Abstract
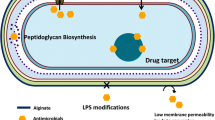
Pseudomonas aeruginosa infection is a significant threat for clinicians. Increasing incidents of resistant biofilm infection result in high mortality rates worldwide. There is a considerable current interest in the field of extracellular DNA (eDNA)–mediated P. aeruginosa biofilm formation. eDNA acts as a glue to make biofilm more stable. This review focuses on the diverse mechanisms and factors, which enhance the eDNA release into the extracellular milieu. Furthermore, eDNA-mediated molecular interactions within the biofilm are emphasized. In addition, drug resistance mechanisms due to the versatility of eDNA are discussed. Spatial physiological diversity is expected due to different metabolic activity of bacterial subpopulation present in P. aeruginosa biofilm layers. In P. aeruginosa, eDNA release is accomplished by cell lysis and OMVs (outer membrane vesicles). eDNA release is a spontaneous and multifactorial process, which may be accomplished by PQS, pyocyanin, and lambda prophage induction. Hydrogen peroxide and pyocin trigger cell death, which may facilitate eDNA release. Lung mucosa of cystic fibrosis patients is enriched with eDNA, which acidifies biofilm and develops P. aeruginosa resistance to aminoglycosides. Further studies on spatial and molecular characterization of bacterial subpopulation in biofilm will shed light on eDNA-biofilm interaction more precisely.
Key Points
• Extracellular DNA (eDNA) is a key component of Pseudomonas aeruginosa biofilm.
• P. aeruginosa eDNA acts as a glue to make biofilm more stronger.
• Bacterial cell death or lysis may be the potential way to release P. aeruginosa eDNA into extracellular milieu.
• P. aeruginosa eDNA contributes to develop resistance to antimicrobials.





Similar content being viewed by others
References
Alipour M, Suntres ZE, Omri A (2009) Importance of DNase and alginate lyase for enhancing free and liposome encapsulatedaminoglycoside activity against Pseudomonas aeruginosa. J Antimicrob Chemother 64:317–325
Alkawash MA, Soothill JS, Schiller NL (2006) Alginate lyase enhances antibiotic killing of mucoid Pseudomonas aeruginosa in biofilms. APMIS 114:131–138
Allesen-Holm M, Barken KB, Yang L, Klausen M, Webb JS, Kjelleberg S, Molin S, Givskov M, Tolker-Nielsen T (2006) Characterization of DNA release in Pseudomonas aeruginosa cultures and biofilms. Mol Microbiol 59:1114–1128
Allocati N, Masulli M, Di Ilio C, De Laurenzi V (2015) Die for the community: an overview of programmed cell death in bacteria. Cell Death Dis 6:e1609
Ascher J, Ceccherini MT, Pantani OL, Agnelli A, Borgogni F, Guerri G, Nannipieri P, Pietramellara G (2009) Sequential extraction and genetic fingerprinting of a forest soil metagenome. Appl Soil Ecol 42:176–181
Aspe M, Jensen L, Melegrito J, Sun M (2012) The role of alginate and extracellular DNA in biofilm-meditated Pseudomonas aeruginosa gentamicin resistance. J Exp Microbiol Immunol 16:42–48
Bahamondez-Canas TF, Heersema LA, Smyth HDC (2019) Current status of in vitro models and assays for susceptibility testing for wound biofilm infections. Biomedicines 7. https://doi.org/10.3390/biomedicines7020034
Banin E, Brady KM, Greenberg EP (2006) Chelator-induced dispersal and killing of Pseudomonas aeruginosa cells in a biofilm. Appl Environ Microbiol 72:2064–2069
Barken KB, Pamp SJ, Yang L, Gjermansen M, Bertrand JJ, Klausen M, Givskov M, Whitchurch CB, Engel JN, Tolker-Nielsen T (2008) Roles of type IV pili, flagellum-mediated motility and extracellular DNA in the formation of mature multicellular structures in Pseudomonas aeruginosa biofilms. Environ Microbiol 10:2331–2343
Bassetti M, Righi E, Viscoli C (2008) Pseudomonas aeruginosa serious infections: mono or combination antimicrobial therapy? Curr Med Chem 15:517–522
Beaumont PE, McHugh B, Gwyer Findlay E, Mackellar A, Mackenzie KJ, Gallo RL, Govan JR, Simpson AJ, Davidson DJ (2014) Cathelicidin host defence peptide augments clearance of pulmonary Pseudomonas aeruginosa infection by its influence on neutrophil function in vivo. PLoS One 9:e99029
Beringer PM, Vinks AA, Jelliffe RW, Shapiro BJ (2000) Pharmacokinetics of tobramycin in adults with cystic fibrosis: implications for once-daily administration. Antimicrob Agents Chemother 44:809–813
