Abstract
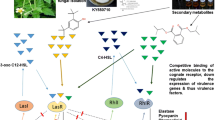
The increasing occurrence of resistance among Pseudomonas aeruginosa clinical isolates necessitates finding alternatives to antibiotics for controlling the infection of such pathogenic bacteria. In this study, lactonase gene ahl-1 from Bacillus weihenstephanensis isolate-P65 was successfully cloned and expressed in Escherichia coli BL21 (DE3) under the control of T7 promoter for utilizing its quorum quenching activity against three multidrug-resistant (MDR) P. aeruginosa clinical isolates. The biological activity of the overexpressed lactonase enzyme (Ahl-1), tested using a synthetic signal and Chromobacterium violaceum CV026 as a biosensor, displayed good catalytic activity using hexanoyl homoserine lactone (HHL) as a substrate and Chromobacterium violaceum (CV026) as a biosensor (77.2 and 133 nm min−1 for the crude and the purified Ahl-lactonase enzymes, respectively). Upon challenging its ability to inhibit the virulence of three MDR P. aeruginosa clinical isolates, recombinant Ahl-1 successfully prevented the accumulation of acylhomoserine lactone signals resulting in a significant reduction in the investigated virulence determinants; protease (from 40 up to 75.5%), pyocyanin (48–75.9%), and rhamnolipids (52.7–63.4%) (P value < 0.05). Ahl-1 also displayed significant inhibitory activities on the swarming motility and biofilm formation of the three tested MDR P. aeruginosa clinical isolates (P value < 0.05). Consequently, Ahl-1 lactonase enzyme in this study is considered a promising therapeutic agent to inhibit P. aeruginosa pathogenicity with no fear of emergence of resistance.







Similar content being viewed by others
References
Aybey A, Demirkan E (2016) Inhibition of quorum sensing-controlled virulence factors in Pseudomonas aeruginosa by human serum paraoxonase. J Med Microbiol 65:105–113. https://doi.org/10.1099/jmm.0.000206
Cao Y, He S, Zhou Z, Zhang M, Mao W, Zhang H, Yaoa B (2012) Orally administered thermostable N-acyl homoserine lactonase from Bacillus sp. strain AI96 attenuates Aeromonas hydrophila infection in zebrafish. Appl Environ Microbiol 78:1899–1908. https://doi.org/10.1128/AEM.06139-11
Chastre J, Fagon JY (2002) Ventilator-associated pneumonia. Am J Respir Crit Care Med 165:867–903. https://doi.org/10.1164/ajrccm.165.7.2105078
Christiaen SE, Matthijs N, Zhang XH, Nelis HJ, Bossier P, Coenye T (2014) Bacteria that inhibit quorum sensing decrease biofilm formation, virulence in Pseudomonas aeruginosa PAO1. Pathog Dis 70:271–279. https://doi.org/10.1111/2049-632X.12124
Dong YH, Wang LH, Xu JL, Zhang HB, Zhang XF, Zhang LH (2001) Quenching quorum-sensing-dependent bacterial infection by an N-acyl homoserine lactonase. Nature 411:813–817. https://doi.org/10.1038/35081101
Essar DW, Eberly L (1990) Evolutionary differences in chromosomal locations of four early genes of tryptophan pathway in fluorescent Pseudomonas: DNA sequences and characterization of Pseudomonas putida trpE and trpGDC. J Bacteriol 172:876–883
Giamarellou H (2002) Prescribing guidelines for severe Pseudomonas infections. J Antimicrob Chemother 49:229–233
Guendouze A, Plener L, Bzdrenga J, Pauline J, Rémy B, Elias M, Lavigne J, Daudé D, Chabrière E (2017) Effect of quorum quenching lactonase in clinical isolates of Pseudomonas aeruginosa and comparison with quorum sensing inhibitors. Front Microbiol 8:227. https://doi.org/10.3389/fmicb.2017.00227
Hanahan D (1983) Studies on transformation of Escherichia coli with plasmids. J Mol Biol 166:557–580
