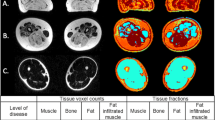
Abstract
Objective
To assess whether changes in MRI-based measures of thigh muscle quality associated with statin use in participants with and without/at-risk of knee osteoarthritis.
Methods
This retrospective cohort study used data from the Osteoarthritis Initiative study. Statin users and non-users were matched for relevant covariates using 1:1 propensity-score matching. Participants were further stratified according to baseline radiographic knee osteoarthritis status. We used a validated deep-learning method for thigh muscle MRI segmentation and calculation of muscle quality biomarkers at baseline, 2nd, and 4th visits. Mean difference and 95% confidence intervals (CI) in longitudinal 4-year measurements of muscle quality biomarkers, including cross-sectional area, intramuscular adipose tissue, contractile percent, and knee extensors and flexors maximum and specific contractile force (force/muscle area) were the outcomes of interest.
Results
After matching, 3772 thighs of 1910 participants were included (1886 thighs of statin-users: 1886 of non-users; age: 62 ± 9 years (average ± standard deviation), range: 45–79; female/male: 1). During 4 years, statin use was associated with a slight decrease in muscle quality, indicated by decreased knee extension maximum (mean-difference, 95% CI: − 1.85 N/year, − 3.23 to − 0.47) and specific contractile force (− 0.04 N/cm2/year, − 0.07 to − 0.01), decreased thigh muscle contractile percent (− 0.03%/year, − 0.06 to − 0.01), and increased thigh intramuscular adipose tissue (3.06 mm2/year, 0.53 to 5.59). Stratified analyses showed decreased muscle quality only in participants without/at-risk of knee osteoarthritis but not those with established knee osteoarthritis.
Conclusions
Statin use is associated with a slight decrease in MRI-based measures of thigh muscle quality over 4 years. However, considering statins’ substantial cardiovascular benefits, these slight muscle changes may be relatively less important in overall patient care.


Similar content being viewed by others
Data availability
The de-identified clinical and demographic information of subjects is publicly available at the osteoarthritis initiative project data repository at https://oai.nih.gov.
References
Fuentes AV, Pineda MD, Venkata KCN. Comprehension of top 200 prescribed drugs in the US as a resource for pharmacy teaching, training and practice. Pharmacy (Basel). 2018;6(2):43.
Collins R, Reith C, Emberson J, Armitage J, Baigent C, Blackwell L, et al. Interpretation of the evidence for the efficacy and safety of statin therapy. Lancet. 2016;388(10059):2532–61.
Haj-Mirzaian A, Mohajer B, Guermazi A, Conaghan PG, Lima JAC, Blaha MJ, et al. Statin use and knee osteoarthritis outcome measures according to the presence of Heberden nodes: results from the Osteoarthritis Initiative. Radiology. 2019;293(2):190557.
Mohajer B, Guermazi A, Conaghan PG, Berenbaum F, Roemer FW, Haj-Mirzaian A, et al. Statin use and MRI subchondral bone marrow lesion worsening in generalized osteoarthritis: longitudinal analysis from Osteoarthritis Initiative data. Eur Radiol. 2022;32(6):3944–53.
Valdes AM, Zhang W, Muir K, Maciewicz RA, Doherty S, Doherty M. Use of statins is associated with a lower prevalence of generalised osteoarthritis. Ann Rheum Dis. 2014;73(5):943–5.
Stroes ES, Thompson PD, Corsini A, Vladutiu GD, Raal FJ, Ray KK, et al. Statin-associated muscle symptoms: impact on statin therapy—European Atherosclerosis Society Consensus Panel Statement on Assessment, Aetiology and Management. Eur Heart J. 2015;36(17):1012–22.
Cohen JD, Brinton EA, Ito MK, Jacobson TA. Understanding statin use in America and gaps in patient education (USAGE): an internet-based survey of 10,138 current and former statin users. J Clin Lipidol. 2012;6(3):208–15.
Law M, Rudnicka AR. Statin safety: a systematic review. Am J Cardiol. 2006;97(8a):52c–60c.
