Abstract
Lovastatin has great medical and economic importance, and its production in Aspergillus terreus is positively regulated at transcriptional level, by reactive oxygen species (ROS) generated during idiophase. To investigate the role of the transcription factor Yap1 in the regulation of lovastatin biosynthesis by ROS, an orthologue of yap1 was identified in A. terreus TUB F-514 and knocked down (silenced) by RNAi. Results confirmed that the selected knockdown strain (Siyap1) showed decreased yap1 expression in both culture systems (submerged and solid-state fermentation). Transformants showed higher sensitivity to oxidative stress. Interestingly, knockdown mutant showed higher ROS levels in idiophase and an important increase in lovastatin production in submerged and solid-state fermentations: 60 and 70% increase, respectively. Furthermore, sporulation also increased by 600%. This suggested that AtYap1 was functioning as a negative regulator of the biosynthetic genes, and that lack of AtYap1 in the mutants would be derepressing these genes and could explain increased production. However, we have shown that lovastatin production is proportional to ROS levels, so ROS increase in the mutants alone could also be the cause of production increase. In this work, when ROS levels were decreased with antioxidant, to the levels shown by the parental strain, the lovastatin production and kinetics were similar to the ones of the parental strain. This means that AtYap1 does not regulate lovastatin biosynthetic genes, and that production increase observed in the knockdown strain was an indirect effect caused by ROS increase. This conclusion is compared with studies on other secondary metabolites produced by other fungal species.
Key points
• ROS regulates lovastatin biosynthesis at transcriptional level, in solid-state, and in submerged fermentations.
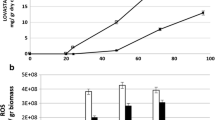
• ATyap1 knockdown mutants showed important lovastatin production increases (60 and 70%) and higher ROS levels.
• When ROS were decreased in the silenced mutant to the parental strain’s level, lovastatin kinetics were identical to the parental strain’s.






Similar content being viewed by others
Data Availability
The authors confirm that the data supporting the findings of this study will be available on reasonable request.
References
Aguirre J, Ríos-Momberg M, Hewitt D, Hansberg W (2005) Reactive oxygen species and development in microbial eukaryotes. Trends Microbiol 13:111–118. https://doi.org/10.1016/j.tim.2005.01.007
Aguirre J, Hansberg W, Navarro R (2006) Fungal responses to reactive oxygen species. Med Mycol 44:101–107. https://doi.org/10.1080/13693780600900080
Ávila N, Tarragó-Castellanos MR, Barrios-González J (2017) Environmental cues that induce the physiology of solid medium: a study on lovastatin production by Aspergillus terreus. J Appl Microbiol 122(4):1029–1038. https://doi.org/10.1111/jam.13391
Baños JG, Tomasini A, Szakács G, Barrios-González J (2009) High lovastatin production by Aspergillus terreus in solid-state fermentation on polyurethane foam: an artificial inert support. J Biosci Bioeng 108:105–110. https://doi.org/10.1016/j.jbiosc.2009.03.006
Barrios-González J (2012) Solid-state fermentation: physiology of solid medium, its molecular basis and applications. Process Biochem 47:175–185. https://doi.org/10.1016/j.procbio.2011.11.016
Barrios-González J, Miranda RU (2010) Biotechnological production and applications of statins. Appl Microbiol Biotechnol 85:869–883. https://doi.org/10.1007/s00253-009-2239-6
Barrios-González J, Baños JG, Covarrubias AA, Garay-Arroyo A (2008) Lovastatin biosynthetic genes of Aspergillus terreus nare expressed differentially in solid-state and in liquid submerged fermentation. Appl Microbiol Biotechnol 79:179–186. https://doi.org/10.1007/s00253-008-1409-2
Barrios-González J, Pérez-Sánchez A, Bibián ME (2020) New knowledge about the biosynthesis of lovastatin and its production by fermentation of Aspergillus terreus. Appl Microbiol Biotechnol 104:8979–8998. https://doi.org/10.1007/s00253-020-10871-x
Bibián ME, Pérez-Sánchez A, Mejía A, Barrios-González J (2020) Penicillin and cephalosporin biosyntheses are also regulated by reactive oxygen species. Appl Microbiol Biotechnol 104(4):1773–1783. https://doi.org/10.1007/s00253-019-10330-2