Hancock RE (1998) Resistance mechanisms in Pseudomonas aeruginosa and other nonfermentative gram-negative bacteria. Clin Infect Dis 27:93–99
Leadbetter JR (2001) Quieting the raucous crowd. Nature 411:748–749. https://doi.org/10.1038/35081216
Lee J, Zhang L (2015) The hierarchy quorum sensing network in Pseudomonas aeruginosa. Protein Cell 6(1):26–41. https://doi.org/10.1007/s13238-014-0100-x
Loeffl J, Stevens DA (2003) Antifungal drug resistance. Clin Infect Dis 36:31–41. https://doi.org/10.1086/344658
Lowry H, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the folin phenol reagent. J Biol Chem 193:265–275
Maschmeyer G, Braveny I (2000) Review of the incidence and prognosis of Pseudomonas aeruginosa infections in cancer patients in the 1990s. Eur J Clin Microbiol Infect Dis 19:915–925
Mesaros N, Nordmann P, Ple’siat P, Roussel-Delvallez M, Eldere JV, Glupczynski Y, Laethem YV, Jacobs F, Lebecque P, Malfroot A, Tulkens PM, Bambeke FV (2007) Pseudomonas aeruginosa: resistance and therapeutic options at the turn of the new millennium. Clin Microbiol Infect 13(6):560–578. https://doi.org/10.1111/j.1469-0691.2007.01681.x
Morens DM, Folkers GK, Fauci AS (2004) The challenge of emerging and re-emerging infectious diseases. Nature 430:242–249. https://doi.org/10.1038/nature02759
Morikawa M, Hirata Y, Imanaka T (2000) A study on the structure-function relationship of lipopeptide biosurfactants. Biochim Biophys Acta 1488(3):211–218
Nicodème M, Grill J, JL G (2005) Extracellular protease activity of different Pseudomonas strains: dependence of proteolytic activity on culture conditions. J Appl Microbiol 99(3):641–648. https://doi.org/10.1111/j.1365-2672.2005.02634.x
Obritsch MD, Fish DN, MacLaren R, Jung R (2005) Nosocomial infections due to MDR Pseudomonas aeruginosa: epidemiology and treatment options. Pharmacotherapy 25:1353–1364. https://doi.org/10.1592/phco.2005.25.10.1353
Osama D, Elkhatib W, Tawfeik A, Aboulwafa M, Hassouna N (2017) Antimicrobial, antibiofilm and immunomodulatory activities of Lactobacillus rhamnosus and Lactobacillus gasseri against some bacterial pathogens. Int J Biotechnol Wellness Ind 6(1):12–21. https://doi.org/10.6000/1927-3037.2017.06.01.2
Overhage J, Bains M, Brazas MD, Hancock RE (2008) Swarming of Pseudomonas aeruginosa is a complex adaptation leading to increased production of virulence factors and antibiotic resistance. J Bacteriol 190(8):2671–2679. https://doi.org/10.1128/JB.01659-07
Rabin N, Zheng Y, Opoku-Temeng C, Du Y, Bonsu E, Sintim HO (2015) Agents that inhibit bacterial biofilm formation. Future Med Chem 7(5):647–671. https://doi.org/10.4155/fmc.15.7
Rashid MH, Kornberg A (2000) Inorganic polyphosphate is needed for swimming, swarming, and twitching motilities of Pseudomonas aeruginosa. Proc Natl Acad Sci U S A 97(9):4885–4890. https://doi.org/10.1073/pnas.060030097
Ravn L, Christensen AB, Molin S, Givskov M, Gram L (2001) Methods for detecting acylated homoserine lactones produced by gram-negative bacteria and their application in studies of AHL-production kinetics. J Microbiol Methods 44(3):239–251
Sakr MM, Aboshanab K, Aboulwafa MM, Hassouna NA (2013) Characterization and complete sequence of lactonase enzyme from Bacillus weihenstephanensis isolate P65 with potential activity against acyl homoserine lactone signal molecules. Biomed Res Int 2013:1–10. https://doi.org/10.1155/2013/192589
Sakr MM, Aboulwafa MM, Aboshanab K, Hassouna NA (2014) Screening and preliminary characterization of the quorum quenching activity of some Bacillus isolates from soil against clinically isolated Pseudomonas aeruginosa homoserine lactones. Malays J Microbiol 10(2):80–91