Binnenkade L, Teichmann L, Thormann KM (2014) Iron triggers λSo prophage induction and release of extracellular DNA in Shewanella oneidensisMR-1 biofilms. Appl Environ Microbiol 80:5304–5316
Bitto NJ, Chapman R, Pidot S, Costin A, Lo C, Choi J, D'Cruze T, Reynolds EC, Dashper SG, Turnbull L, Whitchurch CB, Stinear TP, Stacey KJ, Ferrero RL (2017) Bacterial membrane vesicles transport their DNA cargo into host cells. Sci Rep 7:7072
Blackledge MS, Worthington RJ, Melander C (2013) Biologically inspired strategies for combating bacterial biofilms. Curr Opin Pharmacol 13:699–706
Bomberger JM, Maceachran DP, Coutermarsh BA, Ye S, O'Toole GA, Stanton BA (2009) Long-distance delivery of bacterial virulence factors by Pseudomonas aeruginosa outer membrane vesicles. PLoS Pathog 5:e1000382
Brasseur R, Laurent G, Ruysschaert JM, Tulkens P (1984) Interactions of aminoglycoside antibiotics with negatively charged lipid layers. Biochemical and conformational studies. Biochem Pharmacol 33:629–637
Busscher HJ, Norde W, van der Mei HC (2008) Specific molecular recognition and nonspecific contributions to bacterial interaction forces. Appl Environ Microbiol 74:2559–2564
Carrolo M, Frias MJ, Pinto FR, Melo-Cristino J, Ramirez M (2010) Prophage spontaneous activation promotes DNA release enhancing biofilm formation in Streptococcus pneumoniae. PLoS One 5:e15678
Centers for Disease Control and Prevention (2013) Pseudomonas aeruginosa in healthcare settings. Healthcare-associated infections. https://www.cdc.gov/hai/organisms/pseudomonas.html
Centers for Disease Control and Prevention (2018) Biggest threats and data. Antibiotic / antimicrobial resistance (AR / AMR) https://www.cdc.gov/drugresistance/biggest_threats.html?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.gov%2Fdrugresistance%2Fthreat-report-2013%2Findex.html
Chen F, Chen G, Liu Y, Jin Y, Cheng Z, Liu Y, Yang L, Jin S, Wu W (2017) Pseudomonas aeruginosa oligoribonuclease contributes to tolerance to ciprofloxacin by regulating pyocin biosynthesis. Antimicrob Agents Chemother 61
Chiang WC, Nilsson M, Jensen PO, Hoiby N, Nielsen TE, Givskov M, Tolker-Nielsen T (2013) Extracellular DNA shields against aminoglycosides in Pseudomonas aeruginosa biofilms. Antimicrob Agents Chemother 57:2352–2361
Christie PJ (2001) Type IV secretion: intercellular transfer of macromolecules by systems ancestrally related to conjugation machines. Mol Microbiol 40:294–305
Ciofu O, Tolker-Nielsen T (2019) Tolerance and resistance of Pseudomonas aeruginosa biofilms to antimicrobial agents-how P. aeruginosa can escape antibiotics. Front Microbiol 10:913
Cirz RT, O'Neill BM, Hammond JA, Head SR, Romesberg FE (2006) Defining the Pseudomonas aeruginosa SOS response and its role in the global response to the antibiotic ciprofloxacin. J Bacteriol 188:7101–7110
Cooke AC, Nello AV, Ernst RK, Schertzer JW (2019) Analysis of Pseudomonas aeruginosa biofilm membrane vesicles supports multiple mechanisms of biogenesis. PLoS One 14:e0212275
Costerton JW, Stewart PS, Greenberg EP (1999) Bacterial biofilms: a common cause of persistent infections. Science 284:1318–1322
Das T, Manefield M (2012) Pyocyanin promotes extracellular DNA release in Pseudomonas aeruginosa. PLoS One 7:e46718
Das T, Sharma PK, Busscher HJ, van der Mei HC, Krom BP (2010) Role of extracellular DNA in initial bacterial adhesion and surface aggregation. Appl Environ Microbiol 76:3405–3408
Das T, Kutty SK, Kumar N, Manefield M (2013) Pyocyanin facilitates extracellular DNA binding to Pseudomonas aeruginosa influencing cell surface properties and aggregation. PLoS One 8:e58299
Das T, Sehar S, Koop L, Wong YK, Ahmed S, Siddiqui KS, Manefield M (2014) Influence of calcium in extracellular DNA mediated bacterial aggregation and biofilm formation. PLoS One 9:e91935
Dauros-Singorenko P, Blenkiron C, Phillips A, Swift S (2018) The functional RNA cargo of bacterial membrane vesicles. FEMS Microbiol Lett 365. https://doi.org/10.1093/femsle/fny023
Davies CB, Harrison MD, Huygens F (2017) Pseudomonas aeruginosa Trent and zinc homeostasis. FEMS Microbiol Lett 364. https://doi.org/10.1093/femsle/fnx151
Deatherage BL, Lara JC, Bergsbaken T, Rassoulian Barrett SL, Lara S, Cookson BT (2009) Biogenesis of bacterial membrane vesicles. Mol Microbiol 72:1395–1407
DeLeon S, Clinton A, Fowler H, Everett J, Horswill AR, Rumbaugh KP (2014) Synergistic interactions of Pseudomonas aeruginosa and Staphylococcus aureus in an in vitro wound model. Infect Immun 82:4718–4728
