Abstract
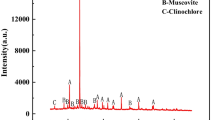
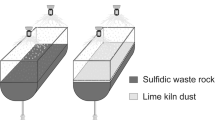
Leaching of two contrasting types of sulphidic tailings in humidity cells has been performed. The release of heavy metals and the oxidation rate have been studied. Tailings from the Laver mine contain a few percent sulphides and lack carbonates, whereas tailings from the Stekenjokk mine are both sulphide- and carbonate-rich. The results showed that in the leachates from the Laver samples, the metal concentrations increased and pH decreased with time, indicating an increased oxidation rate. In the Stekenjokk samples, pH remained high during the experiment, thereby keeping the metal concentrations low in the leachates. The oxidation rate also decreased with time, probably due to Fe-hydroxide coatings on sulphide surfaces. The results show that addition of carbonates and the maintenance of a high pH not only reduce the solubility of heavy metals, but also decrease the oxidation rate of sulphides.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 20 January 1998 · Accepted: 2 April 1998
Rights and permissions
About this article
Cite this article
Holmström, H., Ljungberg, J. & Öhlander, B. Role of carbonates in mitigation of metal release from mining waste. Evidence from humidity cells tests. Environmental Geology 37, 267–280 (1999). https://doi.org/10.1007/s002540050384
Issue Date:
DOI: https://doi.org/10.1007/s002540050384