Abstract
Purpose
Many children who undergo MR of the knee to evaluate traumatic injury may not undergo a separate dedicated evaluation of their skeletal maturity, and we wished to investigate how accurately skeletal maturity could be automatically inferred from knee MRI using deep learning to offer this additional information to clinicians.
Materials and methods
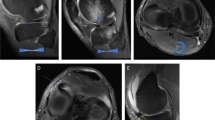
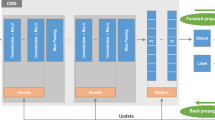
Retrospective data from 894 studies from 783 patients were obtained (mean age 13.1 years, 47% female). Coronal and sagittal sequences that were T1/PD-weighted were included and resized to 224 × 224 pixels. Data were divided into train (n = 673), tune (n = 48), and test (n = 173) sets, and children were separated across sets. The chronologic age was predicted using deep learning approaches based on a long short-term memory (LSTM) model, which took as input DenseNet-121-extracted features from all T1/PD coronal and sagittal slices. Each test case was manually assigned a bone age by two radiology residents using a reference atlas provided by Pennock and Bomar. The patient’s age served as ground truth.
Results
The error of the model’s predictions for chronological age was not significantly different from that of radiology residents (model M.S.E. 1.30 vs. resident 0.99, paired t-test = 1.47, p = 0.14). Pearson correlation between model and resident prediction of chronologic age was 0.96 (p < 0.001).
Conclusion
A deep learning-based approach demonstrated ability to infer skeletal maturity from knee MR sequences that was not significantly different from resident performance and did so in less than 2% of the time required by a human expert. This may offer a method for automatically evaluating lower extremity skeletal maturity automatically as part of every MR examination.




Similar content being viewed by others
References
Mutasa S, Chang PD, Ruzal-Shapiro C, Ayyala R. MABAL: a novel deep-learning architecture for machine-assisted bone age labeling. J Digit Imaging. 2018;31:513–9.
Soudack M, Ben-Shlush A, Jacobson J, Raviv-Zilka L, Eshed I, Hamiel O. Bone age in the 21st century: is Greulich and Pyle’s atlas accurate for Israeli children? [Internet]. Pediatric Radiology. 2012. p. 343–8. Available from: https://doi.org/10.1007/s00247-011-2302-1
Büken B, Şafak AA, Yazıcı B, Büken E, Mayda AS. Is the assessment of bone age by the Greulich-Pyle method reliable at forensic age estimation for Turkish children? Forensic Sci Int. 2007;173:146–53.
Kahleyss S, Hoepffner W, Keller E. Willgerodt H [The determination of bone age by the Greulich-Pyle and Tanner-Whitehouse methods as a basis for the growth prognosis of tall-stature girls]. Padiatr Grenzgeb. 1990;29:137–40.
Bull RK, Edwards PD, Kemp PM, Fry S, Hughes IA. Bone age assessment: a large scale comparison of the Greulich and Pyle, and Tanner and Whitehouse (TW2) methods. Arch Dis Child. 1999;81:172–3.
Larson DB, Chen MC, Lungren MP, Halabi SS, Stence NV, Langlotz CP. Performance of a deep-learning neural network model in assessing skeletal maturity on pediatric hand radiographs. Radiology. 2018;287:313–22.
Halabi SS, Prevedello LM, Kalpathy-Cramer J, Mamonov AB, Bilbily A, Cicero M, et al. The RSNA pediatric bone age machine learning challenge. Radiology. 2019;290:498–503.
Nguyen JC, De Smet AA, Graf BK, Rosas HG. MR imaging-based diagnosis and classification of meniscal tears. Radiographics. 2014;34:981–99.
Pennock AT, Bomar JD, Manning JD. The creation and validation of a knee bone age atlas utilizing MRI. J Bone Joint Surg Am. 2018;100:e20.
