Abstract
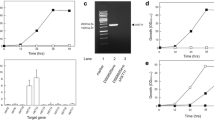
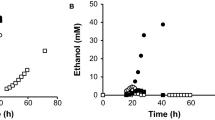
Efficient hexose transporters are essential for the development of industrial yeast strains with high fermentation performance. We previously identified a hexose transporter, CgHxt4, with excellent sugar uptake performance at ultra-high glucose concentrations (200 g/L) in the high sugar fermenting yeast C. glycerinogenes. To understand the working mechanism of this transporter, we constructed 87 mutants and examined their glucose uptake performance. The results revealed that five residues (N321, N322, F325, G426, and P427) are essential for the efficient glucose transport of CgHxt4. Subsequently, we focused our analysis on the roles of N321 and P427. Specifically, N321 and P427 are likely to play a role in glucose coordination and conformational flexibility, respectively. Our results help to expand the application potential of this transporter and provide insights into the working mechanism of yeast hexose transporter.
Key points
• Five residues, transmembrane segments 7 and 10, were found to be essential for CgHxt4.
• N321 and P427 are likely to play a role in glucose coordination and conformational flexibility, respectively.
• Chimeric CgHxt5.4TM7 significantly enhanced the performance of CgHxt5.





Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article (and its supplementary information files).
References
Bailey TL, Boden M, Buske FA, Frith M, Grant CE, Clementi L, Ren J, Li WW, Noble WS (2009) MEME SUITE: tools for motif discovery and searching. Nucleic Acids Res 37(Web Server issue):W202–8 https://doi.org/10.1093/nar/gkp335
Chen X, Fang H, Rao Z, Shen W, Zhuge B, Wang Z, Zhuge J (2008) An efficient genetic transformation method for glycerol producer Candida glycerinogenes. Microbiol Res 163(5):531–537. https://doi.org/10.1016/j.micres.2008.05.003
Deng D, Sun P, Yan C, Ke M, Jiang X, Xiong L, Ren W, Hirata K, Yamamoto M, Fan S, Yan N (2015) Molecular basis of ligand recognition and transport by glucose transporters. Nature 526(7573):391–396. https://doi.org/10.1038/nature14655
Deng D, Xu C, Sun P, Wu J, Yan C, Hu M, Yan N (2014) Crystal structure of the human glucose transporter GLUT1. Nature 510(7503):121–125. https://doi.org/10.1038/nature13306
El-Dalatony MM, Saha S, Govindwar SP, Abou-Shanab RAI, Jeon BH (2019) Biological conversion of amino acids to higher alcohols. Trends Biotechnol 37(8):855–869. https://doi.org/10.1016/j.tibtech.2019.01.011
Farwick A, Bruder S, Schadeweg V, Oreb M, Boles E (2014) Engineering of yeast hexose transporters to transport D-xylose without inhibition by D-glucose. Proc Natl Acad Sci U S A 111(14):5159–5164. https://doi.org/10.1073/pnas.1323464111
Jiang Y, Shen Y, Gu L, Wang Z, Su N, Niu K, Guo W, Hou S, Bao X, Tian C, Fang X (2020) Identification and characterization of an efficient D-xylose transporter in Saccharomyces cerevisiae. J Agric Food Chem 68(9):2702–2710. https://doi.org/10.1021/acs.jafc.9b07113
Kasahara M, Maeda M (1998) Contribution to substrate recognition of two aromatic amino acid residues in putative transmembrane segment 10 of the yeast sugar transporters Gal2 and Hxt2. J Biol Chem 273(44):29106–29112. https://doi.org/10.1074/jbc.273.44.29106
Kasahara M, Shimoda E, Maeda M (1996) Transmembrane segment 10 is important for substrate recognition in Ga12 and Hxt2 sugar transporters in the yeast Saccharomyces cerevisiae. FEBS Lett 389(2):174–178. https://doi.org/10.1016/0014-5793(96)00567-4
Kasahara M, Shimoda E, Maeda M (1997) Amino acid residues responsible for galactose recognition in yeast Gal2 transporter. J Biol Chem 272(27):16721–16724. https://doi.org/10.1074/jbc.272.27.16721
