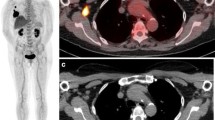
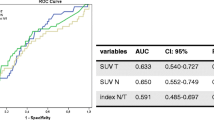
Abstract
Breast cancer continues to be one of the most common cancers in North America and Western Europe. Positron emission tomography with 2-[fluorine-18]-fluoro-2-deoxy-d-glucose (FDG PET) represents a non-invasive functional imaging modality that is based on metabolic characteristics of malignant tumours. In breast cancer, FDG PET is more accurate than conventional methods for staging of distant metastases or local recurrences and enables early assessment of treatment response in patients undergoing primary chemotherapy. Recent data indicate a rationale for the use of FDG PET in cases of asymptomatically elevated tumour marker levels in the presence of uncertain results of conventional imaging. Despite the fact that PET cannot rule out microscopic disease, it does have particular value in providing, in a single examination, a reliable assessment of the true extent of the disease. This technique is complementary to morphological imaging for primary diagnosis, staging and re-staging. It may become the method of choice for the assessment of asymptomatic patients with elevated tumour marker levels. This method, however, cannot replace invasive procedures if microscopic disease is of clinical relevance.


Similar content being viewed by others
References
Avril N, Rose CA, Schelling M, Dose J, Kuhn W, Bense S, Weber W, Ziegler S, Graeff H, Schwaiger M. Breast imaging with positron emission tomography and fluorine-18 fluorodeoxyglucose: use and limitations. J Clin Oncol 2000; 18:3495–3502.
Danforth DN Jr, Aloj L, Carrasquillo JA, Bacharach SL, Chow C, Zujewski J, Whatley M, Galen B, Merino M, Neumann RD. The role of18F-FDG-PET in the local/regional evaluation of women with breast cancer. Breast Cancer Res Treat 2002; 75:135–146.
van Dongen JA, Voogd AC, Fentiman IS, Legrand C, Sylvester RJ, Tong D, van der Schueren E, Helle PA, van Zijl K, Bartelink H. Long-term results of a randomized trial comparing breast-conserving therapy with mastectomy: European Organization for Research and Treatment of Cancer 10801 trial. J Natl Cancer Inst 2000; 19:1143–1150.
Grunfeld E, Mant D, Yudkin P, Dalton RA, Cole D, Stewart J, Fitzpatrick R, Vessey M. Routine follow-up for breast cancer in primary care: randomised trial. BMJ 1996; 13:665–669.
Hathaway PB, Mankoff DA, Maravilla KR, Austin-Seymour MM, Ellis GK, Gralow JR, Cortese AA, Hayes CE, Moe RE. Value of combined FDG PET and MRI imaging in the evaluation of suspected recurrent local-regional breast cancer: preliminary experience. Radiology 1999; 210:807–814.
Schirrmeister H, Kuhn T, Guhlmann A, Santjohanser C, Horster T, Nussle K, Koretz K, Glatting G, Rieber A, Kreienberg R, Buck AC, Reske SN. Fluorine-18 2-deoxy-2-fluoro-d-glucose PET in the preoperative staging of breast cancer: comparison with the standard staging procedures. Eur J Nucl Med 2001; 28:351–358.
Dose J, Bleckmann C, Bachmann S, Bohuslavizki KH, Berger J, Jenicke L, Habermann CR, Janicke F. Comparison of fluorodeoxyglucose positron emission tomography and conventional diagnostic procedures for the detection of distant metastases in breast cancer patients. Nucl Med Commun 2002; 23:857–864.
Ohta M, Tokuda Y, Suzuki Y, Kubota M, Makuuchi H, Tajima T, Nasu S, Suzuki Y, Yasuda S, Shohtsu A. Whole body PET for the evaluation of bony metastases in patients with breast cancer: comparison with99Tcm-MDP bone scintigraphy. Nucl Med Commun 2001; 22:875–879.
Yang SN, Liang JA, Lin FJ, Kao CH, Lin CC, Lee CC. Comparing whole body18F-2-deoxyglucose positron emission tomography and technetium-99m methylene diphosphonate bone scan to detect bone metastases in patients with breast cancer. J Cancer Res Clin Oncol 2002; 128:325–328.
Hayes DF, Bast RC, Desch CE, Fritsche H Jr, Kemeny NE, Jessup JM, Locker GY, Macdonald JS, Mennel RG, Norton L, Ravdin P, Taube S, Winn RJ. Tumor marker utility grading system: a framework to evaluate clinical utility of tumor markers. J Natl Cancer Inst 1996; 88:1456–1466.