Deziel E, Lepine F, Milot S, He J, Mindrinos MN, Tompkins RG, Rahme LG (2004) Analysis of Pseudomonas aeruginosa 4-hydroxy-2-alkylquinolines (HAQs) reveals a role for 4-hydroxy-2-heptylquinoline in cell-to-cell communication. Proc Natl Acad Sci U S A 101:1339–1344
Domenech M, Garcia E (2018) Autolysin-independent DNA release in Streptococcus pneumoniae in vitro biofilms. bioRxiv. https://doi.org/10.1101/322818
Duport C, Baysse C, Michel-Briand Y (1995) Molecular characterization of pyocin S3, a novel S-type pyocin from Pseudomonas aeruginosa. J Biol Chem 270:8920–8927
Flemming HC, Neu TR, Wozniak DJ (2007) The EPS matrix: the “house of biofilm cells”. J Bacteriol 189:7945–7947
Folsom JP, Richards L, Pitts B, Roe F, Ehrlich GD, Parker A, Mazurie A, Stewart PS (2010) Physiology of Pseudomonas aeruginosa in biofilms as revealed by transcriptome analysis. BMC Microbiol 10:294
Frederiksen B, Pressler T, Hansen A, Koch C, Hoiby N (2006) Effect of aerosolized rhDNase (Pulmozyme) on pulmonary colonization in patients with cystic fibrosis. Acta Paediatr 95:1070–1074
Fuxman Bass JI, Gabelloni ML, Alvarez ME, Vermeulen ME, Russo DM, Zorreguieta A, Geffner JR, Trevani AS (2008) Characterization of bacterial DNA binding to human neutrophil surface. Lab Investig 88:926–937
Fuxman Bass JI, Russo DM, Gabelloni ML, Geffner JR, Giordano M, Catalano M, Zorreguieta A, Trevani AS (2010) Extracellular DNA: a major proinflammatory component of Pseudomonas aeruginosa biofilms. J Immunol 184:6386–6395
Ghafoor A, Hay ID, Rehm BH (2011) Role of exopolysaccharides in Pseudomonas aeruginosa biofilm formation and architecture. Appl Environ Microbiol 77:5238–5246
Ghoul M, West SA, Johansen HK, Molin S, Harrison OB, Maiden MCJ, Jelsbak L, Bruce JB, Griffin AS. (2015) Bacteriocin mediated competition in cystic fibrosis lung infections Proc Biol sci 282. https://doi.org/10.1098/rspb.2015.0972
Gi M, Lee KM, Kim SC, Yoon JH, Yoon SS, Choi JY (2015) A novel siderophore system is essential for the growth of Pseudomonas aeruginosa in airway mucus. Sci Rep 5:14644
Gloag ES, Turnbull L, Huang A, Vallotton P, Wang H, Nolan LM, Mililli L, Hunt C, Lu J, Osvath SR, Monahan LG, Cavaliere R, Charles IG, Wand MP, Gee ML, Prabhakar R, Whitchurch CB (2013) Self-organization of bacterial biofilms is facilitated by extracellular DNA. Proc Natl Acad Sci U S A 110:11541–11546
Grande R, Di Marcantonio MC, Robuffo I, Pompilio A, Celia C, Di Marzio L, Paolino D, Codagnone M, Muraro R, Stoodley P, Hall-Stoodley L, Mincione G (2015) Helicobacter pylori ATCC 43629/NCTC 11639 outer membrane vesicles (OMVs) from biofilm and planktonic phase associated with extracellular DNA (eDNA). Front Microbiol 6:1369
Gutu AD, Rodgers NS, Park J, Moskowitz SM (2015) Pseudomonas aeruginosa high-level resistance to polymyxins and other antimicrobial peptides requires cprA, a gene that is disrupted in the PAO1 strain. Antimicrob Agents Chemother 59:5377–5387
Hall CW, Mah TF (2017) Molecular mechanisms of biofilm-based antibiotic resistance and tolerance in pathogenic bacteria. FEMS Microbiol Rev 41:276–301
Halverson TW, Wilton M, Poon KK, Petri B, Lewenza S (2015) DNA is an antimicrobial component of neutrophil extracellular traps. PLoS Pathog 11:e1004593
Hamilton HL, Dominguez NM, Schwartz KJ, Hackett KT, Dillard JP (2005) Neisseria gonorrhoeae secretes chromosomal DNA via a novel type IV secretion system. Mol Microbiol 55:1704–1721
Haussler S, Becker T (2008) The pseudomonas quinolone signal (PQS) balances life and death in Pseudomonas aeruginosa populations. PLoS Pathog 4:e1000166
Hazan R, Sat B, Engelberg-Kulka H (2004) Escherichia coli mazEF-mediated cell death is triggered by various stressful conditions. J Bacteriol 186:3663–3669
Hazan R, Que YA, Maura D, Strobel B, Majcherczyk PA, Hopper LR, Wilbur DJ, Hreha TN, Barquera B, Rahme LG (2016) Auto poisoning of the respiratory chain by a quorum-sensing-regulated molecule favors biofilm formation and antibiotic tolerance. Curr Biol 26:195–206
Heacock-Kang Y, Sun Z, Zarzycki-Siek J, McMillan IA, Norris MH, Bluhm AP, Cabanas D, Fogen D, Vo H, Donachie SP, Borlee BR, Sibley CD, Lewenza S, Schurr MJ, Schweizer HP, Hoang TT (2017) Spatial transcriptomes within the Pseudomonas aeruginosa biofilm architecture. Mol Microbiol 106:976–985
