Abstract
Purpose
To construct an FDG PET/CT metabolic parameter-based model to predict early recurrence of hepatocellular carcinoma (HCC) after liver transplantation (LT).
Methods
A total of 62 patients with HCC after LT were enrolled with a follow-up period of 1 year. Basic clinical, pathology, and laboratory data, CT features (CPLC), and PET metabolic parameters (CPLCP) were collected for model construction. A CPLC nomogram without metabolic parameters and a CPLCP nomogram with metabolic parameters were established. The net reclassification index (NRI) and integrated discrimination improvement (IDI) of the two models were calculated. The constructed model was compared with Milan criteria and University of California San Francisco (UCSF) criteria. The time-dependent area under the receiver operating characteristic curve (time-AUC) was used to compare the efficiency of the models, and the bootstrap method was used to for verification. Harrell’s concordance index (C-index) was used to evaluate the performance of these models. Decision curve analysis (DCA) was used to evaluate the clinical practicability of each model.
Results
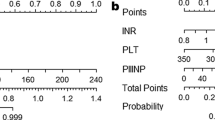
Thirty out of 62 patients experienced a recurrence during the 1-year follow-up. BCLC stage (P = 0.009), MVI (P = 0.032), AFP (P = 0.004), CTdmax (P = 0.033), and MTV (P = 0.039) were the independent predictors. The CPLC nomogram and the CPLCP nomogram were established. Compared with the CPLC nomogram, the NRI of the CPLCP nomogram increased by 38.98% (95% CI = −18.77–60.43%) and the IDI increased by 4.40% (95% CI = −1.00–16.62%). The AUC value of the CPLCP nomogram was higher than those of Milan criteria and UCSF criteria in the time-AUC curve. Moreover, the CPLCP nomogram had a higher C-index (0.774) than other models. Finally, the DCA curve showed that clinical practicability of the CPLCP nomogram outperformed the Milan criteria and UCSF criteria.
Conclusions
The CPLCP nomogram combining basic clinical data, pathology data, laboratory data, CT features, and PET metabolic parameters showed good efficacy and high clinical practicability in predicting the early recurrence of HCC after LT.





Similar content being viewed by others
Data availability
The datasets generated and analysed during the current study are not publicly available due to patient privacy concerns, but they are available from the corresponding author upon reasonable request.
References
Bray F, Ferlay J, Soerjomataram I. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. https://doi.org/10.3322/caac.21492.
Forner A, Reig M, Bruix J. Hepatocellular carcinoma. Lancet. 2018;391(10127):1301–14. https://doi.org/10.1016/s0140-6736(18)30010-2.
Kwong A, Kim WR, Lake JR, Smith JM, Schladt DP, Skeans MA, et al. OPTN/SRTR 2018 annual data report: liver. Am J Transplant. 2020;20(Suppl s1):193–299. https://doi.org/10.1111/ajt.15674.
EASL Clinical Practice Guidelines. Management of hepatocellular carcinoma. J Hepatol. 2018;69(1):182–236. https://doi.org/10.1016/j.jhep.2018.03.019.
Bodzin AS, Lunsford KE, Markovic D, Harlander-Locke MP, Busuttil RW, Agopian VG. Predicting mortality in patients developing recurrent hepatocellular carcinoma after liver transplantation: impact of treatment modality and recurrence characteristics. Ann Surg. 2017;266(1):118–25. https://doi.org/10.1097/sla.0000000000001894.
Sapisochin G, Goldaracena N, Astete S, Laurence JM, Davidson D, Rafael E, et al. Benefit of treating hepatocellular carcinoma recurrence after liver transplantation and analysis of prognostic factors for survival in a large Euro-American series. Ann Surg Oncol. 2015;22(7):2286–94. https://doi.org/10.1245/s10434-014-4273-6.
Liang W, Wu L, Ling X, Schroder PM, Ju W, Wang D, et al. Living donor liver transplantation versus deceased donor liver transplantation for hepatocellular carcinoma: a meta-analysis. Liver Transpl. 2012;18(10):1226–36. https://doi.org/10.1002/lt.23490.
