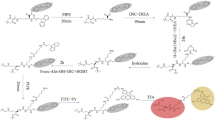
Abstract
Purpose
Radioligand therapy (RLT) targeting prostate-specific membrane antigen (PSMA) is emerging as an effective treatment option for metastatic castration-resistant prostate cancer (mCRPC). An imaging-based method to quantify early treatment responses can help to understand and optimize RLT.
Methods
We developed a self-triggered probe 2 targeting the colocalization of PSMA and caspase-3 for fluorescence imaging of RLT-induced apoptosis.
Results
The probe binds to PSMA potently with a Ki of 4.12 nM, and its fluorescence can be effectively switched on by caspase-3 with a Km of 67.62 μM. Cellular and in vivo studies demonstrated its specificity for imaging radiation-induced caspase-3 upregulation in prostate cancer. To identify the detection limit of our method, we showed that probe 2 could achieve 1.79 times fluorescence enhancement in response to 177Lu-RLT in a medium PSMA-expressing 22Rv1 xenograft model.
Conclusion
Probe 2 can potently bind to PSMA, and the fluorescence signal can be sensitively switched on by caspase-3 both in vitro and in vivo. This method may provide an effective tool to investigate and optimize PSMA-RLT.




Similar content being viewed by others
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
Sung H, Ferlay J, Siegel RL, et al. Global cancer statistics 2020: global estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49.
Litwin MS, Tan HJ. The diagnosis and treatment of prostate cancer: a review. JAMA - J Am Med Assoc. 2017;317:2532–42.
Rebello RJ, Oing C, Knudsen KE, et al. Prostate cancer. Nat Rev Dis Primers. 2021;7:9.
Hofman MS, Violet J, Hicks RJ, et al. [177 Lu]-PSMA-617 radionuclide treatment in patients with metastatic castration-resistant prostate cancer (LuPSMA trial): a single-centre, single-arm, phase 2 study. Lancet Oncol. 2018;19:825–33.
Sartor O, de Bono J, Chi KN, et al. Lutetium-177–PSMA-617 for metastatic castration-resistant prostate cancer. N Engl J Med. 2021;385:1091–103.
Hofman MS, Emmett L, Sandhu S, et al. [177Lu]Lu-PSMA-617 versus cabazitaxel in patients with metastatic castration-resistant prostate cancer (TheraP): a randomised, open-label, phase 2 trial. Lancet. 2021;397:797–804.
Langbein T, Weber WA, Eiber M. Future of theranostics: an outlook on precision oncology in nuclear medicine. J Nucl Med. 2019;60:13S-S19.
Wüstemann T, Haberkorn U, Babich J, Mier W. Targeting prostate cancer: prostate-specific membrane antigen based diagnosis and therapy. Med Res Rev. 2019;39:40–69.
Fendler WP, Rahbar K, Herrmann K, Kratochwil C, Eiber M. 177Lu-PSMA radioligand therapy for prostate cancer. J Nucl Med. 2017;58:1196–200.
Gafita A, Heck MM, Rauscher I, et al. Early prostate-specific antigen changes and clinical outcome after 177Lu-PSMA radionuclide treatment in patients with metastatic castration-resistant prostate cancer. J Nucl Med. 2020;61:1476–83.
Lee BS, Cho YW, Kim GC, et al. Induced phenotype targeted therapy: radiation-induced apoptosis-targeted chemotherapy. J Natl Cancer Inst. 2015;107:dju403.
Fu Q, Li H, Duan D, et al. External-radiation-induced local hydroxylation enables remote release of functional molecules in tumors. Angew Chemie Int Ed Engl. 2020;59:21546–52.
Geng J, Zhang Y, Gao Q, et al. Switching on prodrugs using radiotherapy. Nat Chem. 2021;13:805–10.
Paschalis A, Sheehan B, Riisnaes R, et al. Prostate-specific membrane antigen heterogeneity and DNA repair defects in prostate cancer. Eur Urol. 2019;76:469–78.
Zhang J, Chai X, He XP, Kim HJ, Yoon J, Tian H. Fluorogenic probes for disease-relevant enzymes. Chem Soc Rev. 2019;48:683–722.
Porter AG, Jänicke RU. Emerging roles of caspase-3 in apoptosis. Cell Death Differ. 1999;6:99–104.
