Abstract
Purpose
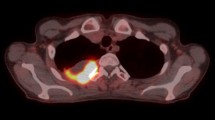
Inflammatory FDG uptake in the lung (PET-pneumonitis) following curative-intent radiotherapy (RT)/chemo-RT (CRT) in non-small cell lung cancer (NSCLC) can pose a challenge in FDG-PET/CT response assessment. The aim of this study is to describe different patterns of PET-pneumonitis to guide the interpretation of FDG-PET/CT and investigate its association with tumor response and overall survival (OS).
Methods
Retrospective analysis was performed on 87 NSCLC patients in three prospective trials who were treated with radical RT (n = 7) or CRT (n = 80), with baseline and post-treatment FDG-PET/CT. Visual criteria were performed for post-treatment FDG-PET/CT response assessment. The grading of PET-pneumonitis was based on relative lung uptake intensity compared to organs of reference and classified as per Deauville score from grade 1–5. Distribution patterns of PET-pneumonitis were defined as follows: A) patchy/sub-pleural; B) diffuse (involving more than a segment); and C) peripheral (diffusely surrounding a photopenic region).
Results
Follow-up FDG-PET/CT scans were performed approximately 3 months (median, 89 days; interquartile range, 79–93) after RT. Overall, PET-pneumonitis was present in 62/87 (71%) of patients, with Deauville 2 or 3 in 12/62 (19%) and 4 or 5 in 50/62 (81%) of patients. The frequency of patterns A, B and C of PET-pneumonitis was 19/62 (31%), 20/62 (32%) and 23/62 (37%), respectively. No association was found between grade or pattern of PET-pneumonitis and overall response at follow-up PET/CT (p = 0.27 and p = 0.56, respectively). There was also no significant association between PET-pneumonitis and OS (hazard ratio [HR], 1.3; 95% confidence interval [CI], 0.6–2.5; p = 0.45). Early FDG-PET/CT response assessment, however, was prognostic for OS (HR, 1.7; 95% CI, 1.2–2.2; p < 0.001).
Conclusion
PET-pneumonitis is common in early post-CRT/RT, but pattern recognition may assist in response assessment by FDG-PET/CT. While FDG-PET/CT is a powerful tool for response assessment and prognostication, PET-pneumonitis does not appear to confound early response assessment or to independently predict OS.



Similar content being viewed by others
References
Hicks RJ. Role of 18F-FDG PET in assessment of response in non-small cell lung cancer. J Nucl Med Off Publ Soc Nucl Med. 2009;50(Suppl 1):31S–42S. https://doi.org/10.2967/jnumed.108.057216.
Mac Manus MP, Hicks RJ, Matthews JP, McKenzie A, Rischin D, Salminen EK, et al. Positron emission tomography is superior to computed tomography scanning for response-assessment after radical radiotherapy or chemoradiotherapy in patients with non-small-cell lung cancer. J Clin Oncol Off J Am Soc Clin Oncol. 2003;21:1285–92. https://doi.org/10.1200/JCO.2003.07.054.
Gotwals P, Cameron S, Cipolletta D, Cremasco V, Crystal A, Hewes B, et al. Prospects for combining targeted and conventional cancer therapy with immunotherapy. Nat Rev Cancer. 2017;17:286–301. https://doi.org/10.1038/nrc.2017.17.
Van Breussegem A, Hendriks JM, Lauwers P, Van Schil PE. Salvage surgery after high-dose radiotherapy. J Thorac Dis. 2017;9:S193–200. https://doi.org/10.21037/jtd.2017.03.88.
Bergsma DP, Salama JK, Singh DP, Chmura SJ, Milano MT. Radiotherapy for oligometastatic lung cancer. Front Oncol. 2017;7:210. https://doi.org/10.3389/fonc.2017.00210.
Hicks RJ, Mac Manus MP, Matthews JP, Hogg A, Binns D, Rischin D, et al. Early FDG-PET imaging after radical radiotherapy for non-small-cell lung cancer: inflammatory changes in normal tissues correlate with tumor response and do not confound therapeutic response evaluation. Int J Radiat Oncol Biol Phys. 2004;60:412–8. https://doi.org/10.1016/j.ijrobp.2004.03.036.
Caulo A, Mirsadraee S, Maggi F, Leccisotti L, van Beek EJ, Bonomo L. Integrated imaging of non-small cell lung cancer recurrence: CT and PET-CT findings, possible pitfalls and risk of recurrence criteria. Eur Radiol. 2012;22:588–606. https://doi.org/10.1007/s00330-011-2299-8.