Hentzer M, Teitzel GM, Balzer GJ, Heydorn A, Molin S, Givskov M (2001) Alginate overproduction affects Pseudomonas aeruginosa biofilm structure and function. J Bacteriol 183:5395–5401
Higgins S, Heeb S, Rampioni G, Fletcher MP, Williams P, Camara M (2018) Differential regulation of the phenazine biosynthetic operons by quorum sensing in Pseudomonas aeruginosa PAO1-N. Front Cell Infect Microbiol 8:252
Hoiby N, Ciofu O, Bjarnsholt T (2010) Pseudomonas aeruginosa biofilms in cystic fibrosis. Future Microbiol 5:1663–1674
Hotterbeekx A, Kumar-Singh S, Goossens H, Malhotra-Kumar S (2017) In vivo and in vitro interactions between Pseudomonas aeruginosa and Staphylococcus spp. Front Cell Infect Microbiol 7:106
Ibanez de Aldecoa AL, Zafra O, Gonzalez-Pastor JE (2017) Mechanisms and regulation of extracellular DNA release and its biological roles in microbial communities. Front Microbiol 8:1390
Irie Y, Roberts AEL, Kragh KN, Gordon VD, Hutchison J, Allen RJ, Melaugh G, Bjarnsholt T, West SA, Diggle SP (2017) The Pseudomonas aeruginosa PSL polysaccharide is a social but noncheatable trait in biofilms. MBio. https://doi.org/10.1128/mBio.00374-17
Jack AA, Khan S, Powell LC, Pritchard MF, Beck K, Sadh H, Sutton L, Cavaliere A, Florance H, Rye PD, Thomas DW, Hill KE (2018) Alginate oligosaccharide-induced modification of the lasI-lasR and rhlI-rhlR quorum-sensing systems in Pseudomonas aeruginosa. Antimicrob Agents Chemother. https://doi.org/10.1128/AAC.02318-17
Jakubovics NS, Shields RC, Rajarajan N, Burgess JG (2013) Life after death: the critical role of extracellular DNA in microbial biofilms. Lett Appl Microbiol 57:467–475
Jennings LK, Storek KM, Ledvina HE, Coulon C, Marmont LS, Sadovskaya I (2015) Pel is a cationic exopolysaccharide that cross-links extracellular DNA in the Pseudomonas aeruginosa biofilm matrix. Proc Natl Acad Sci U S A 112:11353–11358
Johnson L, Horsman SR, Charron-Mazenod L, Turnbull AL, Mulcahy H, Surette MG, Lewenza S (2013) Extracellular DNA-induced antimicrobial peptide resistance in Salmonella enterica serovar Typhimurium. BMC Microbiol 13:115
Jones CJ, Wozniak DJ (2017) Psl produced by mucoid Pseudomonas aeruginosa contributes to the establishment of biofilms and immune evasion. MBio
Jurcisek JA, Brockman KL, Novotny LA, Goodman SD, Bakaletz LO (2017) Nontypeable Haemophilus influenzae releases DNA and DNABII proteins via a T4SS-like complexand ComE of the type IV pilus machinery. Proc Natl Acad Sci U S A 114:E6632–E6641
Kovacha KN, Flemingb D, Rumbaughb KP, Gordon VD (2019) Specific disruption of established P. aeruginosa biofilms using polymer-attacking enzymes. bioRxiv. https://doi.org/10.1101/598979
Kreth J, Vu H, Zhang Y, Herzberg MC (2009) Characterization of hydrogen peroxide-induced DNA release by Streptococcus sanguinis and Streptococcus gordonii. J Bacteriol 191:6281–6291
Kuehn MJ, Kesty NC (2005) Bacterial outer membrane vesicles and the host-pathogen interaction. Genes Dev 19:2645–2655
Kulp A, Kuehn MJ (2010) Biological functions and biogenesis of secreted bacterial outer membrane vesicles. Ann Rev Microbiol 64:163–184
Kumar S, Kolodkin-Gal I, Engelberg-Kulka H (2013) Novel quorum-sensing peptides mediating interspecies bacterial cell death. MBio 4:e00314–e00313
Lamppa JW, Ackerman ME, Lai JI, Scanlon TC, Griswold KE (2011) Genetically engineered alginate lyase-PEG conjugates exhibit enhanced catalytic function and reduced immunoreactivity. PLoS One 6:e17042
Lee JY, Ko KS (2014) Mutations and expression of PmrAB and PhoPQ related with colistin resistance in Pseudomonas aeruginosa clinical isolates. Diagn Microb Infect Dis 78:271–276
Lee J, Zhang L (2015) The hierarchy quorum sensing network in Pseudomonas aeruginosa. Protein Cell 6:26–41
LeRoux M, Kirkpatrick RL, Montauti EI, Tran BQ, Peterson SB, Harding BN, Whitney JC, Russell AB, Traxler B, Goo YA, Goodlett DR, Wiggins PA, Mougous JD (2015) Kin cell lysis is a danger signal that activates antibacterial pathways of Pseudomonas aeruginosa. Elife 4:e05701
Lewenza S (2013) Extracellular DNA-induced antimicrobial peptide resistance mechanisms in Pseudomonas aeruginosa. Front Microbiol 4:21
Liao S, Klein MI, Heim KP, Fan Y, Bitoun JP, Ahn SJ, Burne RA, Koo H, Brady LJ, Wen ZT (2014) Streptococcus mutans extracellular DNA is upregulated during growth in biofilms, actively released via membrane vesicles, and influenced by components of the protein secretion machinery. J Bacteriol 196:2355–2366