Huang Q, Li F, Liu X, et al. Caspase 3-mediated stimulation of tumor cell repopulation during cancer radiotherapy. Nat Med. 2011;17:860–6.
Edgington LE, Berger AB, Blum G, et al. Noninvasive optical imaging of apoptosis by caspase-targeted activity-based probes. Nat Med. 2009;15:967–73.
He X, Hu Y, Shi W, Li X, Ma H. Design, synthesis and application of a near-infrared fluorescent probe for in vivo imaging of aminopeptidase N. Chem Commun. 2017;53:9438–41.
Yuan Y, Kwok RTK, Tang BZ, Liu B. Targeted theranostic platinum(IV) prodrug with a built-in aggregation-induced emission light-up apoptosis sensor for noninvasive early evaluation of its therapeutic responses in situ. J Am Chem Soc. 2014;136:2546–54.
Zeng Z, Liew SS, Wei X, Pu K. Hemicyanine-based near-infrared activatable probes for imaging and diagnosis of diseases. Angew Chemie Int Ed Engl. 2021;60:2–24.
Yuan L, Lin W, Yang Y, Chen H. A unique class of near-infrared functional fluorescent dyes with carboxylic-acid-modulated fluorescence on/off switching: rational design, synthesis, optical properties, theoretical calculations, and applications for fluorescence imaging in living animals. J Am Chem Soc. 2012;134:1200–11.
Cheng D, Peng J, Lv Y, et al. De novo design of chemical stability near-infrared molecular probes for high-fidelity hepatotoxicity evaluation in vivo. J Am Chem Soc. 2019;141:6352–61.
Cheng P, Miao Q, Li J, Huang J, Xie C, Pu K. Unimolecular chemo-fluoro-luminescent reporter for crosstalk-free duplex imaging of hepatotoxicity. J Am Chem Soc. 2019;141:10581–4.
He X, Li L, Fang Y, Shi W, Li X, Ma H. In vivo imaging of leucine aminopeptidase activity in drug-induced liver injury and liver cancer via a near-infrared fluorescent probe. Chem Sci. 2017;8:3479–83.
Tian X, Li Z, Ding N, Zhang J. Near-infrared ratiometric self-assembled theranostic nanoprobe: imaging and tracking cancer chemotherapy. Chem Commun. 2020;56:3629–32.
Rhéaume E, Cohen LY, Uhlmann F, et al. The large subunit of replication factor C is a substrate for caspase-3 in vitro and is cleaved by a caspase-3-like protease during fas-mediated apoptosis. EMBO J. 1997;16:6346–54.
Kozikowski AP, Nan F, Conti P, et al. Design of remarkably simple, yet potent urea-based inhibitors of glutamate carboxypeptidase II (NAALADase). J Med Chem. 2001;44:298–301.
Yang X, Mease RC, Pullambhatla M, et al. Fluorobenzoyllysinepentanedioic acid carbamates: new scaffolds for positron emission tomography (PET) imaging of prostate-specific membrane antigen (PSMA). J Med Chem. 2016;59:206–18.
Duan X, Liu F, Kwon H, et al. (S)-3-(Carboxyformamido)-2-(3-(carboxymethyl)ureido)propanoic acid as a novel PSMA targeting scaffold for prostate cancer imaging. J Med Chem. 2020;63:3563–76.
Müller C, Van Der Meulen NP, Benešová M, Schibli R. Therapeutic radiometals beyond 177Lu and 90Y: production and application of promising α-particle, β–particle, and auger electron emitters. J Nucl Med. 2017;58:91S-S96.
Benesová M, Schäfer M, Bauder-Wüst U, et al. Preclinical evaluation of a tailor-made DOTA-conjugated PSMA inhibitor with optimized linker moiety for imaging and endoradiotherapy of prostate cancer. J Nucl Med. 2015;56:914–20.
Garcia-Calvo M, Peterson EP, Leiting B, Ruel R, Nicholson DW, Thornberry NA. Inhibition of human caspases by peptide-based and macromolecular inhibitors. J Biol Chem. 1998;273:32608–13.
Choi B, Rempala GA, Kim JK. Beyond the Michaelis-Menten equation: accurate and efficient estimation of enzyme kinetic parameters. Sci Rep. 2017;7:17018.
Chang Lee S, Ma JSY, Kim MS, et al. A PSMA-targeted bispecific antibody for prostate cancer driven by a small-molecule targeting ligand. Sci Adv. 2021;7:eabi8193.
