:1624–31.
Kratochwil C, Bruchertseifer F, Rathke H, Hohenfellner M, Giesel FL, Haberkorn U, et al. Targeted α-therapy of metastatic castration-resistant prostate cancer with 225Ac-PSMA-617: swimmer-plot analysis suggests efficacy regarding duration of tumor control. J Nucl Med. 2018;59:795–802.
Sathekge M, Bruchertseifer F, Knoesen O, Reyneke F, Lawal I, Lengana T, et al. 225Ac-PSMA-617 in chemotherapy-naïve patients with advanced prostate cancer: a pilot study. Eur J Nucl Med Mol Imaging. 2019;46:129–38.
Sathekge M, Bruchertseifer F, Vorster M, Lawal IO, Knoesen O, Mahapane J, et al. Predictors of overall and disease-free survival in metastatic castration-resistant prostate cancer patients receiving 225Ac-PSMA-617 radioligand therapy. J Nucl Med. 2020;61:62–9.
Yadav MP, Ballal S, Sahoo RK, Tripathi M, Seth A, Bal C. Efficacy and Safety of 225Ac-PSMA-617 targeted alpha therapy in metastatic castration-resistant prostate cancer patients. Theranostics. 2020;23:9364–77.
Feuerecker B, Tauber R, Knorr K, Heck M, Beheshti A, Sedl C, et al. Activity and adverse events of actinium-225-PSMA-617 in advanced metastatic castration-resistant prostate cancer after failure of lutetium-177-PSMA. Eur Urol. 2021;79:343–50.
Zacherrl MJ, Gildehaus FJ, Mittlmeier L, Boning G, Gosewisch A, Wenter V, et al. First clinical results for PSMA-targeted α-therapy using 225Ac-PSMA-I&T in advanced-mCRPC patients. J Nucl Med. 2021;62:669–74.
Sathekge M, Bruchertseifer F, Vorster M, Lawal I, Knoesen O, Mahapane J, et al. mCRPC patients receiving 222Ac-PSMA-617 therapy in the post androgen deprivation therapy setting: response to treatment and survival analysis. J Nucl Med. Epub ahead of print on February 14, 2022.
Haberkorn U, Giesel F, Morgenstern A, Kratochwil C. The future of radioligand therapy: α, β, or Both? J Nucl Med. 2017;58:1017–8.
Lawal IO, Bruchertseifer F, Vorster M, Morgenstern A, Sathekge MM. Prostate-specific membrane antigen-targeted endoradiotherapy in metastatic prostate cancer. Curr Opin Urol. 2020;30:98–105.
Lawal I, Vorster M, Boshomane T, Ololade K, Ebenhan T, Sathekge M. Metastatic prostate carcinoma presenting as a superscan on 68Ga-PSMA PET/CT. Clin Nucl Med. 2015;40:755–6.
Sathekge MM, Bruchertseifer F, Vorster M, Morgenstern A, Lawal IO. Global experience with PSMA-based alpha therapy in prostate cancer. Eur J Nucl Med Mol Imaging. 2021;49:30–46.
National Cancer Institute – Division of Cancer Treatment and Diagnosis. Common Terminology Criteria for Adverse Events (CTCAE) v5.0. Available from: https://evs.nci.nih.gov/ftp1/CTCAE/About.html. Accessed on 20 December 2021.
Bubley GJ, Carducci M, Dahut W, Dawson N, Daliani D, Eisenberger M, et al. Eligibility and response guidelines for phase II clinical trials in androgen-independent prostate cancer: recommendations from the Prostate-Specific Antigen Working Group. J Clin Oncol. 1999;17:3461–7.
Scher HI, Morris MJ, Stadler WM, Higano C, Basch E, Fizazi K, et al. Trial design and objectives for castration-resistant prostate cancer: updated recommendations from the Prostate Cancer Clinical Trials Working Group 3. J Clin Oncol. 2016;34:1402–18.
Nieder C, Haukland E, Pawinski A, Dalhuag A. Anaemia and thrombocytopenia in patients with prostate cancer and bone metastases. BMC Cancer. 2010;10:284.
Sathekge MM, Bruchertseifer F, Lawal IO, Vorster M, Knoesen O, Lengana T, et al. Treatment of brain metastases of castration-resistant prostate cancer with 225Ac-PSMA-617. Eur J Nucl Med Mol Imaging. 2019;46:1756–7.
Maserumule LC, Mokoala KMG, Hlongwa KN, Ndlovu H, Reed JD, Ismail A, et al. Exceptional initial response of prostate cancer lung metastases to 225Ac-PSMA: A case report. Curr Probl Cancer: Case Rep. 2021;3:100038.
Gafita A, Wang H, Robertson A, Armstrong WR, Zaum R, Weber M, et al. Tumour sink effect in 68Ga-PSMA-11 PET: myth or reality? J Nucl Med. Epub ahead of print on 28 May 2021.
Gaertner FC, Halabi K, Ahmadzadehfar H, Kurpig S, Eppard E, Kotsikopoulos C, et al. Uptake of PSMA-ligand in normal tissues is dependent on tumour load in patients with prostate cancer. Oncotarget. 2017;8:55094–103.
van der Sar ECA, de Keizer B, Lam MGEH, Braat AJAT. Competition (‘steal’ phenomenon) between [68Ga]Ga-PSMA-11 uptake in prostate tumour tissue versus healthy tissue. Pharmaceutics. 2021;13:699.
Hofman MS, Violet J, Hicks RJ, Ferdinandus J, Thang SP, Akhurst T, et al. [177Lu]-PSMA-617 radionuclide treatment in patients with metastatic castration-resistant prostate cancer (LuPSMA trial): a single-centre, single-arm, phase 2 study. Lancet Oncol. 2018;19:825–33.
Gafita A, Fendler WP, Hui W, Sandhu S, Weber M, Esfandiari R, et al. Efficacy and safety of 177Lu-labeled prostate-specific membrane antigen treatment in patients with diffuse bone marrow involvement: a multicentre retrospective study. Eur Urol. 2020;78:148–54.
Groener D, Baumgarten J, Haefele S, Happel C, Klimek K, Mader N, et al. Salvage radioligand therapy with repeated cycles of 177Lu-PSMA-617 in metastatic castration-resistant prostate cancer with diffuse bone marrow involvement. Cancers. 2021;13:4017.
Tranel J, Feng FY, St James S, Hope TA. Effect of microdistribution of alpha and beta-emitters in targeted radionuclide therapies on delivered dose in a GATE model of bone marrow. Phys Med Biol. 2021;66:035016.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
This study was performed per the ethical standard of our institutions and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Consent to participate
All patients gave written informed consent to participate in the study.
Consent for publication
All patients provided informed consent to allow for the publication of their anonymized data collected during this study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Oncology—Genitourinary.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lawal, I.O., Morgenstern, A., Vorster, M. et al. Hematologic toxicity profile and efficacy of [225Ac]Ac-PSMA-617 α-radioligand therapy of patients with extensive skeletal metastases of castration-resistant prostate cancer. Eur J Nucl Med Mol Imaging 49, 3581–3592 (2022). https://doi.org/10.1007/s00259-022-05778-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-022-05778-w