Lin X, Han F, Yang Z, Lu X-z, Yu W-g (2006) A novel alginate lyase 844 with high activity on acetylated alginate of Pseudomonas aeruginosa FRD1 845 from Pseudomonas sp. QD03. World J Microbiol Biotechnol 22:81–88
Ma Q, Zhai Y, Schneider JC, Ramseier TM, Saier MH Jr (2003) Protein secretion systems of Pseudomonas aeruginosa and P fluorescens. Biochim Biophys Acta 1611:223–233
Ma L, Conover M, Lu H, Parsek MR, Bayles K, Wozniak DJ (2009) Assembly and development of the Pseudomonas aeruginosa biofilm matrix. PLoS Pathog 5:e1000354
Malhotra S, Limoli DH, English AE, Parsek MR, Wozniak DJ (2018) Mixed communities of mucoid and nonmucoid Pseudomonas aeruginosa exhibit enhanced resistance to host antimicrobials. MBio
Mann EE, Wozniak DJ (2012) Pseudomonas biofilm matrix composition and niche biology. FEMS Microbiol Rev 36:893–916
Martinez-Aleman SR, Campos-Garcia L, Palma-Nicolas JP, Hernandez-Bello R, Gonzalez GM, Sanchez-Gonzalez A (2017) Understanding the entanglement: neutrophil extracellular traps (NETs) in cystic fibrosis. Fron Cell Infect Microbiol 7:104
Mashburn LM, Whiteley M (2005) Membrane vesicles traffic signals and facilitate group activities in a prokaryote. Nature 437:422–425
Mavrodi DV, Bonsall RF, Delaney SM, Soule MJ, Phillips G, Thomashow LS (2001) Functional analysis of genes for biosynthesis of pyocyanin and phenazine-1-carboxamide from Pseudomonas aeruginosa PAO1. J Bacteriol 183:6454–6465
McFarland KA, Dolben EL, LeRoux M, Kambara TK, Ramsey KM, Kirkpatrick RL, Mougous JD, Hogan DA, Dove SL (2015) A self-lysis pathway that enhances the virulence of a pathogenic bacterium. Proc Natl Acad Sci U S A 112:8433–8438
Michel-Briand Y, Baysse C (2002) The pyocins of Pseudomonas aeruginosa. Biochimie 84:499–510
Miller AK, Brannon MK, Stevens L, Johansen HK, Selgrade SE, Miller SI, Hoiby N, Moskowitz SM (2011) PhoQ mutations promote lipid A modification and polymyxin resistance of Pseudomonas aeruginosa found in colistin-treated cystic fibrosis patients. Antimicrob Agents Chemother 55:5761–5769
Minandri F, Imperi F, Frangipani E, Bonchi C, Visaggio D, Facchini M, Pasquali P, Bragonzi A, Visca P (2016) Role of iron uptake systems in Pseudomonas aeruginosa virulence and airway infection. Infect Immun 84:2324–2335
Mishra M, Byrd MS, Sergeant S, Azad AK, Parsek MR, McPhail L, Schlesinger LS, Wozniak DJ (2012) Pseudomonas aeruginosa Psl polysaccharide reduces neutrophil phagocytosis and the oxidativeresponse by limiting complement-mediated opsonization. Cell Microbiol 14:95–106
Mo CY, Manning SA, Roggiani M, Culyba MJ, Samuels AN, Sniegowski PD, Goulian M, Kohli RM (2016) Systematically altering bacterial SOS activity under stress reveals therapeutic strategies for potentiating antibiotics. mSphere
Moradali MF, Ghods S, Rehm BH (2017) Pseudomonas aeruginosa lifestyle: a paradigm for adaptation, survival, and persistence. Front Cell Infect Microbiol 7:39
Moreau-Marquis S, O'Toole GA, Stanton BA (2009) Tobramycin and FDA-approved iron chelators eliminate Pseudomonas aeruginosa biofilms on cystic fibrosis cells. Am J Respir Cell Mol Biol 41:305–313
Mulcahy H, Charron-Mazenod L, Lewenza S (2008) Extracellular DNA chelates cations and induces antibiotic resistance in Pseudomonas aeruginosa biofilms. PLoS Pathog 4:e1000213
Nair HA, Periasamy S, Yang L, Kjelleberg S, Rice SA (2017) Real time, spatial, and temporal mapping of the distribution of c-di-GMP during biofilm development. J Biol Chem 292:477–487
Nakamura S, Higashiyama Y, Izumikawa K, Seki M, Kakeya H, Yamamoto Y, Yanagihara K, Miyazaki Y, Mizuta Y, Kohno S (2008) The roles of the quorum-sensing system in the release of extracellular DNA, lipopolysaccharide, and membrane vesicles from Pseudomonas aeruginosa. Jpn J Infect Dis 61:375–378
Nakao R, Myint SL, Wai SN, Uhlin BE (2018) Enhanced biofilm formation and membrane vesicle release by Escherichia coli expressing a commonly occurring plasmid gene, kil. Front Microbiol 9:2605
Nakayama K, Takashima K, Ishihara H, Shinomiya T, Kageyama M, Kanaya S, Ohnishi M, Murata T, Mori H, Hayashi T (2000) The R-type pyocin of Pseudomonas aeruginosa is related to P2 phage, and the F-type is related to lambda phage. Mol Microbiol 38:213–231
Nguyen AT, O'Neill MJ, Watts AM, Robson CL, Lamont IL, Wilks A, Oglesby-Sherrouse AG (2014) Adaptation of iron homeostasis pathways by a Pseudomonas aeruginosa pyoverdine mutant in the cystic fibrosis lung. J Bacteriol 196:2265–2276