Fernandez-Sevilla E, Allard MA, Selten J, Golse N, Vibert E, Sa Cunha A, et al. Recurrence of hepatocellular carcinoma after liver transplantation: is there a place for resection? Liver Transpl. 2017;23(4):440–7. https://doi.org/10.1002/lt.24742.
de’Angelis N, Landi F, Carra MC, Azoulay D. Managements of recurrent hepatocellular carcinoma after liver transplantation: a systematic review. World J Gastroenterol. 2015;21(39):11185–98. https://doi.org/10.3748/wjg.v21.i39.11185.
Goldaracena N, Gorgen A, Doyle A, Hansen BE, Tomiyama K, Zhang W, et al. Live donor liver transplantation for patients with hepatocellular carcinoma offers increased survival vs. deceased donation. J Hepatol. 2019;70(4):666–73. https://doi.org/10.1016/j.jhep.2018.12.029.
Verna EC, Patel YA, Aggarwal A, Desai AP, Frenette C, Pillai AA, et al. Liver transplantation for hepatocellular carcinoma: management after the transplant. Am J Transplant. 2020;20(2):333–47. https://doi.org/10.1111/ajt.15697.
Bruix J, Takayama T, Mazzaferro V, Chau GY, Yang J, Kudo M, et al. Adjuvant sorafenib for hepatocellular carcinoma after resection or ablation (STORM): a phase 3, randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2015;16(13):1344–54. https://doi.org/10.1016/s1470-2045(15)00198-9.
Ferrer-Fàbrega J, Forner A, Liccioni A, Miquel R, Molina V, Navasa M, et al. Prospective validation of ab initio liver transplantation in hepatocellular carcinoma upon detection of risk factors for recurrence after resection. Hepatology. 2016;63(3):839–49. https://doi.org/10.1002/hep.28339.
Al-Ameri AAM, Wei X, Wen X, Wei Q, Guo H, Zheng S, et al. Systematic review: risk prediction models for recurrence of hepatocellular carcinoma after liver transplantation. Transpl Int. 2020;33(7):697–712. https://doi.org/10.1111/tri.13585.
Mazzaferro V, Regalia E, Doci R, Andreola S, Pulvirenti A, Bozzetti F, et al. Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N Engl J Med. 1996;334(11):693–9. https://doi.org/10.1056/nejm199603143341104.
Yao FY, Ferrell L, Bass NM, Watson JJ, Bacchetti P, Venook A, et al. Liver transplantation for hepatocellular carcinoma: expansion of the tumor size limits does not adversely impact survival. Hepatology. 2001;33(6):1394–403. https://doi.org/10.1053/jhep.2001.24563.
Zheng SS, Xu X, Wu J, Chen J, Wang WL, Zhang M, et al. Liver transplantation for hepatocellular carcinoma: Hangzhou experiences. Transplantation. 2008;85(12):1726–32. https://doi.org/10.1097/TP.0b013e31816b67e4.
Wang Q, Xia D, Bai W, Wang E, Sun J, Huang M, et al. Development of a prognostic score for recommended TACE candidates with hepatocellular carcinoma: a multicentre observational study. J Hepatol. 2019;70(5):893–903. https://doi.org/10.1016/j.jhep.2019.01.013.
Mazzaferro V, Sposito C, Zhou J, Pinna AD, De Carlis L, Fan J, et al. Metroticket 2.0 model for analysis of competing risks of death after liver transplantation for hepatocellular carcinoma. Gastroenterology. 2018;154(1):128–39. https://doi.org/10.1053/j.gastro.2017.09.025.
Filgueira NA. Hepatocellular carcinoma recurrence after liver transplantation: risk factors, screening and clinical presentation. World J Hepatol. 2019;11(3):261–72. https://doi.org/10.4254/wjh.v11.i3.261.
Mazzaferro V, Llovet JM, Miceli R, Bhoori S, Schiavo M, Mariani L, et al. Predicting survival after liver transplantation in patients with hepatocellular carcinoma beyond the Milan criteria: a retrospective, exploratory analysis. Lancet Oncol. 2009;10(1):35–43. https://doi.org/10.1016/s1470-2045(08)70284-5.