Voogd AC, Nielsen M, Peterse JL, Blichert-Toft M, Bartelink H, Overgaard M, van Tienhoven G, Andersen KW, Sylvester RJ, van Dongen JA, Danish Breast Cancer Cooperative Group. Breast Cancer Cooperative Group of the European Organization for Research and Treatment of Cancer. Differences in risk factors for local and distant recurrence after breast-conserving therapy or mastectomy for stage I and II breast cancer: pooled results of two large European randomized trials. J Clin Oncol 2001; 19:1688–1697.
Lonneux M, Borbath II, Berliere M, Kirkove C, Pauwels S. The place of whole-body PET FDG for the diagnosis of recurrence of breast cancer. Clin Positron Imaging 2000; 3:45–49.
Schwaibold F, Fowble BL, Solin LJ, Schultz DJ, Goodman RL. The results of radiation therapy for isolated local regional recurrence after mastectomy. Int J Radiat Oncol Biol Phys 1991; 21:299–310.
Leborgne F, Leborgne JH, Ortega B, Doldan R, Zubizarreta E. Breast conservation treatment of early stage breast cancer: patterns of failure. Int J Radiat Oncol Biol Phys 1995; 31:765–775.
Katz A, Strom EA, Buchholz TA, Theriault R, Singletary SE, McNeese MD. The influence of pathologic tumor characteristics on locoregional recurrence rates following mastectomy. Int J Radiat Oncol Biol Phys 2001; 50:735–742.
Kolasinska AD, Buscombe JR, Cwikla JB, Holloway B, Parbhoo SP, Davidson T, Hilson AJ. The role of scintimammography and mammography in recurrent breast cancer. Evaluation of their accuracy using ROC curves. Nucl Med Rev Cent East Eur 2001; 4:77–82.
Gilles R, Guinebretiere JM, Lucidarme O, Cluzel P, Janaud G, Finet JF, Tardivon A, Masselot J, Vanel D. Nonpalpable breast tumors: diagnosis with contrast-enhanced subtraction dynamic MR imaging. Radiology 1994; 191:625–631.
Belli P, Costantini M, Romani M, Marano P, Pastore G. Magnetic resonance imaging in breast cancer recurrence. Breast Cancer Res Treat 2002; 73:223–235.
Kamel EM, Wyss MT, Fehr MK, von Schulthess GK, Goerres GW. [18F]-fluorodeoxyglucose positron emission tomography in patients with suspected recurrence of breast cancer. J Cancer Res Clin Oncol 2003; 129:147–153.
Moon DH, Maddahi J, Silvermann HS., Glaspy JA, Phelps ME, Hoh CK. Accuracy of whole-body fluorine-18-FDG PET for the detection of recurrent or metastatic breast carcinoma. J Nucl Med 1997; 39:431–435.
Hubner KF, Smith GT, Thie JA, Bell JL, Nelson HS, Hanna WT. The Potential of F-18-FDG PET in breast cancer. Detection of primary lesions, axillary lymph node metastases, or distant metastases. Clin Positron Imaging 2000; 3:197–205.
Kim TS, Moon WK, Lee DS, Chung JK, Lee MC, Youn YK, Oh SK, Choe KJ, Noh DY. Fluorodeoxyglucose positron emission tomography for detection of recurrent or metastatic breast cancer. World J Surg 2001; 25:829–834.
Eubank WB, Mankoff DA, Takasugi J, Vesselle H, Eary JF, Shanley TJ, Gralow JR, Charlop A, Ellis GK, Lindsley KL, Austin-Seymour MM, Funkhouser CP, Livingston RB.18Fluorodeoxyglucose positron emission tomography to detect mediastinal or internal mammary metastases in breast cancer. J Clin Oncol 2001; 19:3516–3523.
Steinert HC, Hauser M, Allemann F. Non-small cell lung cancer: nodal staging with FDG-PET versus CT with correlative lymph node mapping and sampling. Radiology 1997; 202:441–446.
Cook GJ, Houston S, Rubens R, Maisey MN, Fogelman J. Detection of bone metastases in Breast cancer by FDG PET. Differing metabolic activity in osteoblastic and osteolytic lesions. J Clin Oncol 1998; 16:3375–3379.
Ogunbiyi OA, Flanagan FL, Dehdashti F, Siegel BA, Trask DD, Birnbaum EH, Fleshman JW, Read TE, Philpott GW, Kodner IJ. Detection of recurrent and metastatic colorectal cancer: comparison of positron emission tomography and computed tomography. Ann Surg Oncol 1997; 4:613–620.
The GIVIO Investigators. Impact of follow-up testing on survival and health related quality of life in breast cancer patients. A multicenter randomized controlled trial. JAMA 1994; 271:1587–1592.
Ugrinska A, Bombardieri E, Stockel MP, Crippa F, Pauwels EK. Circulation tumour markers and nuclear medicine imaging modalities: breast, prostate and ovarian cancer. Q J Nucl Med 2002; 46:88–104.