Nichols DP, Durmowicz AG, Field A, Flume PA, VanDevanter DR, Mayer-Hamblett N (2019) Developing inhaled antibiotics in cystic fibrosis: current challenges and opportunities. Ann Am Thorac Soc 16:534–539
O'Brien S, Fothergill JL (2017) The role of multispecies social interactions in shaping Pseudomonas aeruginosa pathogenicity in the cystic fibrosis lung. FEMS Microbiol Lett 364. https://doi.org/10.1093/femsle/fnx128
O'Malley YQ, Reszka KJ, Spitz DR, Denning GM, Britigan BE (2004) Pseudomonas aeruginosa pyocyanin directly oxidizes glutathione and decreases its levels in airway epithelial cells. Am J Physiol Lung Cell Mol Physiol 287:L94–L103
Pallett R, Leslie LJ, Lambert PA, Milic I, Devitt A, Marshall LJ (2019) Anaerobiosis influences virulence properties of Pseudomonas aeruginosa cystic fibrosis isolatesand the interaction with Staphylococcus aureus. Sci Rep 9:6748
Palmgren R, Nielsen PH (1996) Accumulation of DNA in the exopolymeric matrix of activated sludge and bacterial cultures. Water Sci Technol 34:233–240
Penterman J, Singh PK, Walker GC (2014) Biological cost of pyocin production during the SOS response in Pseudomonas aeruginosa. J Bacteriol 196:3351–3359
Perez-Cruz C, Carrion O, Delgado L, Martinez G, Lopez-Iglesias C, Mercade E (2013) New type of outer membrane vesicle produced by the gram-negative bacterium Shewanella vesiculosa M7T: implications for DNA content. Appl Environ Microbiol 79:1874–1881
Petrova OE, Sauer K (2016) Escaping the biofilm in more than one way: desorption, detachment or dispersion. Curr Opin Microbiol 30:67–78
Price-Whelan A, Dietrich LEP, Newman DK (2006) Rethinking secondary metabolism: physiological roles for phenazine antibiotics. Nat Chem Biol 2:71–78
Puca V, Ercolino E, Celia C, Bologna G, Di Marzio L, Mincione G, Marchisio M, Miscia S, Muraro R, Lanuti P, Grande R (2019) Detection and quantification of eDNA-associated bacterial membrane vesicles by flow cytometry. Int J Mol Sci 20. https://doi.org/10.3390/ijms20215307
Qin Z, Ou Y, Yang L, Zhu Y, Tolker-Nielsen T, Molin S, Qu D (2007) Role of autolysin-mediated DNA release in biofilm formation of Staphylococcus epidermidis. Microbiology 153:2083–2092
Rasamiravaka T, Labtani Q, Duez P, El Jaziri M (2015) The formation of biofilms by Pseudomonas aeruginosa: a review of the natural and synthetic compounds interfering with control mechanisms. Biomed Res Int 2015:759348
Recinos DA, Sekedat MD, Hernandez A, Cohen TS, Sakhtah H, Prince AS, Price-Whelan A, Dietrich LE (2012) Redundant phenazine operons in Pseudomonas aeruginosa exhibit environment-dependent expression and differential roles in pathogenicity. Proc Natl Acad Sci U S A 109:19420–19425
Redero M, Lopez-Causape C, Aznar J, Oliver A, Blazquez J, Prieto AI (2018) Susceptibility to R-pyocins of Pseudomonas aeruginosa clinical isolates from cystic fibrosispatients. J Antimicrob Chemother 73:2770–2776
Reichhardt C, Parsek MR (2019) Confocal laser scanning microscopy for analysis of Pseudomonas aeruginosa biofilm architecture and matrix localization. Front Microbiol 10:677
Reid DW, Carroll V, O'May C, Champion A, Kirov SM (2007) Increased airway iron as a potential factor in the persistence of Pseudomonas aeruginosa infection in cystic fibrosis. Eur Respir J 30:286–292
Renelli M, Matias V, Lo RY, Beveridge TJ (2004) DNA-containing membrane vesicles of Pseudomonas aeruginosa PAO1 and their genetic transformation potential. Microbiology 150:2161–2169
Rice KC, Bayles KW (2008) Molecular control of bacterial death and lysis. Microbiol Mol Biol Rev 72:85–109
Rubin BK (2014) Secretion properties, clearance, and therapy in airway disease. Translat Respir Med 2:6
Sano Y, Kageyama M (1981) Purification and properties of an S-type pyocin, pyocin AP41. J Bacteriol 146:733–739
Savari M, Rostami S, Ekrami A, Bahador A (2016) Characterization of toxin-antitoxin (TA) systems in Pseudomonas aeruginosa clinical isolates in Iran. Jundishapur J Microbiol 9:e26627
Schurek KN, Sampaio JL, Kiffer CR, Sinto S, Mendes CM, Hancock RE (2009) Involvement of pmrAB and phoPQ in polymyxin B adaptation and inducible resistance in non-cystic fibrosis clinical isolates of Pseudomonas aeruginosa. Antimicrob Agents Chemother 53:4345–4351
