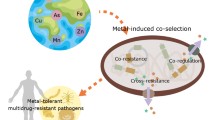
Abstract
Here, we review the possible reasons responsible for the occurrence, maintenance and proliferation of antibiotic resistance genes (ARGs) in the environment, as well as the corresponding mechanisms of their development, diffusion and transfer. Additionally, elimination strategies are also discussed. The factors that influence the development of ARGs are selection pressure, including that from antibiotics, metal and multiple other factors, co-resistance and cross-resistance, microbial consortium structure, nutrients in the environment and oxidative stress responses. Process parameters, transport pathways, and elimination strategies to reduce the health risk caused by ARGs are also reviewed in detail. Moreover, knowledge gaps and future opportunities of ARGs are addressed.
Similar content being viewed by others
References
Allen HK, Donato J, Wang HH, Cloud-Hansen KA, Davies J, Handelsman J (2010) Call of the wild: antibiotics resistance genes in natural environments. Nat Rev Microbiol 8(4):251–259. https://doi.org/10.1038/nrmicro2312
Aminov RI (2011) Horizontal gene exchange in environmental microbiota. Front Microbiol 2(158):1–19. https://doi.org/10.3389/fmicb.2011.00158
Amos GC, Gozzard E, Carter CE, Mead A, Bowes M, Hawkey PM, Zhang LH, Singer AC, Gaze WH, Wellington EMH (2015) Validated predictive modelling of the environmental resistome. ISME J 9(6):1467–1476. https://doi.org/10.1038/ismej.2014.237
Andersson DI (2006) The biological cost of mutational antibiotics resistance: any practical conclusions? Curr Opin Microbiol 9(5):461–465. https://doi.org/10.1016/j.mib.2006.07.002
Andersson DI, Hughes D (2010) Antibiotics resistance and its cost: is it possible to reverse resistance? Nat Rev Microbiol 8(4):260–271. https://doi.org/10.1016/j.mib.2006.07.002
Awad YM, Kim SC, Abd El-Azeem SM, Kim KH, Kim KR, Kim KJ, Jeon C, Lee SS, Ok YS (2014) Veterinary antibiotics contamination in water, sediment, and soil near a swine manure composting facility. Environ Earth Sci 71(3):1433–1440. https://doi.org/10.1007/s12665-013-2548-z
Baharoglu Z, Mazel D (2011) Vibrio cholera triggers SOS and mutugenesis in response to a wide range of antibiotics: a route towards multiresistance. Antimicrob Agents Chemother 55(5):2438–2441. https://doi.org/10.1128/AAC.01549-10
Baker-Austin C, Wright MS, Stepanauskas R, McArthur JV (2006) Co-selection of antibiotics and metal resistance. Trends Microbiol 14(4):176–182. https://doi.org/10.1016/j.tim.2006.02.006
Beaber JW, Hochhut B, Waldor MK (2004) SOS response promotes horizontal dissemination of antibiotics resistance genes. Nature 427(6969):72–74. https://doi.org/10.1038/nature02241
Ben WW, Wang J, Pan X, Qiang ZM (2017a) Dissemination of antibiotics resistance genes and their potential removal by on-farm treatment processes in nine swine feedlots in Shandong Province, China. Chemosphere 167:262–268. https://doi.org/10.1016/j.chemosphere.2016.10.013
Ben WW, Wang J, Cao RK, Yang M, Zhang Y, Qiang ZM (2017b) Distribution of antibiotics resistance in the effluents of ten municipal wastewater treatment plants in China and the effect of treatment processes. Chemosphere 172:392–398. https://doi.org/10.1016/j.chemosphere.2017.01.041
Berg J, Tom-Petersen A, Nybroe O (2005) Copper amendment of agricultural soil selects for bacterial antibiotics resistance in the field. Lett Appl Microbiol 40(2):146–151. https://doi.org/10.1111/j.1472-765X.2004.01650.x