Parker BA, Capizzi JA, Grimaldi AS, Clarkson PM, Cole SM, Keadle J, et al. Effect of statins on skeletal muscle function. Circulation. 2013;127(1):96–103.
Herrett E, Williamson E, Brack K, Beaumont D, Perkins A, Thayne A, et al. Statin treatment and muscle symptoms: series of randomised, placebo controlled n-of-1 trials. BMJ. 2021;372:n135.
Raynauld J-P, Pelletier J-P, Roubille C, Dorais M, Abram F, Li W, et al. Magnetic resonance imaging–assessed vastus medialis muscle fat content and risk for knee osteoarthritis progression: relevance from a clinical trial. Arthritis Care Res. 2015;67(10):1406–15.
Kumar D, Link TM, Jafarzadeh SR, LaValley MP, Majumdar S, Souza RB. Association of quadriceps adiposity with an increase in knee cartilage, meniscus, or bone marrow lesions over three years. Arthritis Care Res. 2021;73(8):1134–9.
Morse CI, Thom JM, Reeves ND, Birch KM, Narici MV. In vivo physiological cross-sectional area and specific force are reduced in the gastrocnemius of elderly men. J Appl Physiol (1985). 2005;99(3):1050–5.
Culvenor AG, Felson DT, Niu J, Wirth W, Sattler M, Dannhauer T, et al. Thigh muscle specific-strength and the risk of incident knee osteoarthritis: the influence of sex and greater body mass index. Arthritis Care Res. 2017;69(8):1266–70.
Culvenor AG, Hamler FC, Kemnitz J, Wirth W, Eckstein F. Brief report: loss of muscle strength prior to knee replacement: a question of anatomic cross-sectional area or specific strength? Arthritis Rheumatol. 2018;70(2):222–9.
Morville T, Dohlmann TL, Kuhlman AB, Sahl RE, Kriegbaum M, Larsen S, et al. Aerobic exercise performance and muscle strength in statin users-the LIFESTAT study. Med Sci Sports Exerc. 2019;51(7):1429–37.
Lawrence JS. The epidemiology of chronic rheumatism. Ann Rheum Dis. 1964;23(1):81.
Rantanen T, Era P, Heikkinen E. Maximal isometric strength and mobility among 75-year-old men and women. Age Ageing. 1994;23(2):132–7.
Peterfy CG, Schneider E, Nevitt M. The osteoarthritis initiative: report on the design rationale for the magnetic resonance imaging protocol for the knee. Osteoarthritis and cartilage. 2008;16(12):1433–1441.
Mohajer B, Dolatshahi M, Moradi K, Najafzadeh N, Eng J, Zikria B, et al. Role of thigh muscle changes in knee osteoarthritis outcomes: Osteoarthritis Initiative data. Radiology. 2022;305(1):169–78.
Ahmad E, McPhee J, Degens H, Yap MH. Automatic segmentation of MRI human thigh muscles: combination of reliable and fast framework methods for quadriceps, femur and marrow segmentation. In Proceedings of the 2018 8th International Conference on Biomedical Engineering and Technology (ICBET '18). Association for Computing Machinery, New York, NY; 2018. p. 31–38
Ogawa M, Yoshiko A, Tanaka N, Koike T, Oshida Y, Akima H. Comparing intramuscular adipose tissue on T1-weighted and two-point Dixon images. PLoS One. 2020;15(4):e0231156–e0231156.
Moser M, AdlAmini D, Echeverri C, Oezel L, Haffer H, Muellner M, et al. Changes in psoas and posterior paraspinal muscle morphology after standalone lateral lumbar interbody fusion: a quantitative MRI-based analysis. Eur Spine J. 2023;32(5):1704–13.
Fortin M, Omidyeganeh M, Battié MC, Ahmad O, Rivaz H. Evaluation of an automated thresholding algorithm for the quantification of paraspinal muscle composition from MRI images. Biomed Eng Online. 2017;16(1):61–61.
Ogier AC, Hostin MA, Bellemare ME, Bendahan D. Overview of MR image segmentation strategies in neuromuscular disorders. Frontiers in Neurology. 2021; 12:625308.