Brakhage AA (2013) Regulation of fungal secondary metabolism. Nat Rev Microbiol 11:21–32. https://doi.org/10.1038/nrmicro2916
Cantoral J, Díez B, Barredo JL, Álvarez E, Martín JF (1987) High–frequency transformation of Penicillium chrysogenum. Nat Biotechnol 5:494–497. https://doi.org/10.1038/nbt0587-494
Cepeda-García C, Domínguez-Santos R, García-Rico RO, Garcia-Estrada C, Cajiao A, Fierro F (2014) Direct involvement of the CreA transcription factor in penicillin biosynthesis and expression of the pcbAB gene in Penicillium chrysogenum. Appl Microbiol Biotechnol 98:7113–7124. https://doi.org/10.1007/s00253-014-5760-1
Delaunay A, Isnard AD, Toledano MB (2000) H2O2 sensing through oxidation of the Yap1 transcription factor. EMBO J 19:5157–5166. https://doi.org/10.1093/emboj/19.19.5157
Fierro F, Kosalkova K, Gutiérrez S, Martin JF (1996) Autonomously replicating plasmids carrying the AMA1 region in Penicillium chrysogenum. Curr Genet 29:482–489. https://doi.org/10.1007/BF02221518
Hong SY, Roze LV, Linz JE (2013) Oxidative stress-related transcription factors in the regulation of secondary metabolism. Toxins 5:683–702. https://doi.org/10.3390/toxins5040683
Kennedy J (1999) Modulation of polyketide synthase activity by accessory proteins during lovastatin biosynthesis. Science 284:1368–1372. https://doi.org/10.1126/science.284.5418.1368
Kuge S, Arita M, Murayama A, Maeta K, Izawa S, Inoue Y, Nomoto A (2001) Regulation of the yeast Yap1p nuclear export signal is mediated by redox signal-induced reversible disulfide bond formation. Mol Biol Cell 21:6139–6150. https://doi.org/10.1128/mcb.21.18.6139-6150.2001
Kwon N-J, Garzia A, Espeso EA, Ugalde U, Yu J-H (2010) FlbC is a putative nuclear C2H2 transcription factor regulating development in Aspergillus nidulans. Molecular Microbiol 77:1203–1219. https://doi.org/10.1111/j.1365-2958.2010.07282.x
Linz J (2011) Stress-related transcription factor AtfB integrates secondary metabolism with oxidative stress response in Aspergilli. J Biol Chem 286:35137–35148. https://doi.org/10.1074/jbc.M111.253468
Marcial J, Pérez de los Santos AI, Fernández FJ, Díaz-Godínez G, Montiel-González AM, Tomasini A (2011) Characterization of an aspartic protease produced by Amylomyces rouxii. Rev Mex Ing Quim Revista mexicana de ingeniería química 10:9–16. http://www.scielo.org.mx/scielo.php?script=sci_arttext&pid=S1665-27382011000100002&lng=es&tlng=en
Mendoza-Martínez AE, Lara-Rojas F, Sánchez O, NapA Aguirre J (2017) Mediates a redox regulation of the antioxidant response, carbon utilization and development in Aspergillus nidulans. Front Microbiol 8:516. https://doi.org/10.3389/fmicb.2017.00516
Mendoza-Martínez AE, Cano-Domínguez N, Aguirre J (2020) Yap1 homologs mediate more than the redox regulation of the antioxidant response in filamentous fungi. Fungal Biol 124:253–262. https://doi.org/10.1016/j.funbio.2019.04.001
Miranda RU, Gómez-Quiroz LE, Mejía A, Barrios-González J (2013) Oxidative state in idiophase links reactive oxygen species (ROS) and lovastatin biosynthesis: differences and similarities in submerged- and solid-state fermentations. Fungal Biol 117:85–93. https://doi.org/10.1016/j.funbio.2012.12.001
Miranda RU, Gómez-Quiroz LE, Mendoza M, Pérez-Sánchez A, Fierro F, Barrios-González J (2014) Reactive oxygen species regulate lovastatin biosynthesis in Aspergillus terreus during submerged and solid-state fermentations. Fungal Biol 118:979–989. https://doi.org/10.1016/j.funbio.2014.09.002
Montibus M, Ducos C, Bonnin-Verdal MN, Bormann J, Ponts N, Richard-Forget F, Barreau C (2013) The bZIP transcription factor Fgap1 mediates oxidative stress response and trichothecene biosynthesis but not virulence in Fusarium graminearum. PLoS ONE 8:83377. https://doi.org/10.1371/journal.pone.0083377
Moore RN, Bigam G, Chan JK, Hogg AM, Nakashima TT, Vederas JC (1985) Biosynthesis of the hypocholesterolemic agent mevinolin by by Aspergillus terreus. Determination of the origin of carbon, hydrogen, and oxygen atoms by carbon-13 NMR and mass spectrometry. J Am Chem Soc 107:3694–3701
Mulder KCL, Mulinari F, Franco OL, Soares MSF, Magalhães BS, Parachin NS (2015) Lovastatin production: from molecular basis to industrial process optimization. Adv Biotechnol 33:648–665. https://doi.org/10.1016/j.biotechadv.2015.04.001
Nak-Jung K, Kwang-Soo S, Jae-Hyuk Y (2010) Characterization of the developmental regulator FlbE in Aspergillus fumigatus and Aspergillus nidulans. Fungal Genet Biol 47:981–993. https://doi.org/10.1016/j.fgb.2010.08.009