Raj A, McCall J, Gane E. Validation of the “Metroticket” predictor in a cohort of patients transplanted for predominantly HBV-related hepatocellular carcinoma. J Hepatol. 2011;55(5):1063–8. https://doi.org/10.1016/j.jhep.2011.01.052.
Kornberg A, Küpper B, Tannapfel A, Büchler P, Krause B, Witt U, et al. Patients with non-[18 F]fludeoxyglucose-avid advanced hepatocellular carcinoma on clinical staging may achieve long-term recurrence-free survival after liver transplantation. Liver Transpl. 2012;18(1):53–61. https://doi.org/10.1002/lt.22416.
Takada Y, Kaido T, Shirabe K, Nagano H, Egawa H, Sugawara Y, et al. Significance of preoperative fluorodeoxyglucose-positron emission tomography in prediction of tumor recurrence after liver transplantation for hepatocellular carcinoma patients: a Japanese multicenter study. J Hepatobiliary Pancreat Sci. 2017;24(1):49–57. https://doi.org/10.1002/jhbp.412.
Hsu CC, Chen CL, Wang CC, Lin CC, Yong CC, Wang SH, et al. Combination of FDG-PET and UCSF criteria for predicting HCC recurrence after living donor liver transplantation. Transplantation. 2016;100(9):1925–32. https://doi.org/10.1097/tp.0000000000001297.
Marrero JA, Fontana RJ, Barrat A, Askari F, Conjeevaram HS, Su GL, et al. Prognosis of hepatocellular carcinoma: comparison of 7 staging systems in an American cohort. Hepatology. 2005;41(4):707–16. https://doi.org/10.1002/hep.20636.
Toso C, Meeberg G, Hernandez-Alejandro R, Dufour JF, Marotta P, Majno P, et al. Total tumor volume and alpha-fetoprotein for selection of transplant candidates with hepatocellular carcinoma: a prospective validation. Hepatology. 2015;62(1):158–65. https://doi.org/10.1002/hep.27787.
Duvoux C, Roudot-Thoraval F, Decaens T, Pessione F, Badran H, Piardi T, et al. Liver transplantation for hepatocellular carcinoma: a model including α-fetoprotein improves the performance of Milan criteria. Gastroenterology. 2012;143(4):986–94.e3; quiz e14–5. https://doi.org/10.1053/j.gastro.2012.05.052.
Giannini EG, Marenco S, Borgonovo G, Savarino V, Farinati F, Del Poggio P, et al. Alpha-fetoprotein has no prognostic role in small hepatocellular carcinoma identified during surveillance in compensated cirrhosis. Hepatology. 2012;56(4):1371–9. https://doi.org/10.1002/hep.25814.
Agopian VG, Harlander-Locke M, Zarrinpar A, Kaldas FM, Farmer DG, Yersiz H, et al. A novel prognostic nomogram accurately predicts hepatocellular carcinoma recurrence after liver transplantation: analysis of 865 consecutive liver transplant recipients. J Am Coll Surg. 2015;220(4):416–27. https://doi.org/10.1016/j.jamcollsurg.2014.12.025.
Kim YI, Paeng JC, Cheon GJ, Suh KS, Lee DS, Chung JK, et al. Prediction of Posttransplantation recurrence of hepatocellular carcinoma using metabolic and volumetric indices of 18F-FDG PET/CT. J Nucl Med. 2016;57(7):1045–51. https://doi.org/10.2967/jnumed.115.170076.
Germani G, Gurusamy K, Garcovich M, Toso C, Fede G, Hemming A, et al. Which matters most: number of tumors, size of the largest tumor, or total tumor volume? Liver Transpl. 2011;17(Suppl 2):S58–66. https://doi.org/10.1002/lt.22336.
Welling TH, Eddinger K, Carrier K, Zhu D, Kleaveland T, Moore DE, et al. Multicenter study of staging and therapeutic predictors of hepatocellular carcinoma recurrence following transplantation. Liver Transpl. 2018;24(9):1233–42. https://doi.org/10.1002/lt.25194.
