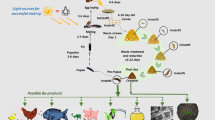
Abstract
The impact of ambient relative humidity (RH) on conidial production of Metarhizium humberi IP 46 microsclerotia (MS) formulated in pellets or granules was investigated, and a promising granular formulation was tested against Aedes aegypti adults to confirm its efficacy. Microcrystalline cellulose (MC) and diatomaceous earth (DE) or a combination of vermiculite (VE), DE and silicon dioxide (SD) were tested as carriers in granular formulations containing MS. A range of 93–96.5% RH was critical for fungal development, and at least 96.5–98.5% RH was required for high conidial production on pellets or granules. Conidial production was clearly higher on pellets and granules prepared with VE than MC as the main carrier. VE granules containing MS were highly active against A. aegypti adults. Most mosquitoes were killed within 6 days after treatment regardless of the exposure time of adults to the formulation (1 min–24 h) or ambient humidity (75 or >98%). Production of conidia on dead adults varied between 7.3 × 106 and 2.2 × 107 conidia/individual, when exposed to MS granules for 12 h and 1 min, respectively. Granular formulations containing VE as the main carrier and MS as the active ingredient of M. humberi have strong potential for use against A. aegypti.
Key points
• High conidial production on granular microsclerotial formulations at >96.5% RH
• Vermiculite is more appropriate as a carrier than microcrystalline cellulose
• Granules with IP 46 microsclerotia are highly active against Aedes aegypti adults
Graphical abstract




Similar content being viewed by others
Data availability
Data available on request.
References
Arthurs SP, Thomas MB (2001) Effects of temperature and relative humidity on sporulation of Metarhizium anisopliae var. acridum in mycosed cadavers of Schistocerca gregaria. J Invertebr Pathol 78:59–65. https://doi.org/10.1006/jipa.2001.5050
Arthurs SP, Thomas MB, Lawton JL (2001) Seasonal patterns of persistence and infectivity of Metarhizium anisopliae var. acridum in grasshopper cadavers in the Sahel. Entomol Exp Appl 100:69–76. https://doi.org/10.1046/j.1570-7458.2001.00849.x
ASTM (American Society for Testing Materials) (1958) Maintaining constant relative humidity by means of aqueous solutions. Standards 6: [F33] 961 p.
Behle RW, Jackson MA (2014) Effect of fermentation media on the production, efficacy, and storage stability of Metarhizium brunneum microsclerotia formulated as a prototype granule. J Econ Entomol 107:582–590. https://doi.org/10.1603/EC13426
Behle RW, Jackson MA, Flor-Weiler LB (2013) Efficacy of a granular formulation containing Metarhizium brunneum F52 (Hypocreales: Clavicipitaceae) microsclerotia against nymphs of Ixodes scapularis (Acari: Ixodidae). J Econ Entomol 106:57–63. https://doi.org/10.1603/EC12226
Behle RW, Richmont DS, Jackson MA, Dunlap CA (2015) Evaluation of Metarhizium brunneum F52 (Hypocreales: Clavicipitaceae) for control of Japanese beetle larvae in turfgrass. J Econ Entomol 108:1587–1595. https://doi.org/10.1093/jee/tov176
Boyette CD, Quimby PC, Connick WJ, Daigle DJ, Fulgham FE (2012) Progress in the production, formulation, and application of mycoherbicides. In: TeBeest D (ed) Microbial control of weeds. Chapman and Hall, New York, pp 209–224
Braga GUL, Flint SD, Miller CD, Anderson AJ, Roberts DW (2001) Variability in response to UV-B among species and strains of Metarhizium isolated from sites at latitudes from 61°N to 54°S. J Invertebr Pathol 78:98–108. https://doi.org/10.1006/jipa.2001.5048
Burges HD (1998) Formulation of microbial biopesticides. Beneficial microorganisms, nematodes and seed treatments. Kluwer Academic Press, Dordrecht.
Chaerunisaa AY, Sriwidodo S, Abdassah M (2019) Microcrystalline cellulose as pharmaceutical excipient. In: Ahmad U, Akhtar J (eds) Pharmaceutical formulation design—recent practices, IntechOpen Limited, London, pp 1−22.
