Abstract
Purpose
Recurrences are frequent in thyroid cancer patients and long-term follow-up is therefore necessary. We evaluated the yield of rhTSH stimulation in three groups of patients, classified according to the UICC/TNM risk stratification and the results of first follow-up testing.
Methods
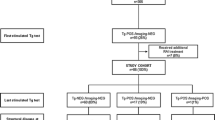
The study population comprised 129 patients referred for rhTSH testing. All had undergone first follow-up testing after thyroid hormone withdrawal (off-T4) within 1 year of 131I ablation. Negative first follow-up testing was defined as Tg <2 ng/ml and no neck uptake on 131I diagnostic whole-body scan. Seventy-five patients had stage I thyroid cancer and negative first follow-up testing (group A), 19 had stage I disease and positive first follow-up testing (group B), and 35 had stage II–IV disease (group C). RhTSH stimulation was performed an average of 6 years after first follow-up testing.
Results
131I diagnostic scanning after rhTSH was negative in all 75 group A patients. Only one group A patient had detectable Tg after rhTSH injection (1.5 ng/ml), but Tg had also been detected at baseline in this patient (1.45 ng/ml). Given the absence of a response to stimulation, suggesting an interference, Tg was reassessed with a different technique and proved to be undetectable (<0.1 ng/ml). Stimulation with rhTSH in group B showed residual Tg in seven patients and residual 131I uptake in the thyroid bed in two patients, but none of these patients had signs of disease progression. Five group C patients (14%) had a positive rhTSH test result, and this was suggestive of disease progression in at least two cases.
Conclusion
The first follow-up testing is essential for prognostic classification after 131I ablation of thyroid cancer. In stage I patients, undetectable Tg and negative 131I scan 1 year after ablation define a large population of subjects who have a very low risk of recurrence and who do not require further stimulation tests. In contrast, periodic rhTSH stimulation tests appear useful in higher-risk patients.

Similar content being viewed by others
References
Schlumberger MJ. Papillary and follicular thyroid carcinoma. N Engl J Med 1998;338:297–306.
Schroeder PR, Haugen BR, Pacini F, Reiners C, Schlumberger M, Sherman SI, et al. A comparison of short-term changes in health-related quality of life in thyroid carcinoma patients undergoing diagnostic evaluation with rhTSH compared to thyroid hormone withdrawal. J Clin Endocrinol Metab 2006;91:878–84.
Mazzaferri EL, Robbins RJ, Spencer CA, Braverman LE, Pacini F, Wartosky L, et al. A consensus report of the role of serum thyroglobulin as a monitoring method for low-risk patients with papillary thyroid carcinoma. J Clin Endocrinol Metab 2003; 88:1433–41.
Schlumberger M, Berg G, Cohen O, Duntas L, Jamar F, Jarzab B, et al. Follow-up of low-risk patients with differentiated thyroid carcinoma: a European perspective. Eur J Endocrinol 2004; 150:105–12.
Kloos RT, Mazzaferri EL. A single recombinant human thyrotropin-stimulated serum thyroglobulin measurement predicts differentiated thyroid carcinoma metastases three to five years later. J Clin Endocrinol Metab 2005;90:5047–57.
David A, Blotta A, Rossi R, Zatelli MC, Bondanelli M, Roti E, et al. Clinical value of different responses of serum thyroglobulin to recombinant human thyrotropin in the follow-up of patients with differentiated thyroid carcinoma. Thyroid 2005;15:267–73.
Cooper DS, Doherty GM, Haugen BR, Kloos RT, Lee SL, Mandel SJ, et al. Management guidelines for patients with thyroid nodules and differentiated thyroid cancer. Thyroid 2006;16:109–42.
Hindie E, Melliere D, Lange F, Hallaj I, de Labriolle-Vaylet C, Jeanguillaume C, et al. Functioning pulmonary metastases of thyroid cancer: does radioiodine influence the prognosis? Eur J Nucl Med Mol Imaging 2003;30:974–81.
Preissner CM, O’Kane DJ, Singh RJ, Morris JC, Grabe SKG. Phantoms in the assay tube: heterophile antibody interferences in serum thyroglobulin assays. J Clin Endocrinol Metab 2003;88:3069–74.