Kasahara T, Ishiguro M, Kasahara M (2004) Comprehensive chimeric analysis of amino acid residues critical for high affinity glucose transport by Hxt2 of Saccharomyces cerevisiae. J Biol Chem 279(29):30274–30278. https://doi.org/10.1074/jbc.m403762200
Kasahara T, Ishiguro M, Kasahara M (2006) Eight amino acid residues in transmembrane segments of yeast glucose transporter Hxt2 are required for high affinity transport. J Biol Chem 281(27):18532–18538. https://doi.org/10.1074/jbc.M602123200
Kasahara T, Kasahara M (2000a) Interaction between the critical aromatic amino acid residues Tyr(352) and Phe(504) in the yeast Ga12 transporter. FEBS Lett 471(1):103–107. https://doi.org/10.1016/S0014-5793(00)01371-5
Kasahara T, Kasahara M (2000b) Three aromatic amino acid residues critical for galactose transport in yeast Gal2 transporter. J Biol Chem 275(6):4422–4428. https://doi.org/10.1074/jbc.275.6.4422
Kasahara T, Kasahara M (2003) Transmembrane segments 1, 5, 7 and 8 are required for high-affinity glucose transport by Saccharomyces cerevisiae Hxt2 transporter. Biochem J 372(Pt 1):247–252. https://doi.org/10.1042/BJ20030044
Kasahara T, Kasahara M (2010) Identification of a key residue determining substrate affinity in the yeast glucose transporter Hxt7: a two-dimensional comprehensive study. J Biol Chem 285(34):26263–26268. https://doi.org/10.1074/jbc.M110.149716
Kasahara T, Maeda M, Ishiguro M, Kasahara M (2007) Identification by comprehensive chimeric analysis of a key residue responsible for high affinity glucose transport by yeast HXT2. J Biol Chem 282(18):13146–13150. https://doi.org/10.1074/jbc.C700041200
Kasahara T, Shimogawara K, Kasahara M (2011) Crucial effects of amino acid side chain length in transmembrane segment 5 on substrate affinity in yeast glucose transporter Hxt7. Biochemistry 50(40):8674–8681. https://doi.org/10.1021/bi200958s
Kelley LA, Mezulis S, Yates CM, Wass MN, Sternberg MJ (2015) The Phyre2 web portal for protein modeling, prediction and analysis. Nat Protoc 10(6):845–858. https://doi.org/10.1038/nprot.2015.053
Koppram R, Tomas-Pejo E, Xiros C, Olsson L (2014) Lignocellulosic ethanol production at high-gravity: challenges and perspectives. Trends Biotechnol 32(1):46–53. https://doi.org/10.1016/j.tibtech.2013.10.003
Kumar S, Stecher G, Li M, Knyaz C, Tamura K (2018) MEGA X: molecular evolutionary genetics analysis across computing platforms. Mol Biol Evol 35(6):1547–1549. https://doi.org/10.1093/molbev/msy096
Li H, Schmitz O, Alper HS (2016) Enabling glucose/xylose co-transport in yeast through the directed evolution of a sugar transporter. Appl Microbiol Biotechnol 100(23):10215–10223. https://doi.org/10.1007/s00253-016-7879-8
Liang Z, Liu D, Lu X, Zong H, Song J, Zhuge B (2018) Identification and characterization from Candida glycerinogenes of hexose transporters having high efficiency at high glucose concentrations. Appl Microbiol Biotechnol 102(13):5557–5567. https://doi.org/10.1007/s00253-018-9027-0
Lomize MA, Pogozheva ID, Joo H, Mosberg HI, Lomize AL (2012) OPM database and PPM web server: resources for positioning of proteins in membranes. Nucleic Acids Res 40(Database issue):D370-D376. https://doi.org/10.1093/nar/gkr703
Madej MG, Sun L, Yan N, Kaback HR (2014) Functional architecture of MFS D-glucose transporters. Proc Natl Acad Sci U S A 111(7):E719–E727. https://doi.org/10.1073/pnas.1400336111
Markham KA, Alper HS (2018) Synthetic biology expands the industrial potential of Yarrowia lipolytica. Trends Biotechnol 36(10):1085–1095. https://doi.org/10.1016/j.tibtech.2018.05.004
Nijland JG, Driessen AJM (2019) Engineering of pentose transport in Saccharomyces cerevisiae for biotechnological applications. Front Bioeng Biotechnol 7:464. https://doi.org/10.3389/fbioe.2019.00464