Schurr MJ (2013) Which bacterial biofilm exopolysaccharide is preferred, Psl or alginate? J Bacteriol 195:1623–1626
Schuster M, Sexton DJ, Diggle SP, Greenberg EP (2013) Acyl-homoserine lactone quorum sensing: from evolution to application. Ann Rev Microbiol 67:43–63
Serra DO, Hengge R (2014) Stress responses go three dimensional - the spatial order of physiological differentiation in bacterial macrocolony biofilms. Environ Microbiol 16:1455–1471
Skopelja-Gardner S, Theprungsirikul J, Lewis KA, Hammond JH, Carlson KM, Hazlett HF, Nymon A, Nguyen D, Berwin BL, Hogan DA, Rigby WFC (2019) Regulation of Pseudomonas aeruginosa-mediated neutrophil extracellular traps. Front Immunol 10:1670
Smith K, Martin L, Rinaldi A, Rajendran R, Ramage G, Walker D (2012) Activity of pyocin S2 against Pseudomonas aeruginosa biofilms. Antimicrob Agents Chemother 56:1599–1601
Steichen CT, Cho C, Shao JQ, Apicella MA (2011) The Neisseria gonorrhoeae biofilm matrix contains DNA, and an endogenous nuclease controls its incorporation. Infect Immun 79:1504–15011
Tahrioui A, Duchesne R, Bouffartigues E, Rodrigues S, Maillot O, Tortuel D, Hardouin J, Taupin L, Groleau MC, Dufour A, Deziel E, Brenner-Weiss G, Feuilloley M, Orange N, Lesouhaitier O, Cornelis P, Chevalier S (2019) Extracellular DNA release, quorum sensing, and PrrF1/F2 small RNAs are key players in Pseudomonas aeruginosa tobramycin-enhanced biofilm formation. NPJ Biofilms Microbiomes 5:15
Tai KP, Kamdar K, Yamaki J, Le VV, Tran D, Tran P, Selsted ME, Ouellette AJ, Wong-Beringer A (2015) Microbicidal effects of alpha- and theta-defensins against antibiotic-resistant Staphylococcus aureus and Pseudomonas aeruginosa. Innate Immun 21:17–29
Tanaka M, Kurata T, Fujisawa C, Ohshima Y, Aoki H, Okazaki O, Hakusui H (1993) Mechanistic study of inhibition of levofloxacin absorption by aluminum hydroxide. Antimicrob Agents Chemother 37:2173–2178
Tang L, Schramm A, Neu TR, Revsbech NP, Meyer RL (2013) Extracellular DNA in adhesion and biofilm formation of four environmental isolates: a quantitative study. FEMS Microbiol Ecol 86:394–403
Tashiro Y, Takaki K, Futamata H (2019) Targeted delivery using membrane vesicles in prokaryotes. Biophys Physicobiol 16:114–120
Tetz VV, Tetz GV (2010) Effect of extracellular DNA destruction by DNase I on characteristics of forming biofilms. DNA Cell Biol 29:399–405
Thomas VC, Thurlow LR, Boyle D, Hancock LE (2008) Regulation of autolysis-dependent extracellular DNA release by Enterococcus faecalis extracellular proteases influences biofilm development. J Bacteriol 190:5690–5698
Trevani AS, Chorny A, Salamone G, Vermeulen M, Gamberale R, Schettini J, Raiden S, Geffner J (2003) Bacterial DNA activates human neutrophils by a CpG-independent pathway. Eur J Immunol 33:3164–3174
Tseng BS, Zhang W, Harrison JJ, Quach TP, Song JL, Penterman J, Singh PK, Chopp DL, Packman AI, Parsek MR (2013) The extracellular matrix protects Pseudomonas aeruginosa biofilms by limiting the penetration of tobramycin. Environ Microbiol 15:2865–2878
Turnbull L, Toyofuku M, Hynen AL, Kurosawa M, Pessi G, Petty NK, Osvath SR, Carcamo-Oyarce G, Gloag ES, Shimoni R, Omasits U, Ito S, Yap X, Monahan LG, Cavaliere R, Ahrens CH, Charles IG, Nomura N, Eberl L, Whitchurch CB (2016) Explosive cell lysis as a mechanism for the biogenesis of bacterial membrane vesicles and biofilms. Nat Commun 7:11220
Vipin C, Mujeeburahiman M, Saptami K, Arun AB, Rekha PD (2019) Synergistic interactions of quercetin with antibiotics against biofilm associated clinical isolates of Pseudomonas aeruginosa in vitro. bioRxiv. https://doi.org/10.1101/601336
Waksman G, Fronzes R (2010) Molecular architecture of bacterial type IV secretion systems. Trends Biochem Sci 35:691–698
Wang S, Liu X, Liu H, Zhang L, Guo Y, Yu S, Wozniak DJ, Ma LZ (2015) The exopolysaccharide Psl-eDNA interaction enables the formation of a biofilm skeleton in Pseudomonas aeruginosa. Environ Microbiol Rep 7:330–340
Watnick P, Kolter R (2000) Biofilm, city of microbes. J Bacteriol 182:2675–2679
Webb JS, Thompson LS, James S, Charlton T, Tolker-Nielsen T, Koch B, Givskov M, Kjelleberg S (2003) Cell death in Pseudomonas aeruginosa biofilm development. J Bacteriol 185:4585–4592
Wei Q, Ma LZ (2013) Biofilm matrix and its regulation in Pseudomonas aeruginosa. Int J Mol Sci 14:20983–21005
Weiss G, Carver PL (2018) Role of divalent metals in infectious disease susceptibility and outcome. Clin Microbiol Infect 24:16–23