Preissner CM, Nguyen HT, Pomeroy MR, et al. Evaluation of thyroglobulin reagents modified to reduce heterophile antibody interference on Beckman Coulter’s Access Immunoassay systems. Presented at the AACC 2004 meeting (presentation number: C-10 AACC).
Giovanella L, Ceriani L. High-sensitivity human thyroglobulin (hTG) immunoradiometric assay in the follow-up of patients with differentiated thyroid cancer. Clin Chem Lab Med 2002;40:480–4.
Zophel K, Wunderlich G, Smith BR. Serum thyroglobulin measurements with a high sensitivity enzyme-linked immunosorbent assay: is there a clinical benefit in patients with differentiated thyroid carcinoma? Thyroid 2003;13:861–5.
Mazzaferri EL, Kloos RT. Is diagnostic iodine-131 scanning with recombinant human TSH useful in the follow-up of differentiated thyroid cancer after thyroid ablation? J Clin Endocrinol Metab 2002;87:1490–8.
Pacini F, Molinaro E, Castagna MG, Agate L, Elisei R, Ceccarelli C, et al. Recombinant human thyrotropin-stimulated serum thyroglobulin combined with neck ultrasonography has the highest sensitivity in monitoring differentiated thyroid carcinoma. J Clin Endocrinol Metab 2003;88:3668–73.
Robbins RJ, Chon JT, Fleisher M, Larson SM, Tuttle RM. Is the serum thyroglobulin response to recombinant human thyrotropin sufficient, by itself, to monitor for residual thyroid carcinoma? J Clin Endocrinol Metab 2002;87:3242–7.
Eustatia-Rutten CF, Smit JW, Romijn JA, van der Kleij-Corssmit EP, Pereira AM, Stokkel MP, et al. Diagnostic value of serum thyroglobulin measurements in the follow-up of differentiated thyroid carcinoma, a structured meta-analysis. Clin Endocrinol (Oxf) 2004;61:61–74.
Ladenson PW, Braverman LE, Mazzaferri EL, Bruker-Davis F, Cooper DS, Garber JR, et al. Comparison of administration of recombinant human thyrotropin with withdrawal of thyroid hormone for radioactive iodine scanning in patients with thyroid carcinoma. N Engl J Med 1997;337:888–96.
Haugen BR, Pacini F, Reiners C, Schlumberger M, Ladenson PW, Shermen SI, et al. A comparison of recombinant human thyrotropin and thyroid hormone withdrawal for the detection of thyroid remnant or cancer. J Clin Endocrinol Metab 1999;84:3877–85.
Cailleux AF, Baudin E, Travagli JP, Ricard M, Schlumberger M. Is diagnostic iodine-131 scanning useful after total thyroid ablation for differentiated thyroid cancer? J Clin Endocrinol Metab 2000;85:175–8.
Pacini F, Capezzone M, Elisei R, Ceccarelli C, Taddei D, Pinchera A, et al. Diagnostic 131-iodine whole-body scan may be avoided in thyroid cancer patients who have undetectable stimulated serum Tg levels after initial treatment. J Clin Endocrinol Metab 2002;87:1499–501.
Rouxel A, Hejblum G, Bernier MO, Boëlle PY, Ménégaux F, Mansour G, et al. Prognostic factors associated with the survival of patients developing loco-regional recurrences of differentiated thyroid carcinomas. J Clin Endocrinol Metab 2004;89:5362–8.
Baudin E, Do Cao C, Cailleux AF, Leboulleuxs S, Travagli JP, Schlumberger M. Positive predictive value of serum thyroglobulin levels, measured during the first year of follow-up after thyroid hormone withdrawal, in thyroid cancer patients. J Clin Endocrinol Metab 2003;88:1107–11.
Acknowledgments
The authors are grateful to professor Philippe Bouchard, Head of the Endocrinology Department and President of the SFE “Société Française d’Endocrinologie”, and to R. Lawrence Sullivan, Jr., MD, at O’Connor Hospital, San Jose, California.
They would also like to thank Nicole Allegrini and all the laboratory staff and technicians of the Nuclear Medicine Department.
The study did not receive any financial support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zanotti-Fregonara, P., Khoury, A., Duron, F. et al. Which thyroid cancer patients need periodic stimulation tests?. Eur J Nucl Med Mol Imaging 34, 541–546 (2007). https://doi.org/10.1007/s00259-006-0279-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-006-0279-z