Coley-Smith JR, Cooke RC (1972) Survival and germination of fungal sclerotia. Annu Rev Phytopathol 9:65–92. https://doi.org/10.1146/annurev.py.09.0900171.000433
Evans HC, Samson RA (1982) Cordyceps species and their anamorphs pathogenic on ants (Formicidae) in tropical forest ecosystems 1. The Cephalotes (Myrmicinae) complex. T Brit Mycol Soc 79:431–453. https://doi.org/10.1016/S0007-1536(82)80037-5
Fernandes ÉKK, Marreto RN, Luz C, Catão AML, Paixão FRS, Santos TR, Mascarin GM (2017) Composição e processo de preparação multiparticulada de natureza sólida contendo microescleródios de Metarhizium anisopliae para controle de artrópodes-praga de importância na saúde humana, veterinária e na agropecuária e seus usos. Brazil. Patent registered BR1020170040615.
Gardescu S, Hajek AE, Goble TA, Jackson MA (2017) Metarhizium microsclerotia and hydrogel versus hydromulch: testing fungal formulations against Asian longhorned beetles. Biocontrol Sci Techn 27:918–930. https://doi.org/10.1080/09583157.2017.1362546
Goble TA, Hajek AE, Jackson MA, Gardescu S (2015) Microsclerotia of Metarhizium brunneum F52 applied in hydromulch for control of Asian longhorned beetles (Coleoptera: Cerambycidae). J Econ Entomol 108:433–443. https://doi.org/10.1093/jee/tov013
Goble TA, Gardescu S, Jackson MA, Hajek AE (2016a) Evaluating different carriers of Metarhizium brunneum F52 microsclerotia for control of adult Asian longhorned beetles (Coleoptera: Cerambycidae). Biocontrol Sci Techn 26:1212–1229. https://doi.org/10.1080/09583157.2016.1192103
Goble TA, Gardescu S, Fisher JJ, Jackson MA, Hajek AE (2016b) Conidial production, persistence and pathogenicity of hydromulch formulations of Metarhizium brunneum F52 microsclerotia under forest conditions. Biol Control 95:83–93. https://doi.org/10.1016/j.biocontrol.2016.01.003
Heinig RL, Thomas MB (2015) Interactions between a fungal entomopathogen and malaria parasites within a mosquito vector. Malaria J 14:22. https://doi.org/10.1186/s12936-014-0526-x
Heinig RL, Paaijmans KP, Hancock PA, Thomas MB (2015) The potential for fungal biopesticides to reduce malaria transmission under diverse environmental conditions. J Appl Ecol 52:1558–1566. https://doi.org/10.1111/1365-2664.12522
Hubner-Campos RF, Leles RN, Rodrigues J, Luz C (2013) Efficacy of entomopathogenic hypocrealean fungi against Periplaneta americana. Parasitol Int 62:517–521. https://doi.org/10.1016/j.parint.2013.07.013
Jackson MA, Jaronski ST (2009) Production of microsclerotia of the fungal entomopathogen Metarhizium anisopliae and their potential for use as a biocontrol agent for soil-inhabiting insects. Mycol Res 113:842–850. https://doi.org/10.1016/j.mycres.2009.03.004
Jackson MA, Payne AR (2016) Liquid culture production of fungal microsclerotia. In: Glare TG, Moran-Diez ME (eds) Microbial based biopesticides. Methods and protocols. Springer Nature, New York, pp 71–83
Jackson MA, Schisler DA (1995) Liquid culture production of microsclerotia of Colletotrichum truncatum for use as bioherbicidal propagules. Mycol Res 99:879–884. https://doi.org/10.1016/S0953-7562(09)80745-4
Kimberly MSA, Seow MH (2017) Mode of infection of Metarhizium spp. fungus and their potential as biological control agents. J Fungi 3(2):30. https://doi.org/10.3390/jof3020030
Lacey LA (2017) Microbial control of medically important mosquitoes in tropical climates. In: Lacey LA (ed) Microbial control of insect and mite pests from theory to practice. Academic Press, London, pp 409–430