Berg J, Thorsen MK, Holm PE, Jensen J, Nybroe O, Brandt KK (2010) Cu exposure under field conditions co-selects for antibiotics resistance as determined by a novel cultivation-independent bacterial community tolerance assay. Environ Sci Technol 44(22):8724–8728. https://doi.org/10.1021/es101798r
Bouzada MLM, Silva VL, Moreira FAS, Silva GA, Diniz CG (2010) Antimicrobial resistance and disinfectants susceptibility of persistent bacteria in a tertiary care hospital. J Microbiol Antimicrob 2:105–112 http://www.academicjournals.org/JMA
Brandt KK, Amézquita A, Backhaus T, Boxall A, Coors A, Heberer T, Lawrence JR, Lazorchak J, Schönfeld J, Snape JR, Zhu YG, Topp E (2015) Ecotoxicological assessment of antibioticss: a call for improved consideration of microorganisms. Environ Int 85:189–205. https://doi.org/10.1016/j.envint.2015.09.013
Cesare A, Eckert EM, Coron G (2016a) Co-selection of antibiotics and heavy metal resistance in freshwater bacteria. J Limnol 75(s2):59–66. https://doi.org/10.4081/jlimnol.2016.1198
Cesare A, Eckert EM, D'Urso S, Bertoni R, Gillan DC, Wattiez R, Corno G (2016b) Co-occurrence of integrase 1, antibiotics and heavy metal resistance genes in municipal wastewater treatment plants. Water Res 94:208–214. https://doi.org/10.1016/j.watres.2016.02.049
Chen J, Michel FC, Sreevatsan S, Morrison M, Yu Z (2010) Occurrence and persistence of erythromycin resistance genes (erm) and tetracycline resistance genes (tet) in waste treatment systems on swine farms. Microb Ecol 60(3):479–486. https://doi.org/10.1007/s00248-010-9634-5
Chen B, Yang Y, Liang X, Yu K, Zhang T, Li X (2013) Metagenomic profiles of antibiotics resistance genes (ARGs) between human impacted estuary and deep ocean sediments. Environ Sci Technol 47(22):12753–12760. https://doi.org/10.1021/es403818e
Chen QL, An XL, Li H, Su JQ, Ma YB, Zhu YG (2016) Long-term field application of sewage sludge increases the abundance of antibiotics resistance genes in soil. Environ Int 92-93:1–10. https://doi.org/10.1016/j.envint.2016.03.026
Cheng WX, Li JN, Wu Y, Xu LK, Su C, Qian YY, Zhu YG, Chen H (2016) Behavior of antibiotics and antibiotics resistance genes in eco-agricultural system: a case study. J Hazard Mater 304:18–25. https://doi.org/10.1016/j.jhazmat.2015.10.037
Codina JC, Cazorla FM, Perez-Garcia A, de Vicente A (2000) Heavy metal toxicity and genotoxicity in water and sewage determined by microbiological methods. Environ Toxicol Chem 19(6):1552–1558. https://doi.org/10.1002/etc.5620190611
Crecchio C, Ruggiero P, Curci M, Colombo C, Palumbo G, Stotzky G (2005) Binding of DNA from Bacillus subtilis on montmorillonite-humic acids-aluminum or iron hydroxypolymers: effects on transformation and protection against DNase. Soil Sci Soc Am J 69(3):834–884. https://doi.org/10.2136/sssaj2004.0166
Czekalski N, Imminger S, Salhi E, Veljkovic M, Kleffel K, Drissner D, Hammes F, Bürgmann H, von Gunten U (2016) Inactivation of antibiotics resistant bacteria and resistance genes by ozone: from laboratory experiments to full-scale wastewater treatment. Environ Sci Technol 50(21):11862–11871. https://doi.org/10.1021/acs.est.6b02640
Diehl D, Lapara TM (2010) Effect of temperature on the fate of genes encoding tetracycline resistance and the integrase of class 1 Integrons within anaerobic and aerobic digesters treating municipal wastewater solids. Environ Sci Technol 44(23):9128–9133. https://doi.org/10.1021/es102765a