Lee D, Hong K-T, Lee W, Khil EK, Lee GY, Choi J-A, et al. Threshold-based quantification of fatty degeneration in the supraspinatus muscle on MRI as an alternative method to Goutallier classification and single-voxel MR spectroscopy. BMC Musculoskelet Disord. 2020;21(1):362.
Ogawa M, Lester R, Akima H, Gorgey AS. Quantification of intermuscular and intramuscular adipose tissue using magnetic resonance imaging after neurodegenerative disorders. Neural Regen Res. 2017;12(12):2100–5.
Bellamy N, Buchanan WW, Goldsmith CH, Campbell J, Stitt LW. Validation study of WOMAC: a health status instrument for measuring clinically important patient relevant outcomes to antirheumatic drug therapy in patients with osteoarthritis of the hip or knee. J Rheumatol. 1988;15(12):1833–40.
Baldwin CE, Paratz JD, Bersten AD. Muscle strength assessment in critically ill patients with handheld dynamometry: an investigation of reliability, minimal detectable change, and time to peak force generation. J Crit Care. 2013;28(1):77–86.
Phillips PS, Haas RH, Bannykh S, Hathaway S, Gray NL, Kimura BJ, et al. Statin-associated myopathy with normal creatine kinase levels. Ann Intern Med. 2002;137(7):581–5.
Biltz NK, Collins KH, Shen KC, Schwartz K, Harris CA, Meyer GA. Infiltration of intramuscular adipose tissue impairs skeletal muscle contraction. J Physiol. 2020;598(13):2669–83.
Loenneke JP, Loprinzi PD. Statin use may reduce lower extremity peak force via reduced engagement in muscle-strengthening activities. Clin Physiol Funct Imaging. 2018;38(1):151–4.
Scott D, Blizzard L, Fell J, Jones G. Statin therapy, muscle function and falls risk in community-dwelling older adults. QJM. 2009;102(9):625–33.
Robles PG, Sussman MS, Naraghi A, Brooks D, Goldstein RS, White LM, et al. Intramuscular fat infiltration contributes to impaired muscle function in COPD. Med Sci Sports Exerc. 2015;47(7):1334–41.
Lee KH, Gao Y, Lau V. Statin-associated anti-3-hydroxy-3-methyl-glutaryl-coenzyme A reductase (HMGCR) myopathy: imaging findings on thigh-muscle magnetic resonance imaging (MRI) in six patients. Muscle Nerve. 2021;64(4):500–4.
Wagner M, Mühldorfer-Fodor M, Prommersberger KJ, Schmitt R. Statin-induced focal myositis of the upper extremity. A report of two cases. Eur J Radiol. 2011;77(2):258–60.
Amit P, Kuiper JH, James S, Snow M. Does statin-treated hyperlipidemia affect rotator cuff healing or muscle fatty infiltration after rotator cuff repair? J Shoulder Elbow Surg. 2021;30(11):2465–74.
Goutallier D, Postel JM, Bernageau J, Lavau L, Voisin MC. Fatty muscle degeneration in cuff ruptures. Pre- and postoperative evaluation by CT scan. Clin Orthop Relat Res. 1994;304:78–83.
Chowdhury R, Khan H, Heydon E, Shroufi A, Fahimi S, Moore C, et al. Adherence to cardiovascular therapy: a meta-analysis of prevalence and clinical consequences. Eur Heart J. 2013;34(38):2940–8.
Veronese N, Koyanagi A, Stubbs B, Cooper C, Guglielmi G, Rizzoli R, et al. Statin use and knee osteoarthritis outcomes: a longitudinal cohort study. Arthritis Care Res (Hoboken). 2019;71(8):1052–8.
Haj-Mirzaian A, Mohajer B, Guermazi A, Conaghan PG, Lima JAC, Blaha MJ, et al. Statin use and knee osteoarthritis outcome measures according to the presence of Heberden nodes: results from the Osteoarthritis Initiative. Radiology. 2019;293(2):396–404.
Hafferty JD, Campbell AI, Navrady LB, Adams MJ, MacIntyre D, Lawrie SM, et al. Self-reported medication use validated through record linkage to national prescribing data. J Clin Epidemiol. 2018;94:132–42.
