Abstract
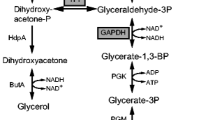
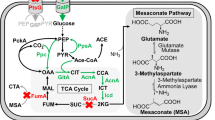
Phosphate is one of the major constituents in growth media. It closely regulates central carbon and energy metabolism. Biochemical reactions in central carbon metabolism are known to be regulated by phosphorylation and dephosphorylation of enzymes. Phosphate scarcity can limit microbial productivity. However, microorganisms are evolved to grow in phosphate starvation environments. This study investigates the effect of phosphate-starved response (PSR) stimuli in wild-type and recombinant Escherichia coli cells cultivated in two different substrates, lactose, and glycerol. Phosphate-starved E. coli culture sustained bacterial growth despite the metabolic burden that emanated from recombinant protein expression albeit with altered dynamics of substrate utilisation. Induction of lactose in phosphate-starved culture led to a 2-fold improvement in product titre of rSymlin and a 2.3-fold improvement in product titre of rLTNF as compared with phosphate-unlimited culture. The results obtained in the study are in agreement with the literature to infer that phosphate starvation or limitation can slow down the microbial growth rate in order to produce recombinant proteins. Further, under PSR conditions, gene expression analysis demonstrated that while selected genes (gapdh, pykF, ppsA, icdA) in glycolysis and pentose phosphate pathway (zwf, gnd, talB, tktA) were up-regulated, other genes in lactose (lacY, lacA) and acetate (ackA, pta) pathway were down-regulated. We have demonstrated that cra, crp, phoB, and phoR are involved in the regulation of central carbon metabolism. We propose a novel cross-regulation between lactose metabolism and phosphate starvation. UDP-galactose, a toxic metabolite that is known to cause cell lysis, has been shown to be significantly reduced due to slow uptake of lactose under PSR conditions. Therefore, E. coli employs a decoupling strategy by limiting growth and redirecting metabolic resources to survive and produce recombinant protein under phosphate starvation conditions.
Key points
• Phosphate starvation controls lactose metabolism, which results in less galactose accumulation.
• Phosphate starvation modulates metabolic flow of central carbon metabolism.
• Product titre improves by 2-fold due to phosphate starvation.
• The approach has been successfully applied to production of two different proteins.








Similar content being viewed by others
References
Borthwick AC, Holms WH, Nimmo HG (1984) The phosphorylation of Escherichia coli isocitrate dehydrogenase in intact cells. Biochem J 222:797–804. https://doi.org/10.1042/bj2220797
Bren A, Park JO, Towbin BD, Dekel E, Rabinowitz JD, Alon U (2016) Glucose becomes one of the worst carbon sources for E. coli on poor nitrogen sources due to suboptimal levels of cAMP. Sci Rep 6:1–10. https://doi.org/10.1038/srep24834
Burnell JN (2010) Cloning and characterization of Escherichia coli DUF299: a bifunctional ADP-dependent kinase-Pi-dependent pyrophosphorylase from bacteria. BMC Biochem 11(1). https://doi.org/10.1186/1471-2091-11-1
Hausjell J, Weissensteiner J, Molitor C, Halbwirth H, Spadiut O (2018) E. coli HMS174 (DE3) is a sustainable alternative to BL21 (DE3). Microb Cell Factories 17:169. https://doi.org/10.1186/s12934-018-1016-6
Huber R, Roth S, Rahmen N, Büchs J (2011) Utilizing high-throughput experimentation to enhance specific productivity of an E. coli T7 expression system by phosphate limitation. BMC Biotechnol 11:22. https://doi.org/10.1186/1472-6750-11-22
Kaleta C, Schäuble S, Rinas U, Schuster S (2013) Metabolic costs of amino acid and protein production in Escherichia coli. Biotechnol J 8:1105–1114. https://doi.org/10.1002/biot.201200267
Kopp J, Slouka C, Ulonska S, Kager J, Fricke J, Spadiut O, Herwig C (2018) Impact of glycerol as carbon source onto specific sugar and inducer uptake rates and inclusion body productivity in E. coli BL21 (DE3). Bioengineering 5:1. https://doi.org/10.3390/bioengineering5010001