Kiess AP, Minn I, Chen Y, et al. Auger radiopharmaceutical therapy targeting prostate-specific membrane antigen. J Nucl Med. 2015;56:1401–7.
Olszewski RT, Bukhari N, Zhou J, et al. NAAG peptidase inhibition reduces locomotor activity and some stereotypes in the PCP model of schizophrenia via group II mGluR. J Neurochem. 2004;89:876–85.
Cho YW, Kim SY, Kwon IC, Kim IS. Complex adaptive therapeutic strategy (CATS) for cancer. J Control Release. 2014;175:43–7.
Zhao Y, Zhang T, Wang YP, et al. ICAM-1 orchestrates the abscopal effect of tumor radiotherapy. Proc Natl Acad Sci USA. 2021;118:e2010333118.
Hofman MS, Lawrentschuk N, Francis RJ, et al. Prostate-specific membrane antigen PET-CT in patients with high-risk prostate cancer before curative-intent surgery or radiotherapy (proPSMA): a prospective, randomised, multicentre study. Lancet. 2020;395:1208–16.
Eder M, Schäfer M, Bauder-Wüst U, et al. 68Ga-complex lipophilicity and the targeting property of a urea-based PSMA inhibitor for PET imaging. Bioconjug Chem. 2012;23:688–97.
Chen Y, Pullambhatla M, Foss CA, et al. 2-(3-{1-carboxy-5-[(6-[18F]fluoro-pyridine-3-carbonyl)-amino]- pentyl}-ureido)-pentanedioic acid, [18F]DCFPyL, a PSMA-based PET imaging agent for prostate cancer. Clin Cancer Res. 2011;17:7645–53.
Zacherl MJ, Gildehaus FJ, Mittlmeier L, et al. First clinical results for PSMA-targeted α-therapy using 225Ac-PSMA-I&T in advanced-mCRPC patients. J Nucl Med. 2021;62:669–74.
Funding
This work was financially supported by the National Natural Science Foundation of China (92059101, 21877004, 81873907, and 81920108020), National Key R&D Program of China (2018YFE0205300 and 2018YFC1313300), Beijing Natural Science Foundation (JQ19026), Clinical Medicine Plus X-Young Scholars Project of Peking University (PKU2020LCXQ029), and the Fundamental Research Funds for the Central Universities (BMU2021RCZX018 and PKU2019LCXQ023). This work was in part supported by the National Research Foundation of Korea (2019R1A6A1A03013807 and 2020R1A2C2005919).
Author information
Authors and Affiliations
Contributions
Xing Yang and Zhaofei Liu conceived and designed research. Hongchuang Xu performed the chemical synthesis. Hongchuang Xu and Xuekang Cai measured the absorption and emission spectrum. Jingming Zhang performed in vitro caspase-3 protein assay and PSMA inhibition experiments. Hyunsoo Ha and Youngjoo Byun performed the docking study. Xiaojiang Duan and Yanpu Wang performed radiolabeling experiments. Xiaojiang Duan performed cell uptake experiments. Yanpu Wang and Ting Zhang designed and performed live cell imaging experiments and SPECT/CT imaging experiments. Yanpu Wang performed the mouse imaging. Hongchuang Xu, Yanpu Wang, Jingming Zhang, Xing Yang, and Zhaofei Liu analyzed data. Xing Yang, Zhaofei Liu, Jingming Zhang, Hongchuang Xu, Yanpu Wang, and Xuekang Cai wrote the original draft. Xing Yang, Zhaofei Liu, Yanpu Wang, Yan Fan, and Zhi Yang reviewed and edited the draft. All authors discussed the results and commented on the manuscript.
Corresponding authors
Ethics declarations
Ethics approval
All animal studies were carried out in conformity to regulations on laboratory animals of the Beijing municipality and approved by the Animal Ethics Committee at Peking University Frist Hospital (Beijing, China), approval number J202163.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Clinical trial registration
Not applicable.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Theragnostic.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xu, H., Wang, Y., Zhang, J. et al. A self-triggered radioligand therapy agent for fluorescence imaging of the treatment response in prostate cancer. Eur J Nucl Med Mol Imaging 49, 2693–2704 (2022). https://doi.org/10.1007/s00259-022-05743-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-022-05743-7