Sambrook J (2000) SDS—plyacrylamide gel electrophoresis of proteins. Cold Harbour Press, Cold harbour
Sambrook J, Russell DW (2001) The Inoue method for preparation and transformation of competent E. coli: “ultra-competent” cells. Cold Spring Harbor Laboratory Press, Cold Spring Harbor
Schuster M, Greenberg EP (2006) A network of networks: quorum-sensing gene regulation in Pseudomonas aeruginosa. J Med Microbiol 296:73–81. https://doi.org/10.1016/j.ijmm.2006.01.036
Sio CF, Otten LG, Cool RH, Diggle SP, Braun PG, Bos R, Daykin M, Camara M, Williams P, Quax WJ (2006) Quorum quenching by an N-acyl-homoserine lactone acylase from Pseudomonas aeruginosa PAO1. Infect Immun 74(3):1673–1682. https://doi.org/10.1128/IAI.74.3.1673-1682.2006
Sligl W, Taylor G, Brindley PG (2006) Five years of nosocomial gram-negative bacteremia in a general intensive care unit: epidemiology, antimicrobial susceptibility patterns, and outcomes. Int J Infect Dis 10:320–325. https://doi.org/10.1016/j.ijid.2005.07.003
Stefani S, Campana S, Cariani L, Carnovale V, Colombo C, Lleo M, Lula V, Minicucci L, Morelli P, Pizzamiglio G, Taccetti G (2017) Relevance of multidrug-resistant Pseudomonas aeruginosa infections in cystic fibrosis. Int J Med Microbiol 307(6):353–362. https://doi.org/10.1016/j.ijmm.2017.07.004
Stepanovic S, Vukovic D, Hola V, Bonaventura GD, Djukic S, Cirkovic C, Ruzicka F (2007) Quantification of biofilm in microtiter plates: overview of testing conditions and practical recommendations for assessment of biofilm production by Staphylococci. APMIS 115(8):891–899. https://doi.org/10.1111/j.1600-0463.2007.apm_630.x
Studier F, Rosenberg A, Dunn J, Dubendorff J (1990) Use of bacteriophage T7 RNA polymerase to direct selective high-level expression of cloned genes. Methods Enzymol 185:60–89
Torres M, Uroz S, Salto R, Fauchery L, Quesada E, Llamas I (2017) HqiA, a novel quorum-quenching enzyme which expands the AHL lactonase family. Sci Rep 7:943. https://doi.org/10.1038/s41598-017-01176-7
Vaisvila R, Morgan RD, Posfai J, Raleigh EA (2001) Discovery and distribution of super-integrons among pseudomonads. Mol Microbiol 42:587–601
Viretta AU, Fussenegger M (2004) Modeling the quorum sensing regulatory network of human-pathogenic Pseudomonas aeruginosa. Biotechnol Prog 20(3):670–678. https://doi.org/10.1021/bp034323l
Wang Y, Yue D, Yong Z, YangBo H, BaoYu Y, ShiYun C (2007) Effects of quorum sensing autoinducer degradation gene on virulence and biofilm formation of Pseudomonas aeruginosa. Sci China C Life Sci 50(3):385–391. https://doi.org/10.1007/s11427-007-0044-y
WHO (2017) Global priority list of antibiotic-resistant bacteria to guide research, discovery and development of new antibiotics. WHO Essential medicines and health products1–7
Acknowledgements
We hereby acknowledge Department of Microbiology and Immunology, Faculty of Pharmacy, Ain Shams University, for providing us with all facilities and support required to perform the practical work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Sakr, M.M., Aboshanab, K.M., Elkhatib, W.F. et al. Overexpressed recombinant quorum quenching lactonase reduces the virulence, motility and biofilm formation of multidrug-resistant Pseudomonas aeruginosa clinical isolates. Appl Microbiol Biotechnol 102, 10613–10622 (2018). https://doi.org/10.1007/s00253-018-9418-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-018-9418-2