Song JY, Lee YN, Kim YS, Kim SG, Jin SJ, Park JM, et al. Predictability of preoperative 18F-FDG PET for histopathological differentiation and early recurrence of primary malignant intrahepatic tumors. Nucl Med Commun. 2015;36(4):319–27. https://doi.org/10.1097/MNM.0000000000000254.
Jiang HY, Chen J, Xia CC, Cao LK, Duan T, Song B. Noninvasive imaging of hepatocellular carcinoma: from diagnosis to prognosis. World J Gastroenterol. 2018;24(22):2348–62. https://doi.org/10.3748/wjg.v24.i22.2348.
Lee SM, Kim HS, Lee S, Lee JW. Emerging role of 18F-fluorodeoxyglucose positron emission tomography for guiding management of hepatocellular carcinoma. World J Gastroenterol. 2019;25(11):1289–306. https://doi.org/10.3748/wjg.v25.i11.1289.
Park JW, Kim JH, Kim SK, Kang KW, Park KW, Choi JI, et al. A prospective evaluation of 18F-FDG and 11C-acetate PET/CT for detection of primary and metastatic hepatocellular carcinoma. J Nucl Med. 2008;49(12):1912–21. https://doi.org/10.2967/jnumed.108.055087.
Ho CL, Yu SC, Yeung DW. 11C-acetate PET imaging in hepatocellular carcinoma and other liver masses. J Nucl Med. 2003;44(2):213–21. https://doi.org/10.1016/S0921-4526(02)01648-4.
Yamamoto Y, Nishiyama Y, Kameyama R, Okano K, Kashiwagi H, Deguchi A, et al. Detection of hepatocellular carcinoma using 11C-choline PET: comparison with 18F-FDG PET. J Nucl Med. 2008;49(8):1245–8. https://doi.org/10.2967/jnumed.108.052639.
Talbot JN, Fartoux L, Balogova S, Nataf V, Kerrou K, Gutman F, et al. Detection of hepatocellular carcinoma with PET/CT: a prospective comparison of 18F-fluorocholine and 18F-FDG in patients with cirrhosis or chronic liver disease. J Nucl Med. 2010;51(11):1699–706. https://doi.org/10.2967/jnumed.110.075507.
Guniganti P, Kierans AS. PET/MRI of the hepatobiliary system: review of techniques and applications. Clin Imaging. 2021;71:160–9. https://doi.org/10.1016/j.clinimag.2020.10.056.
Kang YK, Choi JY, Paeng JC, Kim YI, Kwon HW, Cheon GJ, et al. Composite criteria using clinical and FDG PET/CT factors for predicting recurrence of hepatocellular carcinoma after living donor liver transplantation. Eur Radiol. 2019;29(11):6009–17. https://doi.org/10.1007/s00330-019-06239-z.
Chidambaranathan-Reghupaty S, Fisher PB, Sarkar D. Hepatocellular carcinoma (HCC): epidemiology, etiology and molecular classification. Adv Cancer Res. 2021;149:1–61. https://doi.org/10.1016/bs.acr.2020.10.001.
Author information
Authors and Affiliations
Contributions
All the authors read and approved the final manuscript.
Literature search: Wenjie Miao; Study design: Guangjie Yang, Wei Rao, Zhenguang Wang; Data collection: Yangyang Wang, Yan Lei, Yujun Zhao, Ting Yu, Mingming Yu, Fengyu Wu; Data analysis: Wenjie Miao, Guangjie Yang; Manuscript writing: Wenjie Miao; Manuscript review: Pei Nie, Guangjie Yang, Wei Rao, Zhenguang Wang.
Corresponding authors
Ethics declarations
Research involving human participants
All procedures performed on human participants were in accordance with the ethical standards of the Institutional Research Committee of the Affiliated Hospital of Qingdao University (Approval No. QYFY WZLL 25649) and the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was waived for this retrospective study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Oncology - Digestive tract
Rights and permissions
About this article
Cite this article
Miao, W., Nie, P., Yang, G. et al. An FDG PET/CT metabolic parameter-based nomogram for predicting the early recurrence of hepatocellular carcinoma after liver transplantation. Eur J Nucl Med Mol Imaging 48, 3656–3665 (2021). https://doi.org/10.1007/s00259-021-05328-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-021-05328-w