Mauer MA der, Well EJ, Herrmann J, Groth M, Morlock MM, Maas R, et al. Automated age estimation of young individuals based on 3D knee MRI using deep learning [Internet]. International Journal of Legal Medicine. 2021. p. 649–63. Available from: https://doi.org/10.1007/s00414-020-02465-z
Sauer PJJ, On behalf of the Advocacy and Ethics Group of the European Academy of Paediatrics, Nicholson A, Neubauer D. Age determination in asylum seekers: physicians should not be implicated [Internet]. European Journal of Pediatrics. 2016. p. 299–303. Available from: https://doi.org/10.1007/s00431-015-2628-z
Dallora AL, Kvist O, Berglund JS, Ruiz SD, Boldt M, Flodmark C-E, et al. Chronological age assessment in young individuals using bone age assessment staging and nonradiological aspects: machine learning multifactorial approach. JMIR Med Inform. 2020;8:e18846.
Politzer CS, Bomar JD, Pehlivan HC, Gurusamy P, Edmonds EW, Pennock AT. Creation and validation of a shorthand magnetic resonance imaging bone age assessment tool of the knee as an alternative skeletal maturity assessment. Am J Sports Med. 2021;49:2955–9.
Dedouit F, Auriol J, Rousseau H, Rougé D, Crubézy E, Telmon N. Age assessment by magnetic resonance imaging of the knee: a preliminary study. Forensic Sci Int. 2012;217(232):e1-7.
Margalit A, Cottrill E, Nhan D, Yu L, Tang X, Fritz J, et al. The spatial order of physeal maturation in the normal human knee using magnetic resonance imaging. J Pediatr Orthop. 2019;39:e318–22.
Greulich WW, Pyle SI. Radiographic atlas of skeletal development of the hand and wrist. Stanford: Stanford University Press; 1959.
Rajpurkar P, Irvin J, Zhu K, Yang B, Mehta H, Duan T, et al. CheXNet: Radiologist-level pneumonia detection on chest X-rays with deep learning [Internet]. arXiv [cs.CV]. 2017. Available from: http://arxiv.org/abs/1711.05225
Paszke A, Gross S, Chintala S, Chanan G, Yang E, DeVito Z, et al. Automatic differentiation in PyTorch [Internet]. 2017 [cited 2018 Apr 9]. Available from: https://openreview.net/pdf?id=BJJsrmfCZ
Hochreiter S, Schmidhuber J. Long short-term memory. Neural Comput. 1997;9:1735–80.
Huang G, Liu Z, van der Maaten L, Weinberger KQ. Densely connected convolutional networks. arXiv [csCV] [Internet]. 2018; Available from: https://arxiv.org/pdf/1608.06993.pdf
Bahdanau D, Cho K, Bengio Y. Neural machine translation by jointly learning to align and translate [Internet]. arXiv [cs.CL]. 2014. Available from: http://arxiv.org/abs/1409.0473
Schaffter T, Buist DSM, Lee CI, Nikulin Y, Ribli D, Guan Y, et al. Evaluation of combined artificial intelligence and radiologist assessment to interpret screening mammograms. JAMA Netw Open. 2020;3:e200265.
Badgeley MA, Zech JR, Oakden-Rayner L, Glicksberg BS, Liu M, Gale W, et al. Deep learning predicts hip fracture using confounding patient and healthcare variables. NPJ Digit Med. 2019;2:31.
Mansourvar M, Ismail MA, Raj RG, Kareem SA, Aik S, Gunalan R, et al. The applicability of Greulich and Pyle atlas to assess skeletal age for four ethnic groups. J Forensic Leg Med. 2014;22:26–9.
Loder RT, Estle DT, Morrison K, Eggleston D, Fish DN, Greenfield ML, et al. Applicability of the Greulich and Pyle skeletal age standards to black and white children of today. Am J Dis Child. 1993;147:1329–33.
Eng DK, Khandwala NB, Long J, Fefferman NR, Lala SV, Strubel NA, et al. Artificial intelligence algorithm improves radiologist performance in skeletal age assessment: a prospective multicenter randomized controlled trial. Radiology. 2021;204021.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zech, J.R., Carotenuto, G. & Jaramillo, D. Inferring pediatric knee skeletal maturity from MRI using deep learning. Skeletal Radiol 51, 1671–1677 (2022). https://doi.org/10.1007/s00256-022-04010-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-022-04010-y