Nomura N, Verdon G, Kang HJ, Shimamura T, Nomura Y, Sonoda Y, Hussien SA, Qureshi AA, Coincon M, Sato Y, Abe H, Nakada-Nakura Y, Hino T, Arakawa T, Kusano-Arai O, Iwanari H, Murata T, Kobayashi T, Hamakubo T, Kasahara M, Iwata S, Drew D (2015) Structure and mechanism of the mammalian fructose transporter GLUT5. Nature 526(7573):397–401. https://doi.org/10.1038/nature14909
Pacheco A, Donzella L, Hernandez-Lopez MJ, Almeida MJ, Prieto JA, Randez-Gil F, Morrissey JP, Sousa MJ (2020) Hexose transport in Torulaspora delbrueckii: identification of Igt1, a new dual-affinity transporter. FEMS Yeast Res 20(1):foaa004. https://doi.org/10.1093/femsyr/foaa004
Park MS (2015) Molecular dynamics simulations of the human glucose transporter GLUT1. PLoS ONE 10(4):e0125361. https://doi.org/10.1371/journal.pone.0125361
Paulsen PA, Custodio TF, Pedersen BP (2019) Crystal structure of the plant symporter STP10 illuminates sugar uptake mechanism in monosaccharide transporter superfamily. Nat Commun 10(1):407. https://doi.org/10.1038/s41467-018-08176-9
Pellegrini-Calace M, Maiwald T, Thornton JM (2009) PoreWalker: a novel tool for the identification and characterization of channels in transmembrane proteins from their three-dimensional structure. PLoS Comput Biol 5(7):e1000440. https://doi.org/10.1371/journal.pcbi.1000440
Qureshi AA, Suades A, Matsuoka R, Brock J, McComas SE, Nji E, Orellana L, Claesson M, Delemotte L, Drew D (2020) The molecular basis for sugar import in malaria parasites. Nature 578(7794):321–325. https://doi.org/10.1038/s41586-020-1963-z
Reider Apel A, Ouellet M, Szmidt-Middleton H, Keasling JD, Mukhopadhyay A (2016) Evolved hexose transporter enhances xylose uptake and glucose/xylose co-utilization in Saccharomyces cerevisiae. Sci Rep 6:19512. https://doi.org/10.1038/srep19512
Rodrigues CH, Pires DE, Ascher DB (2018) DynaMut: predicting the impact of mutations on protein conformation, flexibility and stability. Nucleic Acids Res 46(W1):W350–W355. https://doi.org/10.1093/nar/gky300
Rossi G, Sauer M, Porro D, Branduardi P (2010) Effect of HXT1 and HXT7 hexose transporter overexpression on wild-type and lactic acid producing Saccharomyces cerevisiae cells. Microb Cell Fact 9:15. https://doi.org/10.1186/1475-2859-9-15
Roy A, Dement AD, Cho KH, Kim JH (2015) Assessing glucose uptake through the yeast hexose transporter 1 (Hxt1). PLoS ONE 10(3):e0121985. https://doi.org/10.1371/journal.pone.0121985
Roy A, Kim YB, Cho KH, Kim JH (2014) Glucose starvation-induced turnover of the yeast glucose transporter Hxt1. Biochim Biophys Acta 1840(9):2878–2885. https://doi.org/10.1016/j.bbagen.2014.05.004
Screpanti E, Hunte C (2007) Discontinuous membrane helices in transport proteins and their correlation with function. J Struct Biol 159(2):261–267. https://doi.org/10.1016/j.jsb.2007.01.011
Shin HY, Nijland JG, de Waal PP, Driessen AJM (2017) The amino-terminal tail of Hxt11 confers membrane stability to the Hxt2 sugar transporter and improves xylose fermentation in the presence of acetic acid. Biotechnol Bioeng 114(9):1937–1945. https://doi.org/10.1002/bit.26322
Sun L, Zeng X, Yan C, Sun X, Gong X, Rao Y, Yan N (2012) Crystal structure of a bacterial homologue of glucose transporters GLUT1-4. Nature 490(7420):361–366. https://doi.org/10.1038/nature11524
van der Hoek SA, Borodina I (2020) Transporter engineering in microbial cell factories: the ins, the outs, and the in-betweens. Curr Opin Biotechnol 66:186–194. https://doi.org/10.1016/j.copbio.2020.08.002
Vasylyshyn R, Kurylenko O, Ruchala J, Shevchuk N, Kuliesiene N, Khroustalyova G, Rapoport A, Daugelavicius R, Dmytruk K, Sibirny A (2020) Engineering of sugar transporters for improvement of xylose utilization during high-temperature alcoholic fermentation in Ogataea polymorpha yeast. Microb Cell Fact 19(1):96. https://doi.org/10.1186/s12934-020-01354-9