Lacey LA, Grzywacz D, Shapiro-Ilan DI, Frutos R, Brownbridge M, Goettel MS (2015) Insect pathogens as biological control agents: Back to the future. J Invertebr Pathol 132:1–41. https://doi.org/10.1016/j.jip.2015.07.009
Lee SJ, Jeong SK, Jong SY, Kim JC, Nai YS, Kim JS (2015) Biological control of Asian tiger mosquito, Aedes albopictus (Diptera: Culicidae) using Metarhizium anisopliae JEF-003 millet grain. J Asia-Pac Entomol 18:217–221. https://doi.org/10.1016/j.aspen.2015.02.003
Leles RN, Sousa NA, Rocha LFN, Santos AH, Silva HH, Luz C (2010) Pathogenicity of some hypocrealean fungi to adult Aedes aegypti (Diptera: Culicidae). Parasitol Res 107:1271–1274. https://doi.org/10.1007/s00436-010-1991-y
Lima WP, Neto FC, Macoris MLG, Zuccari DAPC, Dibo MR (2009) Estabelecimento de metodologia para alimentação de Aedes aegypti (Diptera-Culicidae) em camundongos swiss e avaliação da toxicidade e do efeito residual do óleo essencial de Tagetes minuta L. (Asteraceae) em populações de Aedes aegypti. Rev Soc Bras Med 42:638–641. https://doi.org/10.1590/S0037-86822009000600005
Lira AC, Mascarin GM, Delalibera IJ (2020) Microsclerotia production of Metarhizium spp. for dual role as plant biostimulant and control of Spodoptera frugiperda through corn seed coating. Fungal Biol 124:689–699. https://doi.org/10.1016/j.funbio.2020.03.011
Luz C, Fargues J (1998) Factors affecting conidial production of Beauveria bassiana from fungus-killed cadavers of Rhodnius prolixus. J Invertebr Pathol 72:97–103. https://doi.org/10.1006/jipa.1998.4774
Luz C, Fargues J (1999) Dependence of the entomopathogenic fungus, Beauveria bassiana, on high humidity for infection of Rhodnius prolixus. Mycopathologia 146:33–41. https://doi.org/10.1023/A:1007019402490
Luz C, Tai MHH, Santos AH, Rocha LFN, Albernaz DAS, Silva HHG (2007) Ovicidal activity of entomopathogenic Hyphomycetes on Aedes aegypti (L.) (Diptera: Culicidae) under laboratory conditions. J Med Entomol 44:799–804. https://doi.org/10.1093/jmedent/44.5.799
Luz C, Rodrigues J, Rocha LFN (2012) Diatomaceous earth and oil enhance effectiveness of Metarhizium anisopliae against Triatoma infestans. Acta Trop 122:29–35. https://doi.org/10.1016/j.actatropica.2011.11.014
Luz C, Rocha LFN, Montalva C, Souza DA, Botelho ABRZ, Lopes RB, Faria M, Delalibera IJ (2019) Metarhizium humberi sp. nov. (Hypocreales: Clavicipitaceae), a new member of the PARB clade in the Metarhizium anisopliae complex from Latin America. J Invertebr Pathol 166:1–9. https://doi.org/10.1016/j.jip.2019.107216
Mascarin GM, Kobori NN, Vital RCJ, Jackson MA, Quintela ED (2014) Production of microsclerotia by Brazilian strains of Metarhizium spp. using submerged liquid culture fermentation. World J Microb Biot 30:1583–1590. https://doi.org/10.1007/s11274-013-1581-0
Mascarin GM, Lopes RB, Fernandes ÉKK, Delalibera I, Luz C, Faria M (2019) Current status and perspectives of fungal entomopathogens used for microbial control of arthropod pests in Brazil. J Invertebr Pathol 165:46–53. https://doi.org/10.1016/j.jip.2018.01.001
Mnyone LL, Russell TL, Lyimo IN, Lwetoijera DW, Kirby MJ, Luz C (2009) First report of Metarhizium anisopliae IP 46 pathogenicity in adult Anopheles gambiae s.s. and An. arabiensis (Diptera; Culicidae). Parasite Vector 2:59. https://doi.org/10.1186/1756-3305-2-59
Moyes CL, Vontas J, Martins AJ, Ng LC, Koou SY, Dusfour I, Raghavendra K, Corbel JPV, David JP, Weetman D (2017) Contemporary status of insecticide resistance in the major Aedes vectors of arboviruses infecting humans. PLoS Neglect Trop D 11(7):e0005625. https://doi.org/10.1371/journal.pntd.0005625