Duan ML, Li HC, Gu J, Tuo XX, Sun W, Qian X, Wan XJ (2017) Effects of biochar on reducing the abundance of oxytetracycline, antibiotics resistance genes, and human pathogenic bacteria in soil and lettuce. Environ Pollut 224:787–795. https://doi.org/10.1016/j.envpol.2017.01.021
Fang HS, Zhang Q, Nie XP, Chen BW, Xiao YD, Zhou QB, Liao W, Liang XM (2017) Occurrence and elimination of antibiotics resistance genes in a long term operation integrated surface flow constructed wetland. Chemosphere 173:99–106. https://doi.org/10.1016/j.chemosphere.2017.01.027
Ferro G, Guarino F, Cicatelli A, Rizzo L (2017) β-Lactams resistance gene quantification in an antibiotics resistant Escherichia coli water suspension treated by advanced oxidation with UV/H2O2. J Hazard Mater 323(5):426–433. https://doi.org/10.1016/j.jhazmat.2016.03.014
Forsberg KJ, Patel S, Gibson MK, Lauber CL, Knight R, Fierer N, Dantas G (2014) Bacterial phylogeny structures soil resistomes across habitats. Nature 509(7502):612–616. https://doi.org/10.1038/nature13377
Gao P, Gu CC, Wei X, Li X, Chen H, Jia HZ, Liu ZL, Xue G, Ma CY (2017) The role of zero valent iron on the fate of tetracycline resistance genes and class 1 integrons during thermophilic anaerobic co-digestion of waste sludge and kitchen waste. Water Res 111:92–99. https://doi.org/10.1016/j.watres.2016.12.047
Ghosh S, LaPara TM (2007) The effects of sub-therapeutic antibiotics use in farm animals on the proliferation and persistence of antibiotics resistance among soil bacteria. ISME J 1(3):191–203. https://doi.org/10.1038/ismej.2007.31
Ghosh S, Ramsden SJ, LaPara TM (2009) The role of anaerobic digestion in controlling the release of tetracycline resistance genes and class 1 integrons from municipal wastewater treatment plants. Appl Microbiol Biotechnol 84(4):791–796. https://doi.org/10.1007/s00253-009-2125-2
Gillings M, Boucher Y, Labbate M, Holmes A, Krishnan S, Holley M, Stokes HW (2008) The evolution of class 1 integrons and the rise of antibiotics resistance. J Bacteriol 190(14):5095–5100. https://doi.org/10.1128/JB.00152-08
Gillings MR, Gaze WH, Smalla K, Tiedje JM Zhu YG (2015) Using the class 1 integron-integrase gene as a proxy for anthropogenic pollution. ISME J 9(6):1269–1279. https://doi.org/10.1038/ismej.2014.226
Guerin É, Cambray G, Sanchez-Alberola N, Campoy S, Erill I, Da Re S, Gonzalez-Zorn B, Barbé J, Ploy MC, Mazel D (2009) The SOS response controls integron recombination. Science 324(5930):1034–1034. https://doi.org/10.1126/science.1172914
Guo XP, Li J, Yang F, Yang J, Yin DQ (2014) Prevalence of sulfonamide and tetracycline resistance genes in drinking water treatment plants in the Yangtze River Delta, China. Sci Total Environ 493:626–631. https://doi.org/10.1016/j.scitotenv.2014.06.035
Guo MT, Yuan QB, Yang J (2015) Insights into the amplification of bacterial resistance to erythromycin in activated sludge. Chemosphere 136:79–85. https://doi.org/10.1016/j.chemosphere.2015.03.085
Guo JH, Li J, Chen H, Bond P, Yuan ZG (2017) Metagenomic analysis reveals wastewater treatment plants as hotspots of antibiotics resistance genes and mobile genetic elements. Water Res 123:467–478. https://doi.org/10.1016/j.watres.2017.07.002
Hall AR, Colegrave N (2008) Decay of unused characters by selection and drift. J Evol Biol 21(2):610–617. https://doi.org/10.1111/j.1420-9101.2007.01473
He LY, Liu YS, Su HC, Zhao JL, Liu SS, Chen J, Liu WR, Ying GG (2014) Dissemination of antibiotics resistance genes in representative broiler feedlots environments: identification of indicator ARGs and correlations with environmental variables. Environ Sci Technol 48(22):13120–13129. https://doi.org/10.1021/es5041267