Vishwakarma P, Banerjee A, Pasrija R, Prasad R, Lynn AM (2018) Phylogenetic and conservation analyses of MFS transporters. 3 Biotech 8(11):462. https://doi.org/10.1007/s13205-018-1476-8
Wang C, Bao X, Li Y, Jiao C, Hou J, Zhang Q, Zhang W, Liu W, Shen Y (2015) Cloning and characterization of heterologous transporters in Saccharomyces cerevisiae and identification of important amino acids for xylose utilization. Metab Eng 30:79–88. https://doi.org/10.1016/j.ymben.2015.04.007
Wang C, Li Y, Qiu C, Wang S, Ma J, Shen Y, Zhang Q, Du B, Ding Y, Bao X (2017) Identification of important amino acids in Gal2p for improving the L-arabinose transport and metabolism in Saccharomyces cerevisiae. Front Microbiol 8:1391. https://doi.org/10.3389/fmicb.2017.01391
Wieczorke R, Krampe S, Weierstall T, Freidel K, Hollenberg CP, Boles E (1999) Concurrent knock-out of at least 20 transporter genes is required to block uptake of hexoses in Saccharomyces cerevisiae. FEBS Lett 464(3):123–128. https://doi.org/10.1016/S0014-5793(99)01698-1
Wisedchaisri G, Park MS, Iadanza MG, Zheng H, Gonen T (2014) Proton-coupled sugar transport in the prototypical major facilitator superfamily protein XylE. Nat Commun 5:4521. https://doi.org/10.1038/ncomms5521
Wu Q, Peng Z, Zhang Y, Yang J (2018) COACH-D: improved protein-ligand binding sites prediction with refined ligand-binding poses through molecular docking. Nucleic Acids Res 46(W1):W438–W442. https://doi.org/10.1093/nar/gky439
Xu X, Liu Y, Du G, Ledesma-Amaro R, Liu L (2020) Microbial chassis development for natural product biosynthesis. Trends Biotechnol 38(7):779–796. https://doi.org/10.1016/j.tibtech.2020.01.002
Yan N (2013) Structural advances for the major facilitator superfamily (MFS) transporters. Trends Biochem Sci 38(3):151–159. https://doi.org/10.1016/j.tibs.2013.01.003
Young E, Poucher A, Comer A, Bailey A, Alper H (2011) Functional survey for heterologous sugar transport proteins, using Saccharomyces cerevisiae as a host. Appl Environ Microbiol 77(10):3311–3319. https://doi.org/10.1128/AEM.02651-10
Young EM, Comer AD, Huang H, Alper HS (2012) A molecular transporter engineering approach to improving xylose catabolism in Saccharomyces cerevisiae. Metab Eng 14(4):401–411. https://doi.org/10.1016/j.ymben.2012.03.004
Young EM, Tong A, Bui H, Spofford C, Alper HS (2014) Rewiring yeast sugar transporter preference through modifying a conserved protein motif. Proc Natl Acad Sci U S A 111(1):131–136. https://doi.org/10.1073/pnas.1311970111
Zhuge J, Fang HY, Wang ZX, Chen DZ, Jin HR, Gu HL (2001) Glycerol production by a novel osmotolerant yeast Candida glycerinogenes. Appl Microbiol Biotechnol 55(6):686–692. https://doi.org/10.1007/s002530100596
Acknowledgements
The authors acknowledge Dr. Eckhard Boles for the gift of the S. cerevisiae EBY.VW4000 strain.
Funding
This work was supported by the National Natural Science Foundation of China (grant numbers 31970033, 31570052, and 21708016).
Author information
Authors and Affiliations
Contributions
Y. Q. and C. L. designed and conducted the study and wrote the original manuscript. X. L. analyzed the data and contributed to writing. H. Z. supervised the project and provided funding. B. Z. directed the study, reviewed the manuscript, and provided funding.
Corresponding author
Ethics declarations
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Qiao, Y., Li, C., Lu, X. et al. Identification of key residues for efficient glucose transport by the hexose transporter CgHxt4 in high sugar fermentation yeast Candida glycerinogenes. Appl Microbiol Biotechnol 105, 7295–7307 (2021). https://doi.org/10.1007/s00253-021-11567-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-021-11567-6