Muniz-Paredes F, Miranda-Hernandez F, Loera O (2017) Production of conidia by entomopathogenic fungi: from inoculants to final quality tests. World J Microb Biot 33:57. https://doi.org/10.1007/s11274-017-2229-2
Rai M (2009) Advances in fungal biotechnology. IK International Publishing House, New Delhi
Rocha LFN, Luz C (2011) Activity of Metarhizium spp. and Isaria spp. from the Central Brazilian Cerrado against Triatoma infestans nymphs. T Roy Soc Trop Med H 105:417–419. https://doi.org/10.1016/j.trstmh.2011.04.012
Rocha LFN, Inglis PW, Humber RA, Kipnis A, Luz C (2013) Occurrence of Metarhizium spp. in Central Brazilian soils. J Bas Microbiol 53:251–259. https://doi.org/10.1002/jobm.201100482
Rocha LFN, Sousa NA, Rodrigues J, Catão AML, Marques CS, Fernandes ÉKK, Luz C (2015) Efficacy of Tolypocladium cylindrosporum against Aedes aegypti eggs, larvae and adults. J Appl Microbiol 119:1412–1419. https://doi.org/10.1111/jam.12945
Rodrigues J, Borges PR, Fernandes ÉKK, Luz C (2019) Activity of additives and their effect in formulations of Metarhizium anisopliae s.l. IP 46 against Aedes aegypti adults and on post mortem conidiogenesis. Acta Trop 193:192–198. https://doi.org/10.1016/j.actatropica.2019.03.002
Rogge SA, Mayerhofer J, Enkerli J, Bacher S, Grabenweger G (2017) Preventive application of an entomopathogenic fungus in cover crops for wireworm control. BioControl 62:613–623. https://doi.org/10.1007/s10526-017-9816-x
Sandler N, Rantanen J, Heinämäki J, Römer M, Marola M, Yliruusi J (2005) Pellet manufacturing by extrusion-spheronization using process analytical technology. AAPS Pharm Sci Tech 6:174–183. https://doi.org/10.1208/pt060226
Santos AH, Tai MHH, Rocha LFN, Silva HHG, Luz C (2009) Dependence of Metarhizium anisopliae on high humidity for ovicidal activity on Aedes aegypti. Biol Control 50:37–42. https://doi.org/10.1016/j.biocontrol.2009.01.018
Scholte E-J, Knols BGJ, Takken W (2006) Infection of the malaria mosquito Anopheles gambiae with the entomopathogenic fungus Metarhizium anisopliae reduces blood feeding and fecundity. J Invertebr Pathol 91:43–49. https://doi.org/10.1016/j.jip.2005.10.006
Silva RO, Silva HHG, Luz C (2004) Effect of Metarhizium anisopliae isolated from soil samples of the Central Brazilian cerrado against Aedes aegypti larvae under laboratory conditions. Rev Pat Trop 33:207–213. https://doi.org/10.5216/rpt.v33i2.3446
Skinner M, Gouli S, Frank CE, Parker BL, Kim JS (2012) Management of Frankliniella occidentalis (Thysanoptera: Thripidae) with granular formulations of entomopathogenic fungi. Biol Control 63:246–252. https://doi.org/10.1016/j.biocontrol.2012.08.004
Snetselaar J, Andriessen R, Suer RA, Osinga AJ, Knols BGJ, Farenhorst M (2014) Development and evaluation of a novel contamination device that targets multiple life-stages of Aedes aegypti. Parasite Vector 7:200. https://doi.org/10.1186/1756-3305-7-200
Song S (2018) Fungal microsclerotia development: essential prerequisites, influencing factors, and molecular mechanism. Appl Microbiol Biotechnol 102:9873–9880. https://doi.org/10.1007/s00253-018-9400-z
Song Z, Yin Y, Jiang S, Liu J, Wang Z (2014) Optimization of culture medium for microsclerotia production by Nomuraea rileyi and analysis of their viability for use as a mycoinsecticide. BioControl 59:597–605. https://doi.org/10.1007/s10526-014-9589-4
Song Z, Lin Y, Du F, Yin Y, Wang Z (2017) Statistical optimisation of process variables and large-scale production of Metarhizium rileyi (Ascomycetes: Hypocreales) microsclerotia in submerged fermentation. Mycology 8:39–47. https://doi.org/10.1080/21501203.2017.1279688