He LY, Ying GG, Liu YS, Su HC, Chen J, Liu SS, Zhao JL (2016) Discharge of swine wastes risks water quality and food safety: antibiotics and antibiotics resistance genes from swine sources to the receiving environments. Environ Int 92-93:210–219. https://doi.org/10.1016/j.envint.2016.03.023
Heuer H, Schmitt H, Smalla K (2011) Antibiotics resistance gene spread due to manure application on agricultural fields. Curr Opin Microbiol 14(3):236–243. https://doi.org/10.1016/j.mib.2011.04.009
Hu HW, Wang JT, Li J, Shi XZ, Ma YB, Chen D, He JZ (2016a) Field-based evidence for copper contamination induced changes of antibiotics resistance in agricultural soils. Environ Microbiol 18(11):3896–3909. https://doi.org/10.1111/1462-2920.13370
Hu Q, Zhang XX, Jia SY, Huang KL, Tang JY, Shi P, Ye L, Ren HQ (2016b) Metagenomic insights into ultraviolet disinfection effects on antibiotics resistome in biologically treated wastewater. Water Res 101:309–317. https://doi.org/10.1016/j.watres.2016.05.092
Hu HW, Wang JT, Li J, Shi XZ, Ma YB, Chen DL, He JZ (2017) Long-term nickel contamination increases the occurrence of antibiotics resistance genes in agricultural soils. Environ Sci Technol 51(2):790–800. https://doi.org/10.1021/acs.est.6b03383
Huang MH, Zhang W, Zheng Y, Zhang W (2014) Correlation among extracellular polymeric substances, tetracycline resistant bacteria and tetracycline resistance genes under trace tetracycline. Chemosphere 117(117C):658–662. https://doi.org/10.1016/j.chemosphere.2014.09.078
Huang X, Liu C, Li K, Su J, Zhu G, Liu L (2015) Performance of vertical up-flow constructed wetlands on swine wastewater containing tetracyclines and tet genes. Water Res 70:109–117. https://doi.org/10.1016/j.watres.2014.11.048
Ji X, Shen Q, Liu F, Ma J, Xu G, Wang Y, Wu M (2012) Antibiotics resistance gene abundances associated with antibiotics and heavy metals in animal manures and agricultural soils adjacent to feedlots in Shanghai; China. J Hazard Mater 235-236(15):178–185. https://doi.org/10.1016/j.jhazmat.2012.07.040
Johnsen PJ, Townsend JP, Bohn T, Simonsen GS, Sundsfjord A, Nielsen KM (2009) Factors affecting the reversal of antimicrobial drug resistance. Lancet Infect Dis 9(6):357–364. https://doi.org/10.1016/S1473-3099(09)70105-7
Kim S, Yun Z, Ha UH, Lee S, Park H, Kwon EE, Cho Y, Choung S, Oh J, Medriano CA, Chandran K (2014) Transfer of antibiotics resistance plasmids in pure and activated sludge cultures in the presence of environmentally representative micro-contaminant concentrations. Sci Total Environ 468:813–820. https://doi.org/10.1016/j.scitotenv.2013.08.100
Klumper U, Riber L, Dechesne A, Sannazzarro A, Hansen LH, Sorensen SJ, Smets BF (2015) Broad host range plasmids can invade an unexpectedly diverse fraction of a soil bacterial community. ISME J 9(4):934–945. https://doi.org/10.1038/ismej.2014.191
Knapp CW, McCluskey SM, Singh BK, Campbell CD, Hudson G, Graham DW (2011) Antibiotics resistance gene abundances correlated with metal and geochemical conditions in archived Scottish soils. PLoS One 6:e27300. https://doi.org/10.1371/journal.pone.0027300
Kolpin DW, Furlong ET, Meyer MT, Thurman EM, Zaugg SD, Barber LB, Buxton HT (2002) Pharmaceuticals, hormones, and other organic wastewater contaminants in US streams, 1999-2000: a national reconnaissance. Environ Sci Technol 36(6):1202–1211. https://doi.org/10.1021/es011055j
