Abstract
Objective
To study non-diagnostic CT-guided musculoskeletal biopsies and take steps to minimize them. Specifically we asked: (1) What malignant diagnoses have a higher non-diagnostic rate? (2) What factors of a non-diagnostic biopsy may warrant more aggressive pursuit? (3) Do intra-procedural frozen pathology (FP) or point-of-care (POC) cytology reduce the non-diagnostic biopsy rate?
Materials and methods
This study was IRB-approved and HIPAA-compliant. We retrospectively reviewed 963 consecutive CT-guided musculoskeletal biopsies. We categorized pathology results as malignant, benign, or non-diagnostic and recorded use of FP or POC cytology. Initial biopsy indication, final diagnosis, method of obtaining the final diagnosis of non-diagnostic biopsies, age of the patient, and years of biopsy attending experience were recorded. Groups were compared using Pearson’s χ2 test or Fisher’s exact test.
Results
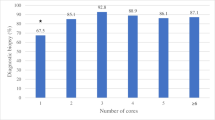
In all, 140 of 963 (15 %) biopsies were non-diagnostic. Lymphoma resulted in more non-diagnostic biopsies (P < 0.0001). While 67% of non-diagnostic biopsies yielded benign diagnoses, 33% yielded malignant diagnoses. Patients whose percutaneous biopsy was indicated due to the clinical context without malignancy history almost always generated benign results (96 %). Whereas 56% of biopsies whose indication was an imaging finding of a treatable lesion were malignant, 20% of biopsies whose indication was a history of malignancy were malignant. There was no statistically significant difference in the nondiagnostic biopsy rates of pediatric versus adult patients (P = 0.8) and of biopsy attendings with fewer versus more years of experience (P = 0.5). The non-diagnostic rates of biopsies with FP (8 %), POC cytology (25 %), or neither (24 %) were significantly different (P < 0.0001).
Conclusion
Lymphoma is the malignant diagnosis most likely to result in a non-diagnostic biopsy. If the clinical and radiologic suspicion for malignancy is high, repeat biopsy is warranted. If the clinical context suggests a benign lesion, a non-diagnostic biopsy may be considered reassuring. Frozen pathology may decrease the non-diagnostic biopsy rate.
Similar content being viewed by others
References
Dupuy DE, Rosenberg AE, Punyaratabandhu T, Tan MH, Mankin HJ. Accuracy of CT-guided needle biopsy of musculoskeletal neoplasms. AJR Am J Roentgenol. 1998;171:759–62.
Altuntas AO, Slavin J, Smith PJ, Schlict SM, Powell GJ, Ngan S, et al. Accuracy of computed tomography guided core needle biopsy of musculoskeletal tumours. ANZ J Surg. 2005;75:187–91.
Fraser-Hill MA, Renfrew DL. Percutaneous needle biopsy of musculoskeletal lesions, 1: effective accuracy and diagnostic utility. AJR Am J Roentgenol. 1992;158:809–12.
Fraser-Hill MA, Renfrew DL, Hilsenrath PE. Percutaneous needle biopsy of musculoskeletal lesions, 2: cost-effectiveness. AJR Am J Roentgenol. 1992;158:813–8.
Pohlig F, Kirchhoff C, Lenze U, Schauwecker J, Burgkart R, Rechl H, et al. Percutaneous core needle biopsy versus open biopsy in diagnostics of bone and soft tissue sarcoma: a retrospective study. Eur J Med Res. 2012;17:29.
Skrzynski MC, Biermann JS, Montag A, Simon MA. Diagnostic accuracy and charge-savings of outpatient core needle biopsy compared with open biopsy of musculoskeletal tumors. J Bone Joint Surg Am. 1996;78:644–9.
Springfield DS, Rosenberg A. Biopsy: complicated and risky. J Bone Joint Surg Am. 1996;78:639–43.
Yao L, Nelson SD, Seeger LL, Eckardt JJ, Eilber FR. Primary musculoskeletal neoplasms: effectiveness of core-needle biopsy. Radiology. 1999;212:682–6.
