Abstract
Objective. A meta-analysis was carried out of clinical trials published between 1987 and 2001 in respect of the clinical pharmacology and safety as well as the diagnostic efficacy of gadolinium-DTPA (Gd-DTPA) for direct intra-articular injection before MRI examination.
Design. Scientific papers (clinical, postmortem and experimental studies) and information from the manufacturer regarding intra-articular injection of Gd-DTPA that addressed questions of mode of action, optimal concentration and dose, elimination and safety were reviewed. Clinical studies were classified according to their study design. The sensitivity, specificity and accuracy of MR arthrography (MRA) were compared with a "gold standard" (arthroscopy, arthrotomy) and other radiological evidence for different joints.
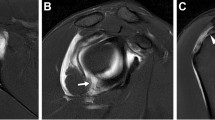
Results. Fifty-two clinical studies of the overall 112 studies addressed aspects of diagnostic efficacy of MRA in patients or in healthy volunteers. The shoulder was the most assessed joint (29 of 52 studies). Good (>80%) or even excellent (90–100%) sensitivity, specificity and accuracy were found for MRA in most indications, especially for the shoulder and knee joints and induced extension of rotator cuff lesions, labrum abnormalities and postoperative meniscal tears. Two millimoles per liter has proven to be the best concentration for intra-articular administration of Gd-DTPA. After passive complete diffusion from the joint within 6–24 h, complete and rapid renal elimination takes place after intra-articular injection. Local safety proved to be excellent after intra-articular administration of Gd-DTPA. Regarding systemic tolerance almost no side effects have been reported, but the same safety considerations apply for intra-articular administration of Gd-DTPA as for intravenous injection.
Conclusions. The diagnostic efficacy of intra-articular MRA in most clinical conditions affecting major joints is greater than that of plain MRI. In some diagnostic problems MRA achieves almost the same sensitivity and specificity as the surgical gold standard. Given a sterile application, the intra-articular administration of Gd-DTPA in a concentration of 2 mmol/l prior to MRI is a safe procedure.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Schulte-Altedorneburg, .G., Gebhard, .M., Wohlgemuth, .W. et al. MR arthrography: pharmacology, efficacy and safety in clinical trials. Skeletal Radiol 32, 1–12 (2003). https://doi.org/10.1007/s00256-002-0595-8
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00256-002-0595-8