Abstract
Aim
In intermediate- or high-risk prostate cancer (PC) patients, to avoid extended pelvic lymph node dissection (ePLND), the updated Briganti nomogram is recommended with the cost of missing 1.5 % of patients with lymph node invasion (LNI). Is it possible to reduce the percentage of unexpected LNI patients (nomogram false negative)? We used the isotopic sentinel lymph node (SLN) technique systematically associated with laparoscopic ePLND to assess the potential value of isotopic SLN method to adress this point.
Methods
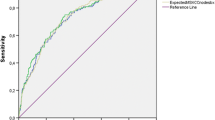
Two hundred and two consecutive patients had procedures with isotopic SLN detection associated with laparoscopic ePLND for high or intermediate risk of PC. The area under the curve (AUC) of the receiver operating characteristics (ROC) analysis was used to quantify the accuracy of different models as: the updated Briganti nomogram, the percentage of positive cores, and an equation of the best predictors of LNI. We tested the model cutoffs associated with an optimal negative predictive value (NPV) and the best cutoff associated with avoiding false negative SLN detection, in order to assist the clinician’s decision of when to spare ePLND.
Results
LNI was detected in 35 patients (17.2 %). Based on preoperative primary Gleason grade and percentage of positive cores, a bivariate model was built to calculate a combined score reflecting the risk of LNI. For the Briganti nomogram, the 5 % probability cutoff avoided ePLND in 53 % (108/202) of patients, missing three LNI patients (8.6 %), but all were detected by the SLN technique. For our bivariate model, the best cutoff was <10, leaving no patient with LNI due to positive SLN detection (four patients = 11.4 %), and avoiding ePLND in 52 % (105/202) of patients.
Conclusion
For patients with a low risk of LNI determined using the updated Briganti nomogram or bivariate model, SLN technique could be used alone for lymph node staging in intermediate- or high-risk PC patients.



Similar content being viewed by others
References
Bubendorf L, Schöpfer A, Wagner U, Sauter G, Moch H, Willi N, et al. Metastatic patterns of prostate cancer: an autopsy study of 1,589 patients. Hum Pathol. 2000;31:578–83.
Cheng L, Zincke H, Blute M, Bergstralh E, Scherer B, Bostwick D. Risk of prostate carcinoma death in patients with lymph node metastasis. Am Cancer Soc. 2001;91:66–73.
Heidenreich A, Aus G, Bolla M, Joniau S, Matveer VB, Schmid HP, et al. EAU guidelines on prostate cancer. Eur Urol. 2008;53:68–80.
Briganti A, Chun FK-H, Salonia A, Suardi N, Gallina A, Da Pozzo LF, et al. Complications and other surgical outcomes associated with extended pelvic lymphadenectomy in men with localized prostate cancer. Eur Urol. 2006;50:1006–13.
Van Baelen A, Mottet N, Spahn M, Briganti A, Gontero P, Joniau S. Sense and nonsense of an extended pelvic lymph node dissection in prostate cancer. Adv Urol. 2012;2012:983058.
Briganti A, Capitanio U, Abdollah F, Gallina A, Suardi N, Bianchi M, et al. Assessing the risk of lymph node invasion in patients with intermediate risk prostate cancer treated with extended pelvic lymph node dissection. A novel prediction tool. Prostate. 2012;72(5):499–506.
Briganti A, Suardi N, Gallina A, Abdollah F, Montorsi F. Pelvic lymph node dissection in prostate cancer: the mystery is taking shape. Eur Urol. 2013;63(3):459–61.
Heidenreich A, Bastian PJ, Bellmunt J, Bolla M, Joniau S, van der Kwast T, et al. EAU guidelines on prostate cancer. part 1: screening, diagnosis, and local treatment with curative intent-update 2013. Eur Urol. 2014;65(1):124–37.
Briganti A, Larcher A, Abdollah F, Capitanio U, Gallina A, Suardi N, et al. Updated nomogram predicting lymph node invasion in patients with prostate cancer undergoing extended pelvic lymph node dissection: the essential importance of percentage of positive cores. Eur Urol. 2012;61(3):480–7.
Acar C, Kleinjan GH, van den Berg NS, Wit EM, van Leeuwen FW, van der Poel HG. Advances in sentinel node dissection in prostate cancer from a technical perspective. Int J Urol. 2015;22(10):898–909.
Winter A, Kneib T, Henke RP, Wawroschek F. Sentinel lymph node dissection in more than 1200 prostate cancer cases: rate and prediction of lymph node involvement depending on preoperative tumor characteristics. Int J Urol. 2013;21(1):58–63.
Rousseau C, Rousseau T, Campion L, Lacoste J, Aillet G, Potiron E, et al. Laparoscopic sentinel lymph node versus hyperextensive pelvic dissection for staging clinically localized prostate carcinoma: a prospective study of 200 patients. J Nucl Med. 2014;55(5):753–8.
D’Amico AV, Whittington R, Malkowicz SB, Tomaszewski JE, Schultz D, Wein A. Outcome based staging for clinically localizedadenocarcinomaof the prostate. J Urol. 1997;158:1422–6.
Rousseau T, Lacoste J, Pallardy A, Campion L, Bridji B, Mouaden A, et al. Laparoscopic sentinel lymph node (SLN) dissection for clinically localized prostate carcinoma: results obtained in the first 70 patients. Prog Urol. 2011;22:30–7.
Rousseau C, Rousseau T, Bridji B, et al. Laparoscopic sentinel lymph node (SLN) versus extensive pelvic dissection for clinically localized prostate carcinoma. Eur J Nucl Med Mol Imaging. 2012;39(2):291–9.
Fujisawa M, Miyake H. Significance of micrometastases in prostate cancer. Surg Oncol. 2008;17:247–52.
Chéreau E, Bezu C, Gligorov J, Sakr R, Antoine M, Daraï E, et al. Impact of immunohistochemical analysis of sentinel lymph node biopsy on breast cancer management. Anticancer Res. 2012;32:3403–9.
Hansen J, Rink M, Bianchi M, Kluth LA, Tian Z, Ahyai SA, et al. External validation of the updated Briganti nomogram to predict lymph node invasion in prostate cancer patients undergoing extended lymph node dissection. Prostate. 2013;73(2):211–8.
Gacci M, Schiavina R, Lanciotti M, Masieri L, Serni S, Vagnoni V, et al. External validation of the updated nomogram predicting lymph node invasion in patients with prostate cancer undergoing extended pelvic lymph node dissection. Urol Int. 2013;90(3):277–82.
Dell’Oglio P, Abdollah F, Suardi N, Gallina A, Cucchiara V, Vizziello D, et al. External validation of the European association of urology recommendations for pelvic lymph node dissection in patients treated with robot-assisted radical prostatectomy. J Endourol. 2014;28(4):416–23.
Acknowledgments
This work has been supported by grants from the French National Agency for Research called Investissements d’Avenir, Labex IRON no. ANR-11-LABX-0018-01 and Equipex ArronaxPlus no. ANR-11-EQPX-0004.
The authors’ gratitude goes to the patients of the study, the nuclear medicine technologists at the ICO Cancer Center, N. Fleury (clinical research associate) and E. Cerato (Project manager).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Rousseau, C., Rousseau, T., Mathieu, C. et al. Laparoscopic sentinel lymph node dissection in prostate cancer patients: the additional value depends on preoperative data. Eur J Nucl Med Mol Imaging 43, 1849–1856 (2016). https://doi.org/10.1007/s00259-016-3397-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-016-3397-2