Pérez-Pérez WD, Carrasco-Navarro U, García-Estrada C, Kosalková K, Gutiérrez-Ruíz MC, Barrios-González J, Fierro F (2022) bZIP transcription factors PcYap1 and PcRsmA link oxidative stress response to secondary metabolism and development in Penicillium chrysogenum. Microb Cell Fact 21(1):50. https://doi.org/10.1186/s12934-022-01765-w
Pérez-Sánchez A, Uribe-Carvajal S, Cabrera-Orefice A, Barrios-González J (2017) Key role of alternative oxidase in lovastatin solid-state fermentation. Appl Microbiol Biotechnol 101(19):7347–7356. https://doi.org/10.1007/s00253-017-8452-9
Pérez-Sánchez A, Bibián ME, Barrios-González J (2022) The Biosynthesis of Penicillin and Cephalosporin C are Regulated by ROS at Transcriptional Level. Curr Microbiol 79:243. https://doi.org/10.1007/s00284-022-02935-0
Reverberi M, Zjalic S, Ricelli A, Punelli F, Camera E, Fabbri C, Picardo M, Fanelli C, Fabbri AA (2008) Modulation of antioxidant defense in Aspergillus parasiticus is involved in aflatoxin biosynthesis: a role for the ApyapA gene. Eukaryotic Cell 7:988–1000. https://doi.org/10.1128/EC.00228-07
Reverberi M, Gazzetti K, Punelli F, Scarpari M, Zjalic S, Ricelli A (2012) Aoyap1 regulates OTA synthesis by controlling cell redox balance in Aspergillus ochraceus. Appl Microbiol Biotechnol 95:1293–304. https://doi.org/10.1007/s00253-012-3985-4
Rodrigues-Pousada C, Devaux F, Caetano SM, Pimentel C, da Silva S, Cordeiro AC (2019) Yeast AP-1 like transcription factors (Yap) and stress response: a current overview. Microb Cell 6:267–285. https://doi.org/10.15698/mic2019.06.679
Roze LV, Hong SY, Linz JE (2013) Aflatoxin biosynthesis: current frontiers. Annu Rev Food Sci Technol 4:293–311. https://doi.org/10.1146/annurev-food-083012-123702
Signs SA, File TM, Tan JS (1984) High-pressure liquid chromatographic method for analysis of cephalosporins. Antimicrob Agents Chemother 26:652–655. https://doi.org/10.1128/AAC.26.5.652
Subhan M, Faryal R, Macreadie I (2016) Exploitation of Aspergillus terreus for the production of natural statins. J Fungi 2:13. https://doi.org/10.3390/jof2020013
Suryanarayan S (2003) Current industrial practice in solid state fermentations for secondary metabolite production: the Biocon India experience. J Biomech Eng 13:189–195. https://doi.org/10.1016/S1369-703X(02)00131-6
Szakács G, Morovján G, Tengerdy RP (1998) Production of lovastatin by a wild strain of Aspergillus terreus. Biotechnol Lett 20:411–41. https://doi.org/10.1023/A:1005391716830
Turrens JF (2003) Mitochondrial formation of reactive oxygen species. J Physiol 552:335–344. https://doi.org/10.1111/j.1469-7793.2003.00335.x
Ullán RV, Godio RP, Teijeira F, Vaca I, García-Estrada C, Feltrer R (2008) RNA-silencing in Penicillium chrysogenum and Acremonium chrysogenum: validation studies using beta-lactam genes expression. J Microbiol Methods 75:209–218. https://doi.org/10.1016/j.mimet.2008.06.001
Wagschal K, Yoshizawa Y, Witter DJ, Liu Y, Vederas JC (1996) Biosynthesis of ML-236C and the hypocholesterolemic agents compactin by Penicillium aurantiogriseum and lovastatin by Aspergillus terreus: determination of the origin of carbon, hydrogen and oxygen atoms by CNMR spectrometry and observation of unusual labelling of acetate-derived oxygens by O2. J Chem Soc 19:2357–2363. https://doi.org/10.1039/P19960002357
Yan C, Lee LH, Davis LI (1998) Crm1p mediates regulated nuclear export of a yeast AP-1- like transcription factor. EMBO J 17:7416–7429. https://doi.org/10.1093/emboj/17.24.741
Funding
This study was funded by Consejo Nacional de Ciencia y Tecnología (CONACyT), Mexico, project CB-2013-01 222028. A. Pérez-Sánchez received scholarships from CONACyT No. 283909.
Author information
Authors and Affiliations
Contributions
JB and AP conceived and designed research. AP conducted experiments. RUM contributed new analytical tools. AM analyzed data. JB and AP prepared the draft and revised the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pérez-Sánchez, A., Mejía, A., Miranda-Labra, R.U. et al. Role of AtYap1 in the reactive oxygen species regulation of lovastatin production in Aspergillus terreus. Appl Microbiol Biotechnol 107, 1439–1451 (2023). https://doi.org/10.1007/s00253-023-12382-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-023-12382-x