Sudarski S, Henzler T, Schoenberg SO. Post-therapeutic positron emission tomography/computed tomography for early detection of non-small cell lung cancer recurrence. Transl Lung Cancer Res. 2013;2:295–303. https://doi.org/10.3978/j.issn.2218-6751.2013.05.02.
Turgeon G, Iravani A, Akhurst T, Beaulieu A, Callahan J, Bressel M, et al. What FDG-PET response-assessment method best predicts survival after curative-intent chemoradiation in non-small cell lung cancer (NSCLC): EORTC, PERCIST, Peter Mac or Deauville criteria? J Nucl Med. 2018. https://doi.org/10.2967/jnumed.118.214148.
Chun SG, Hu C, Choy H, Komaki RU, Timmerman RD, Schild SE, et al. Impact of intensity-modulated radiation therapy technique for locally advanced non-small-cell lung cancer: a secondary analysis of the NRG oncology RTOG 0617 randomized clinical trial. J Clin Oncol Off J Am Soc Clin Oncol. 2017;35:56–62. https://doi.org/10.1200/JCO.2016.69.1378.
Mac Manus MP, Everitt S, Bayne M, Ball D, Plumridge N, Binns D, et al. The use of fused PET/CT images for patient selection and radical radiotherapy target volume definition in patients with non-small cell lung cancer: results of a prospective study with mature survival data. Radiother Oncol J Eur Soc Ther Radiol Oncol. 2013;106:292–8. https://doi.org/10.1016/j.radonc.2012.12.018.
Everitt S, Ball D, Hicks RJ, Callahan J, Plumridge N, Trinh J, et al. Prospective study of serial imaging comparing fluorodeoxyglucose positron emission tomography (PET) and fluorothymidine PET during radical chemoradiation for non-small cell lung cancer: reduction of detectable proliferation associated with worse survival. Int J Radiat Oncol Biol Phys. 2017;99:947–55. https://doi.org/10.1016/j.ijrobp.2017.07.035.
Siva S, Callahan J, Kron T, Martin OA, MacManus MP, Ball DL, et al. A prospective observational study of Gallium-68 ventilation and perfusion PET/CT during and after radiotherapy in patients with non-small cell lung cancer. BMC Cancer. 2014;14:740. https://doi.org/10.1186/1471-2407-14-740.
De Ruysscher D, Houben A, Aerts HJ, Dehing C, Wanders R, Ollers M, et al. Increased (18)F-deoxyglucose uptake in the lung during the first weeks of radiotherapy is correlated with subsequent radiation-induced lung toxicity (RILT): a prospective pilot study. Radiother Oncol J Eur Soc Ther Radiol Oncol. 2009;91:415–20. https://doi.org/10.1016/j.radonc.2009.01.004.
Antonia SJ, Villegas A, Daniel D, Vicente D, Murakami S, Hui R, et al. Durvalumab after chemoradiotherapy in stage III non-small-cell lung cancer. N Engl J Med. 2017;377:1919–29. https://doi.org/10.1056/NEJMoa1709937.
Arnett AL, Packard AT, Mara K, Mansfield AS, Wigle DA, Haddock MG, et al. FDG-PET parameters as predictors of pathologic response and nodal clearance in patients with stage III non-small cell lung cancer receiving neoadjuvant chemoradiation and surgery. Practical Radiat Oncol. 2017;7:e531–e41. https://doi.org/10.1016/j.prro.2017.04.013.
Badiyan SN, Roach MC, Chuong MD, Rice SR, Onyeuku NE, Remick J, et al. Combining immunotherapy with radiation therapy in thoracic oncology. J Thorac Dis. 2018;10:S2492–S507. https://doi.org/10.21037/jtd.2018.05.73.
Turgeon GA, Weickhardt A, Azad AA, Solomon B, Siva S. Radiotherapy and immunotherapy: a synergistic effect in cancer care. Med J Aust. 2019;210:47–53. https://doi.org/10.5694/mja2.12046.
Antonia SJ, Villegas A, Daniel D, Vicente D, Murakami S, Hui R, et al. Overall survival with durvalumab after chemoradiotherapy in stage III NSCLC. N Engl J Med. 2018;379:2342–50. https://doi.org/10.1056/NEJMoa1809697.
Siva S, MacManus M, Kron T, Best N, Smith J, Lobachevsky P, et al. A pattern of early radiation-induced inflammatory cytokine expression is associated with lung toxicity in patients with non-small cell lung cancer. PLoS One. 2014;9:e109560. https://doi.org/10.1371/journal.pone.0109560.