Li D, Zeng SY, He M, Gu AZ (2016) Water disinfection byproducts induce antibiotics resistance-role of environmental pollutants in resistance phenomena. Environ Sci Technol 50(6):3193–3201. https://doi.org/10.1021/acs.est.5b05113
Li LG, Xia Y, Zhang T (2017a) Co-occurrence of antibiotics and metal resistance genes revealed in complete genome collection. ISME J 11(3):651–662. https://doi.org/10.1038/ismej.2016.155
Li N, Sheng GP, Lu YZ, Zeng RJ, Yu HQ (2017b) Removal of antibiotics resistance genes from wastewater treatment plant effluent by coagulation. Water Res 111:204–212. https://doi.org/10.1016/j.watres.2017.01.010
Li HC, Duan ML, Gu J, Zhang YJ, Qian X, Ma J, Zhang RR, Wang XJ (2017c) Effects of bamboo charcoal on antibiotics resistance genes during chicken manure composting. Ecotoxicol Environ Saf 140:1–6. https://doi.org/10.1016/j.ecoenv.2017.01.007
Liu MM, Zhang Y, Yang M, Tian Z, Ren LR, Zhang SJ (2012) Abundance and distribution of tetracycline resistance genes and mobile elements in an oxytetracycline production wastewater treatment system. Environ Sci Technol 46(14):7551–7557. https://doi.org/10.1021/es301145m
Lopatkin AJ, Huang SQ, Smith RP, Srimani JK, Sysoeva TA, Bewick S, Karig DK, You LC (2016) Antibiotics as a selective driver for conjugation dynamics. Nat Microbiol 1(6):1–8. https://doi.org/10.1038/nmicrobiol.2016.44
Luo Y, Mao DQ, Rysz M, Zhou QX, Zhang HJ, Xu L, Alvarez PJJ (2010) Trends in antibiotics resistance genes occurrence in the Haihe River, China. Environ Sci Technol 44(19):7220–7225. https://doi.org/10.1021/es100233w
Ma Y, Wilson CA, Novak JT, Riffat R, Aynur S, Murthy S, Pruden A (2011) Effect of various sludge digestion conditions on sulfonamide, macrolide, and tetracycline resistance genes and class I integrons. Environ Sci Technol 45(18):7855–7861. https://doi.org/10.1021/es200827t
Marcato CE, Pinelli E, Cecchi M, Winterton P, Guiresse M (2009) Bioavailability of Cu and Zn in raw and anaerobically digested pig slurry. Ecotoxicol Environ Saf 72(5):1538–1544. https://doi.org/10.1016/j.ecoenv.2008.12.010
Marti E, Huerta B, Rodríguez-Mozaz S, Barceló D, Jofre J, Balcázar JL (2014) Characterization of ciprofloxacin-resistant isolates from a wastewater treatment plant and its receiving river. Water Res 61:67–76. https://doi.org/10.1016/j.watres.2014.05.006
Martínez JL (2008) Antibiotics and antibiotics resistance genes in natural environments. Science 321(5887):365–367. https://doi.org/10.1126/science.1159483
Martínez JL, Coque TM, Baquero F (2015) What is a resistance gene? Ranking risk in resistomes. Nat Rev Microbiol 13(2):116–123. https://doi.org/10.1038/nrmicro3399
Mazel D (2004) Integrons and the origin of antibiotics resistance gene cassettes. ASM News 70(11):520–525
McKinney CW, Pruden A (2012) Ultraviolet disinfection of antibiotics resistant bacteria and their antibiotics resistance genes in water and wastewater. Environ Sci Technol 46(24):13393–13400. https://doi.org/10.1021/es303652q
McKinney CW, Loftin KA, Meyer MT, Davis JG, Pruden A (2010) tet and sul antibiotics resistance genes in livestock lagoons of various operation type, configuration, and antibiotics occurrence. Environ Sci Technol 44(16):6102–6109. https://doi.org/10.1021/es9038165
Michael I, Rizzo L, McArdell CS, Manaia CM, Merlin C, Schwartz T, Dagot C, Fatta-Kassinos D (2013) Urban wastewater treatment plants as hotspots for the release of antibiotics in the environment: a review. Water Res 47(3):957–995. https://doi.org/10.1016/j.watres.2012.11.027