Lee SJ, Trostel A, Le P, Harinarayanan R, FitzGerald PC, Adhya S (2009) Cellular stress created by intermediary metabolite imbalances. Proc Natl Acad Sci U S A 106:19515–19520. https://doi.org/10.1073/pnas.0910586106
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Marzan LW, Shimizu K (2011) Metabolic regulation of Escherichia coli and its phoB and phoR genes knockout mutants under phosphate and nitrogen limitations as well as at acidic condition. Microb Cell Factories 10:39. https://doi.org/10.1186/1475-2859-10-39
Merchant SS, Helmann JD (2012) Elemental economy: microbial strategies for optimizing growth in the face of nutrient limitation. In: Advances in microbial physiology. Elsevier, pp 91–210
Murray EL, Conway T (2005) Multiple regulators control expression of the Entner-Doudoroff aldolase (Eda) of Escherichia coli. J Bacteriol 187:991–1000. https://doi.org/10.1128/JB.187.3.991-1000.2005
Ogawa T, Mori H, Tomita M, Yoshino M (2007) Inhibitory effect of phosphoenolpyruvate on glycolytic enzymes in Escherichia coli. Res Microbiol 158:159–163. https://doi.org/10.1016/j.resmic.2006.11.003
Rao N, Torriani A (1990) Molecular aspects of phosphate transport in Escherichia coli. Mol Microbiol 4:1083–1090. https://doi.org/10.1111/j.1365-2958.1990.tb00682.x
Schuhmacher T, Löffler M, Hurler T, Takors R (2014) Phosphate limited fed-batch processes: impact on carbon usage and energy metabolism in Escherichia coli. J Bacteriol 190:96–104. https://doi.org/10.1016/j.jbiotec.2014.04.025
Shimizu K (2013) Metabolic regulation of a bacterial cell system with emphasis on Escherichia coli metabolism. ISRN Biochem 2013
Valgepea K, Adamberg K, Nahku R, Lahtvee P-J, Arike L, Vilu R (2010) Systems biology approach reveals that overflow metabolism of acetate in Escherichia coli is triggered by carbon catabolite repression of acetyl-CoA synthetase. BMC Syst Biol 4:166. https://doi.org/10.1186/1752-0509-4-166
Wang Y, Du P, Gan R, Li Z, Ye Q (2005) Fed-batch cultivation of Escherichia coli YK537 (pAET-8) for production ofphoA promoter-controlled human epidermal growth factor. Biotechnol Bioprocess Eng 10:149–154. https://doi.org/10.1007/BF02932585
Wanner B (1993) Gene regulation by phosphate in enteric bacteria. J Cell Biochem 51:47–54. https://doi.org/10.1002/jcb.240510110
Youngquist JT, Korosh TC, Pfleger BF (2017) Functional genomics analysis of free fatty acid production under continuous phosphate limiting conditions. J Ind Microbiol Biotechnol 44:759–772. https://doi.org/10.1007/s10295-016-1846-3
Zhang Y, Li Z, Ye Q (2010) Enhanced production of human epidermal growth factor under control of the phoA promoter by acetate-tolerant Escherichia coli DB15 in a chemically defined medium. Biotechnol Bioprocess Eng 15:626–634. https://doi.org/10.1007/s12257-009-3049-5
Zhao G, Winkler ME (1994) An Escherichia coli K-12 tktA tktB mutant deficient in transketolase activity requires pyridoxine (vitamin B6) as well as the aromatic amino acids and vitamins for growth. J Bacteriol 176:6134–6138. https://doi.org/10.1128/jb.176.19.6134-6138.1994
Funding
This work was supported by the Department of Biotechnology, Ministry of Science and Technology under schemes of the Centre of Excellence for Biopharmaceutical Technology (BT/COE/34/SP15097/2015).
Data availabilityThe data that support the findings of this study are available from the corresponding author upon reasonable request.
Author information
Authors and Affiliations
Contributions
KAP and ASR conceived and designed research. KAP and ASC conducted experiments. WHK contributed to gene expression analysis. KAP, ASC, and WHK wrote the first draft. ASR was responsible for project supervision, acquiring funding, and creating the final version of the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with humans or animals performed by any of the authors.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(PDF 217 kb)
Rights and permissions
About this article
Cite this article
Pandi, K., Chauhan, A.S., Khan, W.H. et al. Phosphate starvation controls lactose metabolism to produce recombinant protein in Escherichia coli. Appl Microbiol Biotechnol 104, 9707–9718 (2020). https://doi.org/10.1007/s00253-020-10935-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-10935-y