Abravan A, Eide HA, Knudtsen IS, Londalen AM, Helland A, Malinen E. Assessment of pulmonary (18)F-FDG-PET uptake and cytokine profiles in non-small cell lung cancer patients treated with radiotherapy and erlotinib. Clin Transl Radiat Oncol. 2017;4:57–63. https://doi.org/10.1016/j.ctro.2017.04.002.
Weller A, O’Brien MER, Ahmed M, Popat S, Bhosle J, McDonald F, et al. Mechanism and non-mechanism based imaging biomarkers for assessing biological response to treatment in non-small cell lung cancer. Eur J Cancer. 2016;59:65–78. https://doi.org/10.1016/j.ejca.2016.02.017.
Jahangiri P, Pournazari K, Torigian DA, Werner TJ, Swisher-McClure S, Simone CB 2nd, et al. A prospective study of the feasibility of FDG-PET/CT imaging to quantify radiation-induced lung inflammation in locally advanced non-small cell lung cancer patients receiving proton or photon radiotherapy. Eur J Nucl Med Mol Imaging. 2019;46:206–16. https://doi.org/10.1007/s00259-018-4154-5.
Guerrero T, Johnson V, Hart J, Pan T, Khan M, Luo D, et al. Radiation pneumonitis: local dose versus [18F]-fluorodeoxyglucose uptake response in irradiated lung. Int J Radiat Oncol Biol Phys. 2007;68:1030–5. https://doi.org/10.1016/j.ijrobp.2007.01.031.
Siva S, Callahan JW, Kron T, Chesson B, Barnett SA, Macmanus MP, et al. Respiratory-gated (4D) FDG-PET detects tumour and normal lung response after stereotactic radiotherapy for pulmonary metastases. Acta Oncol. 2015;54:1105–12. https://doi.org/10.3109/0284186X.2015.1027409.
Ypsilantis PP, Siddique M, Sohn HM, Davies A, Cook G, Goh V, et al. Predicting response to neoadjuvant chemotherapy with PET imaging using convolutional neural networks. PLoS One. 2015;10:e0137036. https://doi.org/10.1371/journal.pone.0137036.
Funding
No funding was received for this study. However, the 18F-FLT/18F-FDG study (Australian Clinical Trials Registry: ACTRN12611001283965) work was supported by the National Health and Medical Research Council (NHMRC) (grant number APP1003895) and the Victorian Cancer Agency. The 68Ga-Ventilation/perfusion PET (Universal Trial Number: U1111-1138-4421) study was supported by funding from the NHMRC (grant number APP1038399) and from the Cancer Australia Priority-driven Collaborative Cancer Research Scheme (Project No. 1060919). Professor Hicks is supported by an NHMRC Practitioner Fellowship (APP1108050).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
We wish to confirm that there are no known conflicts of interest associated with this publication. A Iravani, GA Turgeon, T Akhurst, JW Callahan, M Bressel, SJ Everitt and MP Mac Manus have no disclosure. Through his institution, DL Ball has an advisory role for Pfizer Australia. RJ Hicks has ownership interest in Telix Radiopharmaceuticals. Through his institution, S Siva has an advisory role for Astellas Pharmaceuticals and Janssen Pharmaceuticals and receives research funding from Varian Medical Systems and Merck Sharp & Dohme Pharmaceuticals. S Siva received speakers’ bureau, travel/accommodations/expenses from Bristol-Myers Squibb Pharmaceuticals as well as travel/accommodation/expenses from Astellas Pharmaceuticals.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
The registration number for the prospective studies were as follows, PET-planning study: Peter Mac protocol 03/55; the 18F-FLT/18F-FDG study: Australian Clinical Trials Registry no. ACTRN12611001283965; and the 68Ga-ventilation/perfusion PET study: universal trial number U1111-1138-4421. The Peter MacCallum Cancer Clinical Research and Ethics Committee approved this retrospective study and waived the requirement to obtain informed consent (approval number: PMCC 17/43R). All patients had previously provided written informed consent to undergo their respective prospective studies.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Oncology – Chest
Electronic supplementary material
ESM 1
(DOCX 19 kb)
Rights and permissions
About this article
Cite this article
Iravani, A., Turgeon, GA., Akhurst, T. et al. PET-detected pneumonitis following curative-intent chemoradiation in non-small cell lung cancer (NSCLC): recognizing patterns and assessing the impact on the predictive ability of FDG-PET/CT response assessment. Eur J Nucl Med Mol Imaging 46, 1869–1877 (2019). https://doi.org/10.1007/s00259-019-04388-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-019-04388-3