Sousa NA, Lobo LS, Rodrigues J, Luz C (2013) New insights on the effectiveness of Metarhizium anisopliae formulation and application against Aedes aegypti eggs. Lett Appl Microbiol 57:193–199. https://doi.org/10.1111/lam.12097
Sprenkel RK, Brooks WM (1977) Winter survival of the entomogenous fungus Nomuraea rileyi in North Carolina. J Invertebr Pathol 29:262–266. https://doi.org/10.1016/S0022-2011(77)80031-1
Thomas MB (2018) Biological control of human disease vectors: a perspective on challenges and opportunities. BioControl 63:61–69. https://doi.org/10.1007/s10526-017-9815-y
Throne JE, Weaker DK, Chew V, Baker JE (1995) Probit analysis of correlated data: multiple observations over time at one concentration. J Econ Entomol 88:1510–1512. https://doi.org/10.1093/jee/88.5.1510
Vega FE, Goettel MS, Blackwell M, Chandler D, Jackson MA, Keller S, Koike M, Maniania NK, Monzón A, Ownley BH, Pell JK, Rangel DEN, Roy HE (2009) Fungal entomopathogens: new insights on their ecology. Fung Ecol 2:149–159. https://doi.org/10.1016/j.funeco.2009.05.001
Wang C, Wang S (2017) Insect pathogenic fungi: genomics, molecular interactions, and genetic improvements. Annu Rev Entomol 62:73–90. https://doi.org/10.1146/annurev-ento-031616-035509
Weaver S, Scott C, Charlier C, Vasilakis N, Lecuit M (2018) Zika, chikungunya, and other emerging vector-borne viral diseases. Annu Rev Med 69:6.1–6.14. https://doi.org/10.1146/annurev-med-050715-105122
Willetts HJ, Bullock S (1992) Developmental biology of sclerotia. Mycol Res 96:801–816. https://doi.org/10.1016/S0953-7562(09)81027-7
Winston PW, Bates DH (1960) Saturated solutions for the control of humidity in biological research. Ecology 41:232–237. https://doi.org/10.2307/1931961
Zhou X, Feng MG (2009) Sporulation, storage and infectivity of obligate aphid pathogen Pandora nouryi grown on novel granules of broomcorn millet and polymer gel. J Appl Microbiol 107:1847–1856. https://doi.org/10.1111/j.1365-2672.2009.04363.x
Acknowledgements
The authors thank Elen Regozino Muniz and Prof. Walquíria Arruda (ICB, UFG) for technical assistance, and Prof. Eliana M. Lima, coordinator of FARMATEC-UFG for providing facilities.
Funding
This study was supported by the National Council for Scientific and Technological Development, CNPq, Universal (14/2012 48329/2012-0), Coordination of the Improvement of Higher Education, CAPES/MEC, CNPq/MCTI and Brazilian Ministry of Health, Decit/SCTIE/MS (440506/2016) and scholarships to JR, AC and FP (CNPq) and to AS, JM and TS (CAPES). We also thank CNPq for the grants PQ 308850/2015-7 to ÉF and PQ 303096/2019-5 to CL.
Author information
Authors and Affiliations
Contributions
CL, RM, GM, ÉF and RH conceived the research. JR, AC, FP, TS, JM, RM, GM, ÉF and CL designed the experiments. JR, AC and AS performed the experiments. JR, AC and JM assisted in the insect rearing and analysed the data. JR, AC, RH and CL wrote the paper. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
JR, AC, AS, FP, TS, JM, RM, GM, ÉF, RH and CL declare that they have no conflict of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Rodrigues, J., Catão, A.M.L., dos Santos, A.S. et al. Relative humidity impacts development and activity against Aedes aegypti adults by granular formulations of Metarhizium humberi microsclerotia. Appl Microbiol Biotechnol 105, 2725–2736 (2021). https://doi.org/10.1007/s00253-021-11157-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-021-11157-6