Abstract
Purpose
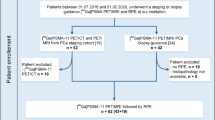
Given the good correlation between PSMA expression and intraglandular tumour aggressiveness based on immunohistochemistry, there is increasing interest in 68Ga-PSMA-11 PET/MRI for staging prostate cancer (PCA). Therefore, accurate knowledge of prostate anatomy as well as normal distribution of PSMA within the prostate gland is becoming essential. The aim of this study was to investigate the physiological intraprostatic distribution of 68Ga-PSMA-11.
Methods
We retrospectively analysed all patients who underwent a staging 68Ga-PSMA-11 PET/MRI scan between June 2016 and January 2018 for high-risk PCA, underwent radical prostatectomy in our institution, and gave written consent for further data analysis. In each patient, standardized volumes of interest (VOIs) were placed bilaterally in the central, transition and peripheral zones within the zonal anatomy according to T2 weighted sequences in the axial and coronal planes. VOIs were only placed if they were safely within healthy tissue without spillover from the PCA. SUVmax and SUVmean were determined and their differences among the regions were assessed using the Wilcoxon signed-ranks test.
Results
Of 283 consecutive patients scanned with 68Ga-PSMA-11 PET/MR, 31 were analysed. A total of 133 VOIs were placed, 46 in the central zone, 41 in the transition zone and 46 in the peripheral zone. Differences in SUVmax between the central zone (mean 3.9 ± 0.58) and transition zone (mean 3.2 ± 0.59) and between the central zone and peripheral zone (mean 2.7 ± 0.54) were statistically significant (both p < 0.001).
Conclusion
Our results suggest that higher 68Ga-PSMA-11 accumulation in the central zone than in the transition and peripheral zones is normal, and leads to a pattern resembling “Mickey Mouse ears” on 68Ga-PSMA-11 PET. This pattern could be helpful in avoiding false-positive interpretations of PET scans.





Similar content being viewed by others
References
Cabarrus MC, Westphalen AC. Multiparametric magnetic resonance imaging of the prostate – a basic tutorial. Transl Androl Urol. 2017;6:376–86. https://doi.org/10.21037/tau.2017.01.06.
Becker AS, Cornelius A, Reiner CS, Stocker D, Ulbrich EJ, Barth BK, et al. Direct comparison of PI-RADS version 2 and version 1 regarding interreader agreement and diagnostic accuracy for the detection of clinically significant prostate cancer. Eur J Radiol. 2017;94:58–63. https://doi.org/10.1016/j.ejrad.2017.07.016.
Afshar-Oromieh A, Avtzi E, Giesel FL, Holland-Letz T, Linhart HG, Eder M, et al. The diagnostic value of PET/CT imaging with the (68)Ga-labelled PSMA ligand HBED-CC in the diagnosis of recurrent prostate cancer. Eur J Nucl Med Mol Imaging. 2015;42:197–209. https://doi.org/10.1007/s00259-014-2949-6.
Eiber M, Maurer T, Souvatzoglou M, Beer AJ, Ruffani A, Haller B, et al. Evaluation of hybrid 68Ga-PSMA ligand PET/CT in 248 patients with biochemical recurrence after radical prostatectomy. J Nucl Med. 2015;56:668–74. https://doi.org/10.2967/jnumed.115.154153.
Afshar-Oromieh A, Holland-Letz T, Giesel FL, Kratochwil C, Mier W, Haufe S, et al. Diagnostic performance of (68)Ga-PSMA-11 (HBED-CC) PET/CT in patients with recurrent prostate cancer: evaluation in 1007 patients. Eur J Nucl Med Mol Imaging. 2017;44:1258–68. https://doi.org/10.1007/s00259-017-3711-7.
Kasperzyk JL, Finn SP, Flavin R, Fiorentino M, Lis R, Hendrickson WK, et al. Prostate-specific membrane antigen protein expression in tumor tissue and risk of lethal prostate cancer. Cancer Epidemiol Biomarkers Prev. 2013;22:2354–63. https://doi.org/10.1158/1055-9965.EPI-13-0668.
Marchal C, Redondo M, Padilla M, Caballero J, Rodrigo I, Garcia J, et al. Expression of prostate specific membrane antigen (PSMA) in prostatic adenocarcinoma and prostatic intraepithelial neoplasia. Histol Histopathol. 2004;19:715–8. https://doi.org/10.14670/HH-19.715.
Eiber M, Nekolla SG, Maurer T, Weirich G, Wester HJ, Schwaiger M. (68)Ga-PSMA PET/MR with multimodality image analysis for primary prostate cancer. Abdom Imaging. 2015;40:1769–71. https://doi.org/10.1007/s00261-014-0301-z.
Zamboglou C, Drendel V, Jilg CA, Rischke HC, Beck TI, Schultze-Seemann W, et al. Comparison of 68Ga-HBED-CC PSMA-PET/CT and multiparametric MRI for gross tumour volume detection in patients with primary prostate cancer based on slice by slice comparison with histopathology. Theranostics. 2017;7:228–37. https://doi.org/10.7150/thno.16638.
Rahbar K, Weckesser M, Huss S, Semjonow A, Breyholz HJ, Schrader AJ, et al. Correlation of intraprostatic tumor extent with 68Ga-PSMA distribution in patients with prostate cancer. J Nucl Med. 2016;57:563–7. https://doi.org/10.2967/jnumed.115.169243.
Eiber M, Weirich G, Holzapfel K, Souvatzoglou M, Haller B, Rauscher I, et al. Simultaneous 68Ga-PSMA HBED-CC PET/MRI improves the localization of primary prostate cancer. Eur Urol. 2016;70:829–36. https://doi.org/10.1016/j.eururo.2015.12.053.