Bast RC Jr, Ravdin P, Hayes DF, Bates S, Fritsche H Jr, Jessup JM, Kemeny N, Locker GY, Mennel RG, Somerfield MR, American Society of Clinical Oncology Tumor Markers Expert Panel. 2000 update of recommendations for the use of tumor markers in breast and colorectal cancer: clinical practice guidelines of the American Society of Clinical Oncology. J Clin Oncol 2001; 19:1865–1878.
Chan DW, Beveridge RA, Muss H, Fritsche HA, Hortobagyi G, Theriault R, Kiang D, Kennedy BJ, Evelegh M. Use of Truquant BR radioimmunoassay for early detection of breast cancer recurrence in patients with stage II and stage III disease. J Clin Oncol 1997; 15:2322–2328.
Kokko R, Holli K, Hakama M. Ca 15-3 in the follow-up of localised breast cancer: a prospective study. Eur J Cancer 2002; 38:1189–1193.
Gion M, Boracchi P, Dittadi R, Biganzoli E, Peloso L, Mione R, Gatti C, Paccagnella A, Marubini E. Prognostic role of serum CA15.3 in 362 node-negative breast cancers. An old player for a new game. Eur J Cancer 2002; 38:1181–1188.
Gion M, Barioli P, Mione R, Cappelli G, Vignati G, Fortunato A, Saracchini S, Biasioli R, Gulisano M. Tumor markers in breast cancer follow-up: a potentially useful parameter still awaiting definitive assessment. Forza Operativa Nazionale sul Carcinoma Mammario (FONCaM). Ann Oncol 1995; 6 Suppl 2:31–35.
Ravaioli A, Pasini G, Polselli A, Papi M, Tassinari D, Arcangeli V, Milandri C, Amadori D, Bravi M, Rossi D, Fattori PP, Pasquini E, Panzini I. Staging of breast cancer: new recommended standard procedure. Breast Cancer Res Treat 2002; 72:53–60.
Molina R, Jo J, Filella X, Zanon G, Farrus B, Munoz M, Latre ML, Pahisa J, Velasco M, Fernandez P, Estape J, Ballesta AM. C-erbB-2, CEA and CA 15.3 serum levels in the early diagnosis of recurrence of breast cancer patients. Anticancer Res 1999; 19:2551–2555.
De La Lande B, Hacene K, Floiras JL, Alatrakchi N, Pichon MF. Prognostic value of CA 15.3 kinetics for metastatic breast cancer. Int J Biol Markers 2002; 17:231–238.
Soletormos G, Nielsen D, Schioler V, Mouridsen H, Dombernowsky P. Monitoring different stages of breast cancer using tumour markers CA 15-3, CEA and TPA. Eur J Cancer 2004; 40:481–486.
Liu CS, Shen YY, Lin CC, Yen RF, Kao CH. Clinical impact of [18F]FDG-PET in patients with suspected recurrent breast cancer based on asymptomatically elevated tumor marker serum levels: a preliminary report. Jpn J Clin Oncol 2002; 32:244–247.
Pecking AP, Mechelany-Corone C, Bertrand-Kermorgant F, Alberini JL, Floiras JL, Goupil A, Pichon MF. Detection of occult disease in breast cancer using fluorodeoxyglucose camera-based positron emission tomography. Clin Breast Cancer 2001; 2:229–234.
Suarez M, Perez-Castejon MJ, Jimenez A, Domper M, Ruiz G, Montz R, Carreras JL. Early diagnosis of recurrent breast cancer with FDG-PET in patients with progressive elevation of serum tumor markers. Q J Nucl Med 2002; 46:113–121.
Chang WC, Hung YC, Kao CH, Yen RF, Shen YY, Lin CC. Usefulness of whole body positron emission tomography (PET) with18F-fluoro-2-deoxyglucose (FDG) to detect recurrent ovarian cancer based on asymptomatically elevated serum levels of tumor marker. Neoplasma 2002; 49:329–333.
Vranjesevic D, Filmont JE, Meta J, Silverman DH, Phelps ME, Rao J, Valk PE, Czernin J. Whole-body18F-FDG PET and conventional imaging for predicting outcome in previously treated breast cancer patients. J Nucl Med 2002; 43:325–329.
Siggelkow W, Zimny M, Faridi A, Petzold K, Buell U, Rath W. The value of positron emission tomography in the follow-up for breast cancer. Anticancer Res 2003; 23:1859–1867.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Siggelkow, W., Rath, W., Buell, U. et al. FDG PET and tumour markers in the diagnosis of recurrent and metastatic breast cancer. Eur J Nucl Med Mol Imaging 31 (Suppl 1), S118–S124 (2004). https://doi.org/10.1007/s00259-004-1534-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-004-1534-9