Abstract
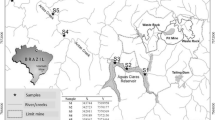
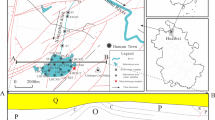
Sulfur-isotope (δ34S) values and weight (%) of acid-volatile sulfur (AVS), chrome-reducible sulfide (CRS), elemental sulfur (ES), and acid-soluble sulfates were determined in Balya Mine ore rock, mine wastes, and Kocacay River and Lake Manyas sediments. Estimation of isotopic fractionation (Δ34S) between product sulfate and initial CRS (pyrite) was used to evaluate the progress of sulfide oxidation in the mine-waste area. Water- and acid-soluble sulfate produced from different mine-waste samples, such as metallurgical waste (MW) and waste rock (WR), in laboratory experiments also shows distinct δ34S values and allows identification of the acid-mine-drainage sources in the mine-waste area. Average δ34SSO4values are −1.43‰ for MW (n=4) and +2.06‰ for WR (n=8). Short (24 hr) and long (60 days) term leach experiments were considered using alternating wet/dry conditions to simulate sulfate-production capacity and metal-discharge characteristics for MW and WR piles. Release of heavy metals follows the order of Pb2+ >Mn2+ >Zn2+ >Cu2+ for these pile samples. Values of δ34SSO4 for river water that was collected after 3–4 h of heavy rainfall are close to values of δ34SSO4 for water-soluble sulfates from mine-waste piles used in laboratory leach experiments.




Similar content being viewed by others
References
Öslund P, Torssander P, Mörth CM, Claesson S (1995) Lead an sulfur isotope dilution during dispersion from the Falun mining area. J Geochem Explor 52:91–95
Agdemir N, Kirikoglu S, Lehmann B, Tietze J (1994) Petrology and alteration geochemistry of the epithermal Balya Pb–Zn–Ag deposits, NW Turkey. Miner Deposita 29:366–371
Akyol Z (1982) Geology, mineralogy and ore potential of the Balikesir–Balya region (in Turkish) Istanbul. Earth Sci Rev 3(1–2):168–169
Aykol A, Budakoglu M (in press) Metal dispersion in sediments of the Kocacay River draining the historic Balya Pb–Zn Mine-debris sites, NW Anatolia, Turkey. Neues Jahrbuch fuer Mineralogie-Abhandlungen
Aykol A, Budakoglu M, Kumral M, Turan M, Gultekin AH, Esenli V, Yavuz F, Orgun Y (2003) Heavy metal pollution and acid drainage from the abandoned Balya Pb–Zn Mine. Environ Geol 45(2):198–208
Bruchert V, Pratt LM (1996) Contemporaneous early diagenetic formation of organic and nonorganic sulfur in estuarine sediments from St. Andrew Bay, Florida, USA. Geochim Cosmochim Acta 60(13):2325–2332
Buckley AN, Woods R (1987) The surface oxidation of pyrite. Appl Surf Sci 27:437–452
Fry B, Ruf W, Gest H, Hayes JM (1988) Sulfur isotope effects associated with oxidation of sulfide by O2 in aqueous-solution. Chem Geol 73(3):205–210
Fu M, Changkakoti A, Krouse HR, Gray J, Kwak TAP (1991) An oxygen, hydrogen, sulfur, and carbon isotope study of carbonate-replacement (skarn) tin deposits of the Dachang tin field, China. Econ Geol 86:1683–1703
Gleisner M, Herbert RB (2002) Sulfide mineral oxidation in freshly processed tailings: batch experiments. J Geochem Explor 76(3):139–153
Hsu SC, Maynard JB (1999) The use of sulfur isotopes to monitor the effectiveness of constructed wetlands in controlling acid mine drainage. Environ Eng Policy 1:223–233
Jorgensen BB (1983) The microbial iron cycle, Microbial Geochemistry. In: Krumbein WE (ed) Blackwell Scientific Publications, London, pp 329
Nealson KH (1983) The microbial sulphur cycle, Microbial Geochemistry. In: Krumbein WE (ed) Blackwell Scientific Pubications, London pp 329
Panettiere P, Cortecci G, Dinelli E, Bencini A, Guidi M (2000) Chemistry and sulfur isotopic composition of precipitation at Bologna, Italy. Appl Geochem 15(10):1455–1467
Parafiniuk J (1996) Sulfate minerals and their origin in the weathering zone of the pyrite-bearing schists at Wiesciszowice (Rudawy Janowickie Mts, Western Sudetes). Acta Geologica Polonica 46(3–4):353–414
Pašava J, Křibek B, Dobeš P, Vavřin I, Žák K, Delian F, Tao Z, Boiron MC (2003) Tin-polymetallic sulfide deposits in the eastern part of the Dachang tin field (South China) and the role of black shales in their origin. Miner Deposita 38:39–66
Rimstidt JD, Vaughan DJ (2003) Pyrite oxidation: a state of the art assessment of the reaction mechanism. Geochim Cosmochim Acta 67(5):873–880
Sanchez AG, Ayuso EA, De Blas OJ (1999) Sorption of heavy metals from industrial waste water by low-cost mineral silicates. Clay Miner 34(3):469–477
Schwertmann U, Taylor RM (1995) Iron oxides. In: Dixon JB, Weed SB (eds) Minerals in soil environments. Soil Science Society of America Inc, pp 379–438
Taylor BE, Wheeler MC (1994) Sulfur-isotope and oxygen-isotope geochemistry of acid-mine drainage in the western United-States field and experimental studies revisited. ACS Symp Ser 550:481–514
Taylor BE, Wheeler MC, Nordstrom DK (1984) Oxygen and sulfur compositions of sulphate in acid mine drainage: evidence for oxidation mechanisms. Nature 308:538–541
Toran L, Harris RF (1989) Interpretation of sulfur and oxygen isotopes in biological and abiological sulfide oxidation. Geochim Cosmochim Acta 53(9):12341–2348
Van Stempvoort DR, Reardon EJ, Fritz P (1990) Fractionation of sulfur and oxygen isotopes in sulfate soil sorption. Geochim Cosmochim Acta 54:2817–2826
Zaback DA, Pratt LM (1992) Isotopic and speciation of sulfur in the Miocene Monterey formation-Re-evaluation of sulfur reactions during early diagenesis in marine environments. Geochim Cosmochim Acta 56(2):763–774
Acknowledgements
Financial support was provided by the Istanbul Technical University Foundation. Isotope analyses carried out at the Department of Geological Science, Indiana University. The authors wish to specially thank Dr. Atilla Aykol, Jon Fong, Dr. Brandy Anglen, and Dr. Scout Struck for their technical support, also David Dye for Raman spectra studies.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Budakoglu, M., Pratt, L.M. Sulfur-isotope distribution and contamination related to the Balya Pb–Zn Mine in Turkey. Environ Geol 47, 773–781 (2005). https://doi.org/10.1007/s00254-004-1202-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00254-004-1202-1