Mueller AM, Liakoni E, Schneider C, Burkard T, Jick SS, Krähenbühl S, et al. The risk of muscular events among new users of hydrophilic and lipophilic statins: an observational cohort study. J Gen Intern Med. 2021;36(9):2639–47.
Chan DC, Gan SK, Wong AT, Barrett PH, Watts GF. Association between skeletal muscle fat content and very-low-density lipoprotein-apolipoprotein B-100 transport in obesity: effect of weight loss. Diabetes Obes Metab. 2014;16(10):994–1000.
Hollingsworth KG, Garrood P, Eagle M, Bushby K, Straub V. Magnetic resonance imaging in Duchenne muscular dystrophy: longitudinal assessment of natural history over 18 months. Muscle Nerve. 2013;48(4):586–8.
Tustison NJ, Avants BB, Cook PA, Zheng Y, Egan A, Yushkevich PA, et al. N4ITK: improved N3 bias correction. IEEE Trans Med Imaging. 2010;29(6):1310–20.
Manini TM, Clark BC, Nalls MA, Goodpaster BH, Ploutz-Snyder LL, Harris TB. Reduced physical activity increases intermuscular adipose tissue in healthy young adults. Am J Clin Nutr. 2007;85(2):377–84.
Hogrel J-Y, Barnouin Y, Azzabou N, Butler-Browne G, Voit T, Moraux A, et al. NMR imaging estimates of muscle volume and intramuscular fat infiltration in the thigh: variations with muscle, gender, and age. Age. 2015;37(3):60.
Acknowledgements
The Osteoarthritis Initiative, a collaborative project between public and private sectors, includes five contracts N01-AR-2-2258, N01-AR-2-2259, N01-AR-2-2260, N01-AR-2-2261, and N01-AR-2-2262. This project is conducted by the Osteoarthritis Initiative project investigators and is financially supported by the National Institutes of Health (NIH). Merck Research Laboratories, Novartis Pharmaceuticals Corporation, GlaxoSmithKline, and Pfizer, Inc. were the private funding partners.
In preparing this manuscript, osteoarthritis initiative project publicly available datasets were used. The results of this work do not necessarily reflect the opinions of the osteoarthritis initiative project investigators, the NIH, or the private funding partners.
Funding
This research was supported by the NIH National Institute of Aging (NIA) under Award Number P01AG066603 and NIH National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS) under Award Number R01AR079620-01.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
The medical ethics review boards of the University of California, San Francisco (Approval Number: 10–00532) and the four clinical centers of Osteoarthritis Initiative project recognized the project as Health Insurance Portability and Accountability Act (HIPAA)-compliant. This project was in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments, and all individuals gave their informed consent prior to their inclusion in the study. The authors of this manuscript certify that they comply with the ethical guidelines for authorship and publishing in the Journal of Cachexia, Sarcopenia, and Muscle.
Patient consent
Subjects have given informed consent before participating in the Osteoarthritis Initiative (OAI) project.
Conflict of interest
None of the authors has any conflicting personal or financial relationships with the organization that sponsored the research (NIH) that could have influenced the results of this study. FWR is chief marketing officer and shareholder of Boston Imaging Core Lab (BICL), LLC, and consultant to Calibr — California Institute of Biomedical Research and Grünenthal GmbH. AG is a shareholder of BICL and consultant to Pfizer, TissueGene, MerckSerono, Novartis, Regeneron, and AstraZeneca. SD reported that he received funding from Toshiba Medical Systems (for consultation) and grants from GERRAF and Carestream Health (for a clinical trial study). PGC is supported in part through the UK National Institute for Health Research (NIHR) Leeds Biomedical Research Centre. The views expressed are those of the authors and not necessarily those of the National Health Service, the NIHR, or the Department of Health. Other authors declare that they did not have any competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mohajer, B., Moradi, K., Guermazi, A. et al. Statin use and longitudinal changes in quantitative MRI-based biomarkers of thigh muscle quality: data from Osteoarthritis Initiative. Skeletal Radiol 53, 683–695 (2024). https://doi.org/10.1007/s00256-023-04473-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-023-04473-7