Abstract
Purpose
Renal function can be quantified by both laboratory and scintigraphic methods. In the case of small animal diagnostics, scintigraphic image-based methods are ideal since they can assess split renal function, work noninvasively, and can be repeated. The aim of this study is to validate a 18F-PET-based method to quantify renal function in rats.
Materials and methods
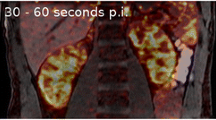
Fluoride clearance was calculated from a dynamic whole body listmode acquisition of 60 min length in a small animal PET scanner following an i.v. injection of 15 MBq 18F-fluoride. Volumes of interest (VOIs) were placed in the left ventricle and the bladder as well as traced around the kidney contours. The respective time–activity curves (TAC) were calculated. The renal 18F-clearance was calculated by the ratio of the total renal excreted activity (bladder VOI) and the integral of the blood TAC. PET-derived renal function was validated by intraindividual measurements of creatinine clearance (n = 23), urea clearance (n = 23), and tubular excretion rate (TER-MAG3). The split renal function was derived from the injection of the clinically available radionuclide 99mTc-mercaptotriglycine by blood sampling and planar renography (n = 8).
Results
In all animals studied, PET revealed high-quality TACs. PET-derived renal fluoride clearance was linearly correlated with intraindividual laboratory measures (PET vs. creatinine: r = 0.78; PET vs. urea: r = 0.73; PET vs. TER-MAG3: r = 0.73). Split function was comparable (18F-PET vs. MAG3-renography: r = 0.98). PET-derived measures were highly reproducible.
Conclusions
18F-PET is able to noninvasively assess renal function in rats and provides a significant potential for serial studies in different experimental scenarios.







Similar content being viewed by others
References
Rahn KH, Heidenreich S, Bruckner D. How to assess glomerular function and damage in humans. J Hypertens 1999;17:309–17.
Levey AS. Measurement of renal function in chronic renal disease. Kidney Int 1990;38:167–84.
Berlyne GM, Varley H, Nilwarangkur S, Hoerni M. Endogenous creatinine clearance and glomerula filtration rate. Lancet 1964;22:874–6.
Brochner-Mortensen J. Current status on assessment and measurement of glomerular filtration rate. Clin Physiol 1985;5:1–17.
Durand E, Prigent A. The basics of renal imaging and function studies. Q J Nucl Med 2002;46:249–67.
Boubaker A, Prior JO, Meuwly JY, Bischof-Delaloye A. Radionuclide investigations of the urinary tract in the era of multimodality imaging. J Nucl Med 2006;47:1819–36.
Lorenz JN. Considerations for the evaluation of renal function in genetically engineered mice. Curr Opin Nephrol Hypertens 2001;10:65–9.
Spak CJ, Berg U, Ekstrand J. Renal clearance of flouride in children and adolescents. Pediatrics 1985;75(3):575–9.
Whitford GM, Pashley DH, Stringer GI. Fluoride renal clearance: a pH-dependent event. Am J Physiol 1976;230:527–32.
Whitford GM. Intake and metabolism of fluoride. Adv Dent Res 1994;8:5–14.
Maisey M. Radionuclide renography: a review. Curr Opin Nephrol Hypertens 2003;12:649–52.
Wieland BW, Bida GT, Padgett HC, Go H. Current status of CTI target systems for the production of PET radiochemicals. Proceedings of the 3rd Workshop on Targetry and Target Chemistry, Vancouver, British Columbia, Canada, 19–23 June 1989.
Schäfers KP, Reader AJ, Kriens M, Knoess C, Schober O, Schafers M. Performance evaluation of the 32-module quadHIDAC small animal PET scanner. J Nucl Med 2005;46:996–1004.
Bubeck B, Piepenburg R, Grethe U, Ehrig B, Hahn K. A new principle to normalize plasma concentrations allowing single-sample clearance determinations in both children and adults. Eur J Nucl Med 1992;19:511–6.
Smuts D. B. Ein experimenteller Beitrag zur Berechnung der Körperoberfläche von Maus, Meerschweinchen und Kaninchen. Pflügers Archiv Eur J Physiol 1933;1:105–10.
Bland JM, Altman DG. Statistical methods for assessing agreement between two methods of clinical measurement. Lancet 1986;1:307–10.
Charkes ND, Brookes M, Makler PT Jr. Studies of skeletal tracer kinetics: II. Evaluation of a five-compartment model of [18F]fluoride kinetics in rats. J Nucl Med 1979;20:1150–7.
Park-Holohan SJ, Blake GM, Fogelman I. Quantitative studies of bone using (18)F-fluoride and (99 m)Tc-methylene diphosphonate: evaluation of renal and whole-blood kinetics. Nucl Med Commun 2001;22:1037–44.
Yang B, Bankir L. Urea and urine concentrating ability: new insights from studies in mice. Am J Physiol Renal Physiol 2005;288:881–96.
Darling IM, Morris ME. Evaluation of “true” creatinine clearance in rats reveals extensive renal secretion. Pharm Res 1991;8:1318–22.
Hawkins RA, Choi Y, Huang SC, Hoh CK, Dahlbom M, Schiepers C, Satyamurthy N, Barrio JR, Phelps ME. Evaluation of the skeletal kinetics of fluorine-18-fluoride ion with PET. J Nucl Med 1992;33:633–42.
Nemzek JA, Bolgos GL, Williams BA, Remick DG. Differences in normal values for murine white blood cell counts and other hematological parameters based on sampling site. Inflamm Res 2001;50(10):523–7.
Jarnberg PO, Ekstrand J, Irestedt L. Renal fluoride excretion and plasma fluoride levels during and after enflurane anesthesia are dependent on urinary pH. Anesthesiology 1981;54:48–52.
Rouch AJ, Whitford GM, Campbell HT. Fluoride flux in the rabbit CCD: a pH-dependent event. Kidney Int 1992;41:342–9.
Yagil Y, Myers BD, Jamison RL. Course and pathogenesis of postischemic acute renal failure in the rat. Am J Physiol 1988;255:257–64.
Kharasch ED, Frink EJ Jr, Artru A, Michalowski P, Rooke GA, Nogami W. Long-duration low-flow sevoflurane and isoflurane effects on postoperative renal and hepatic function. Anesth Analg 2001;93:1511–20.
Acknowledgements
The authors are grateful to Marilyn Law, Anne Kanzog, and Christine Bätza for excellent support, to Daniel Burkert and Sven Fatum for producing 18F-fluoride, and to Liam Bergin for language editing of the manuscript. The study was supported in part by the Interdisciplinary Centre of Clinical Research IZKF Münster (ZPG 4b), the Innovative Medizinische Forschung (IMF) Münster and the Else-Kröner-Fresenius Foundation (P22/05//A43/05//F00).
Author information
Authors and Affiliations
Corresponding author
Additional information
Uta Schnöckel and Stefan Reuter contributed equally to this study.
Rights and permissions
About this article
Cite this article
Schnöckel, U., Reuter, S., Stegger, L. et al. Dynamic 18F-fluoride small animal PET to noninvasively assess renal function in rats. Eur J Nucl Med Mol Imaging 35, 2267–2274 (2008). https://doi.org/10.1007/s00259-008-0878-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-008-0878-y