Roh J-L, Lee Y-W, Kim J-M. Clinical utility of fine-needle aspiration for diagnosis of head and neck lymphoma. Eur J Surg Oncol. 2008;34:817–21.
Didolkar MM, Anderson ME, Hochman MG, Rissmiller JG, Goldsmith JD, Gebhardt MG, et al. Image guided core needle biopsy of musculoskeletal lesions: are nondiagnostic results clinically useful? Clin Orthop Relat Res. 2013;471:3601–9.
Ogilvie CM, Torbert JT, Finstein JL, Fox EJ, Lackman RD. Clinical utility of percutaneous biopsies of musculoskeletal tumors. Clin Orthop Relat Res. 2006;450:95–100.
Potter BK. CORR Insights®: image guided core needle biopsy of musculoskeletal lesions—are nondiagnostic results clinically useful? Clin Orthop Relat Res. 2013;471:3610–1.
Wu JS, Goldsmith JD, Horwich PJ, Shetty SK, Hochman MG. Bone and soft-tissue lesions: what factors affect diagnostic yield of image-guided core-needle biopsy? Radiology. 2008;248:962–70.
Yang J, Frassica FJ, Fayad L, Clark DP, Weber KL. Analysis of nondiagnostic results after image-guided needle biopsies of musculoskeletal lesions. Clin Orthop Relat Res. 2010;468:3103–11.
Gustafson P, Dreinhöfer KE, Rydholm A. Soft tissue sarcoma should be treated at a tumor center: a comparison of quality of surgery in 375 patients. Acta Orthop Scand. 1994;65:47–50.
Barman I, Dingari NC, Saha A, McGee S, Galindo LH, Liu W, et al. Application of Raman spectroscopy to identify microcalcifications and underlying breast lesions at stereotactic core needle biopsy. Cancer Res. 2013;73:3206–15.
Choi SH, Chae EJ, Kim J-E, Kim EY, Oh SY, Hwang HJ, et al. Percutaneous CT-guided aspiration and core biopsy of pulmonary nodules smaller than 1 cm: analysis of outcomes of 305 procedures from a tertiary referral center. AJR Am J Roentgenol. 2013;201:964–70.
Lorenz JM. Updates in percutaneous lung biopsy: new indications, techniques and controversies. Semin Intervent Radiol. 2012;29:319–24.
Adams J, Wu HH. The utility of fine-needle aspiration in the diagnosis of primary and metastatic tumors to the lung: a retrospective examination of 1,032 cases. Acta Cytol. 2012;56:590–5.
Londoño DC, Wuerstle MC, Thomas AA, Salazar LE, Hsu J-WY, Danial T, et al. Accuracy and implications of percutaneous renal biopsy in the management of renal masses. Perm J. 2013;17:4–7.
Park SY, Park BK, Kim CK, Kwon GY. Ultrasound-guided core biopsy of small renal masses: diagnostic rate and limitations. J Vasc Interv Radiol. 2013;24:90–6.
Dedhia PH, Rubio GA, Cohen MS, Miller BS, Gauger PG, Hughes DT. Potential effects of molecular testing of indeterminate thyroid nodule fine needle aspiration biopsy on thyroidectomy volume. World J Surg. 2014
Tatomirovic Z, Skuletic V, Bokun R, Trimcev J, Radic O, Cerovic S, et al. Fine needle aspiration cytology in the diagnosis of head and neck masses: accuracy and diagnostic problems. J BUON. 2009;14:653–9.
Fung AD, Collins JA, Campassi C, Ioffe OB, Staats PN. Performance characteristics of ultrasound-guided fine-needle aspiration of axillary lymph nodes for metastatic breast cancer employing rapid on-site evaluation of adequacy: analysis of 136 cases and review of the literature. Cancer Cytopathol. 2013
Mahony GT, Mahony BS. Low nondiagnostic rate for fine-needle capillary sampling biopsy of thyroid nodules: a singular experience. J Ultrasound Med. 2013;32:2155–61.