Uprimny C, Kroiss AS, Decristoforo C, Fritz J, von Guggenberg E, Kendler D, et al. (68)Ga-PSMA-11 PET/CT in primary staging of prostate cancer: PSA and Gleason score predict the intensity of tracer accumulation in the primary tumour. Eur J Nucl Med Mol Imaging. 2017;44:941–9. https://doi.org/10.1007/s00259-017-3631-6.
Vargas HA, Akin O, Franiel T, Goldman DA, Udo K, Touijer KA, et al. Normal central zone of the prostate and central zone involvement by prostate cancer: clinical and MR imaging implications. Radiology. 2012;262:894–902. https://doi.org/10.1148/radiol.11110663.
Sekine T, Barbosa FD, Sah BR, Mader CE, Delso G, Burger IA, et al. PET/MR outperforms PET/CT in suspected occult tumors. Clin Nucl Med. 2017;42:E88–95. https://doi.org/10.1097/Rlu.0000000000001461.
Wollenweber SD, Ambwani S, Delso G, Lonn AHR, Mullick R, Wiesinger F, et al. Evaluation of an atlas-based PET head attenuation correction using PET/CT and MR patient data. IEEE Trans Nucl Sci. 2013;60:3383–90. https://doi.org/10.1109/Tns.2013.2273417.
Wollenweber SD, Ambwani S, Lonn AHR, Shanbhag DD, Thiruvenkadam S, Kaushik S, et al. Comparison of 4-class and continuous fat/water methods for whole-body, MR-based PET attenuation correction. IEEE Trans Nucl Sci. 2013;60:3391–8. https://doi.org/10.1109/Tns.2013.2278759.
Hricak H, Dooms GC, McNeal JE, Mark AS, Marotti M, Avallone A, et al. MR imaging of the prostate gland: normal anatomy. AJR Am J Roentgenol. 1987;148:51–8. https://doi.org/10.2214/ajr.148.1.51.
Argani P, Walsh PC, Epstein JI. Analysis of the prostatic central zone in patients with unilateral absence of wolffian duct structures: further evidence of the mesodermal origin of the prostatic central zone. J Urol. 1998;160:2126–9.
Leung CS, Srigley JR. Distribution of lipochrome pigment in the prostate gland: biological and diagnostic implications. Hum Pathol. 1995;26:1302–7.
Chen ME, Johnston DA, Tang K, Babaian RJ, Troncoso P. Detailed mapping of prostate carcinoma foci: biopsy strategy implications. Cancer. 2000;89:1800–9.
Mai KT, Belanger EC, Al-Maghrabi HM, Robertson S, Wang D, Margnean C. Primary prostatic central zone adenocarcinoma. Pathol Res Pract. 2008;204:251–8. https://doi.org/10.1016/j.prp.2007.11.002.
Cohen RJ, Shannon BA, Phillips M, Moorin RE, Wheeler TM, Garrett KL. Central zone carcinoma of the prostate gland: a distinct tumor type with poor prognostic features. J Urol. 2008;179:1762–7; discussion 7. https://doi.org/10.1016/j.juro.2008.01.017.
Afshar-Oromieh A, Haberkorn U, Schlemmer HP, Fenchel M, Eder M, Eisenhut M, et al. Comparison of PET/CT and PET/MRI hybrid systems using a 68Ga-labelled PSMA ligand for the diagnosis of recurrent prostate cancer: initial experience. Eur J Nucl Med Mol Imaging. 2014;41:887–97. https://doi.org/10.1007/s00259-013-2660-z.
Lutje S, Blex S, Gomez B, Schaarschmidt BM, Umutlu L, Forsting M, et al. Optimization of acquisition time of 68Ga-PSMA-ligand PET/MRI in patients with local and metastatic prostate cancer. PLoS One. 2016;11:e0164392. https://doi.org/10.1371/journal.pone.0164392.
Afshar-Oromieh A, Wolf M, Haberkorn U, Kachelriess M, Gnirs R, Kopka K, et al. Effects of arm truncation on the appearance of the halo artifact in (68)Ga-PSMA-11 (HBED-CC) PET/MRI. Eur J Nucl Med Mol Imaging. 2017;44:1636–46. https://doi.org/10.1007/s00259-017-3718-0.
Acknowledgments
The authors acknowledge the technicians Marlena Hofbauer, Miguel Porto, Sofia Kaltsuni, Tobias Oblasser, Sabrina Epp, Michele Hug and Melanie Thüringer for their excellent work providing high-quality PET/MRI data. The Department of Nuclear Medicine holds an institutional Research Contract with GE Healthcare.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
I.A.B. received research grants and speaker honoraria from GE Healthcare. All other authors declare no conflicts of interest.
Ethical approval
This retrospective study was approved by the local ethics committee (BASEC Nr. 2016-02231).
Informed consent
All patients gave a written informed general consent for retrospective analysis of their data.
Electronic supplementary material
Supplementary Fig. 1
All patients with high urinary activity (SUVmax >25) are included to demonstrate the lack of any halo artefact despite high accumulation of 68Ga-PSMA-11 in the bladder. Axial and coronal slices are shown at the level of the bladder using a PET window of 0–8 g/ml in (a) patient 13 (SUVmax 27), (b) patient 17 (SUVmax 32), (c) patient 9 (SUVmax 33), and (d) patient 23 (SUVmax 57) (GIF 125 kb)
Rights and permissions
About this article
Cite this article
Pizzuto, D.A., Müller, J., Mühlematter, U. et al. The central zone has increased 68Ga-PSMA-11 uptake: “Mickey Mouse ears” can be hot on 68Ga-PSMA-11 PET. Eur J Nucl Med Mol Imaging 45, 1335–1343 (2018). https://doi.org/10.1007/s00259-018-3979-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-018-3979-2