Wessel AK, Liew J, Kwon T, Marcotte EM, Whiteley M (2013) Role of Pseudomonas aeruginosa peptidoglycan-associated outer membrane proteins in vesicle formation. J Bacteriol 195:213–219
Wessel AK, Arshad TA, Fitzpatrick M, Connell JL, Bonnecaze RT, Shear JB, Whiteley M (2014) Oxygen limitation within a bacterial aggregate. MBio 5:e00992
Whitchurch CB, Tolker-Nielsen T, Ragas PC, Mattick JS (2002) Extracellular DNA required for bacterial biofilm formation. Science 295:1487
Williamson KS, Richards LA, Perez-Osorio AC, Pitts B, McInnerney K, Stewart PS, Franklin MJ (2012) Heterogeneity in Pseudomonas aeruginosa biofilms includes expression of ribosome hibernationfactors in the antibiotic-tolerant subpopulation and hypoxia-induced stress response in the metabolically active population. J Bacteriol 194:2062–2073
Wilton M, Charron-Mazenod L, Moore R, Lewenza S (2015) Extracellular DNA acidifies biofilms and induces aminoglycoside resistance in Pseudomonas aeruginosa. Antimicrob Agents Chemother 60:544–553
Wilton M, Wong MJQ, Tang L, Liang X, Moore R, Parkins MD, Lewenza S, Dong TG (2016) Chelation of membrane-bound cations by extracellular DNA activates the type VI secretion system in Pseudomonas aeruginosa. Infect Immun 84:2355–2361
Wilton M, Halverson TWR, Charron-Mazenod L, Parkins MD, Lewenza S (2018) Secreted phosphatase and deoxyribonuclease are required by Pseudomonas aeruginosa to defend against neutrophil extracellular traps. Infect Immun. https://doi.org/10.1128/IAI.00403-18
Woith E, Fuhrmann G, Melzig MF (2019) Extracellular vesicles—Connecting kingdoms. Int J Mol Sci 20:5695
Wysoczynski-Horita CL, Boursier ME, Hill R, Hansen K, Blackwell HE, Churchill MEA (2018) Mechanism of agonism and antagonism of the Pseudomonas aeruginosa quorum sensing regulator QscR with non-native ligands. Mol Microbiol 108:240–257
Yan S, Wu G (2019) Can biofilm be reversed through quorum sensing in Pseudomonas aeruginosa? Front Microbiol 10:1582
Yang L, Barken KB, Skindersoe ME, Christensen AB, Givskov M, Tolker-Nielsen T (2007) Effects of iron on DNA release and biofilm development by Pseudomonas aeruginosa. Microbiology 153:1318–1328
Young RL, Malcolm KC, Kret JE, Caceres SM, Poch KR, Nichols DP, Taylor-Cousar JL, Saavedra MT, Randell SH, Vasil ML, Burns JL, Moskowitz SM, Nick JA (2011) Neutrophil extracellular trap (NET)-mediated killing of Pseudomonas aeruginosa: evidence of acquired resistance within the CF airway, independent of CFTR. PLoS One 6:e23637
Zafra O, Lamprecht-Grandio M, de Figueras CG, Gonzalez-Pastor JE (2012) Extracellular DNA release by undomesticated Bacillus subtilis is regulated by early competence. PLoS One 7:e48716
Zemke AC, Bomberger JM (2016) Microbiology: social suicide for a good cause. Curr Biol 26:R80–R82
Zhang W, Sun J, Ding W, Lin J, Tian R, Lu L, Liu X, Shen X, Qian PY (2015) Extracellular matrix-associated proteins form an integral and dynamic system during Pseudomonas aeruginosa biofilm development. Front Cell Infect Microbiol 5:40
Zweig M, Schork S, Koerdt A, Siewering K, Sternberg C, Thormann K, Albers SV, Molin S, van der Does C (2014) Secreted single-stranded DNA is involved in the initial phase of biofilm formation by Neisseria gonorrhoeae. Environ Microbiol 16:1040–1052
Acknowledgments
I would like to express my sincere gratitude to Prof. Chandan K. Sen, Indiana Center for Regenerative Medicine and Engineering, University School of Medicine, Department of Surgery, Indiana University, Indianapolis, IN 46202, USA; and Prof. Daniel Wozniak, Departments of Microbial Infection and Immunity, Microbiology, Center for Microbial Interface Biology, The Ohio State University, Columbus, OH 43210, USA.
Author information
Authors and Affiliations
Contributions
SS conceived and designed the research. SS analyzed the data and wrote the manuscript.
Corresponding author
Ethics declarations
Ethical statement
This study does not contain any work related to human participants or animals.
Conflict of interest
The author declares that there is no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Sarkar, S. Release mechanisms and molecular interactions of Pseudomonas aeruginosa extracellular DNA. Appl Microbiol Biotechnol 104, 6549–6564 (2020). https://doi.org/10.1007/s00253-020-10687-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-020-10687-9