Zhao H, Xie Z, Zhou Z-L, Sui X-Z, Wang J. Diagnostic value of endobronchial ultrasound-guided transbronchial needle aspiration in intrapulmonary lesions. Chin Med J. 2013;126:4312–5.
Odejide OO, Cronin AM, DeAngelo DJ, Bernazzoli ZA, Jacobson JO, Rodig SJ, et al. Improving the quality of bone marrow assessment: impact of operator techniques and use of a specimen preparation checklist. Cancer. 2013;119:3472–8.
Hunter S, Samir A, Eisner B, Gervais D, Maher M, Hahn P, et al. Diagnosis of renal lymphoma by percutaneous image guided biopsy: experience with 11 cases. J Urol. 2006;176:1952–6. discussion 1956.
Na DG, Kim J, Sung JY, Baek JH, Jung KC, Lee H, et al. Core-needle biopsy is more useful than repeat fine-needle aspiration in thyroid nodules read as nondiagnostic or atypia of undetermined significance by the Bethesda system for reporting thyroid cytopathology. Thyroid. 2012;22:468–75.
Simsek GG, Ertuğrul DT, Guresci S, Şimşek H. Is there a role for on-site evaluation of thyroid fine needle aspiration to reduce the nondiagnostic rate? Endocr Pathol. 2013;24:57–61.
Cermak TS, Wang B, DeBrito P, Carroll J, Haddad N, Sidawy MK. Does on-site adequacy evaluation reduce the nondiagnostic rate in endoscopic ultrasound-guided fine-needle aspiration of pancreatic lesions? Cancer Cytopathol. 2012;120:319–25.
Virayavanich W, Ringler MD, Chin CT, Baum T, Giaconi JC, O’Donnell RJ, et al. CT-guided biopsy of bone and soft-tissue lesions: role of on-site immediate cytologic evaluation. J Vasc Interv Radiol. 2011;22:1024–30.
Salomao M, Remotti H, Allendorf JD, Poneros JM, Sethi A, Gonda TA, et al. Fine-needle aspirations of pancreatic serous cystadenomas: improving diagnostic yield with cell blocks and α-inhibin immunohistochemistry. Cancer Cytopathol. 2014;122:33–9.
Deonizio JMD, Guitart J. The role of molecular analysis in cutaneous lymphomas. Semin Cutan Med Surg. 2012;31:234–40.
Anderson MD, Abel TW, Moots PL. Re-evaluation of nondiagnostic biopsies of suspected low-grade glioma using isocitrate dehydrogenase 1 mutation immunohistochemistry. Neuro-Oncology. 2013;15:811–3.
Nielsen JA, Lager DJ, Lewin M, Weber JJ, Roberts CA. Incidence of diagnostic change in colorectal polyp specimens after deeper sectioning at 2 different laboratories staffed by the same pathologists. Am J Clin Pathol. 2013;140:231–7.
Li QK, Nugent SL, Straseski J, Cooper D, Riedel S, Askin FB, et al. Thyroglobulin measurements in fine-needle aspiration cytology of lymph nodes for the detection of metastatic papillary thyroid carcinoma. Cancer Cytopathol. 2013;121:440–8.
Brenner RJ, Fajardo L, Fisher PR, Dershaw DD, Evans WP, Bassett L, et al. Percutaneous core biopsy of the breast: effect of operator experience and number of samples on diagnostic accuracy. AJR Am J Roentgenol. 1996;166:341–6.
Source of funding
N/A
Compliance with ethical standards
N/A
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chang, C.Y., Huang, A.J., Bredella, M.A. et al. Percutaneous CT-guided needle biopsies of musculoskeletal tumors: a 5-year analysis of non-diagnostic biopsies. Skeletal Radiol 44, 1795–1803 (2015). https://doi.org/10.1007/s00256-015-2235-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00256-015-2235-0