Miller JH, Novak JT, Knocke WR, Pruden A (2016) Survival of antibiotics resistant bacteria and horizontal gene transfer control antibiotics resistance gene content in anaerobic digesters. Front Microbiol 7:263. https://doi.org/10.3389/fmicb.2016.00263
Ochman H, Lawrence JG, Groisman EA (2000) Lateral gene transfer and the nature of bacterial innovation. Nature 405(6784):299–304. https://doi.org/10.1038/35012500
Peak N, Knapp CW, Yang RK, Hanfelt MM, Smith MS, Aga DS, Graham DW (2007) Abundance of six tetracycline resistance genes in wastewater lagoons at cattle feedlots with different antibiotics use strategies. Environ Microbiol 9(1):143–151. https://doi.org/10.1111/j.1462-2920.2006.01123.x
Pruden A, Pei R, Storteboom H, Carlson KH (2006) Antibiotics resistance genes as emerging contaminants: studies in northern Colorado. Environ Sci Technol 40(23):7445–7450. https://doi.org/10.1021/es060413l
Qian X, Sun W, Gu J, Wang XJ, Sun JJ, Yin YN, Duan ML (2016) Variable effects of oxytetracycline on antibiotics resistance gene abundance and the bacterial community during aerobic composting of cow manure. J Hazard Mater 315:61–69. https://doi.org/10.1016/j.jhazmat.2016.05.002
Salyers A, Shoemaker NB (2006) Reservoirs of antibiotics resistance genes. Anim Biotechnol 17(2):137–146. https://doi.org/10.1080/10495390600957076
Sanderson H, Fricker C, Brown RS, Majury A, Liss SN (2016) Antibiotics resistance genes as an emerging environmental contaminant. Environ Rev 24:205–218. https://doi.org/10.1139/er-2015-0069
Selvam A, Xu D, Zhao Z, Wong JWC (2012) Fate of tetracycline, sulfonamide and fluoroquinolone resistance genes and the changes in bacterial diversity during composting of swine manure. Bioresour Technol 126:383–390. https://doi.org/10.1016/j.biortech.2012.03.045
Séveno NA, Kallifidas D, Smalla K, van Elsas JD, Collard JM, Karagouni AD, Wellington EMH (2002) Occurrence and reservoirs of antibiotics resistance genes in the environment. Rev Med Microbiol 13(1):15–27. https://doi.org/10.1007/s10646-017-1853-9
Shi ZJ, Hu HY, Shen YY, Xu JJ, Shi ML, Jin RC (2017) Long-term effects of oxytetracycline (OTC) on the granule-based anammox: process performance and occurrence of antibiotic resistance genes. Biochem Eng J 127:110–118. https://doi.org/10.1016/j.bej.2017.08.009
Song JX, Rensing C, Holm PE, Virta M, Brandt KK (2017) Comparison of metals and tetracycline as selective agents for development of tetracycline resistant bacterial communities in agricultural soil. Environ Sci Technol 51(5):3040–3047. https://doi.org/10.1021/acs.est.6b05342
Stepanauskas R, Glenn TC, Jagoe CH, Tuckfield RC, Lindell AH, King CJ, McArthur JV (2005) Elevated microbial tolerance to metals and antibiotics in metal-contaminated industrial environments. Environ Sci Technol 39(10):3671–3678. https://doi.org/10.1021/es048468f
Stepanauskas R, Glenn TC, Jagoe CH, Tuckfield RC, Lindell AH, King CJ, McArthur JV (2006) Co-selection for microbial resistance to metals and antibiotics in freshwater microcosms. Environ Microbiol 8(9):1510–1514. https://doi.org/10.1111/j.1462-2920.2006.01091.x
Su JQ, Ou-Yang WY, Bei W, Huang FY, Zhao Y, Xu HJ, Zhu YG (2015) Antibiotics resistome and its association with bacterial communities during sewage sludge composting. Environ Sci Technol 49(12):7356–7363. https://doi.org/10.1021/acs.est.5b01012
Sui QW, Zhang JY, Chen MX, Tong J, Wang R, Wei YS (2016) Distribution of antibiotics resistance genes (ARGs) in anaerobic digestion and land application of swine wastewater. Environ Pollut 213:751–759. https://doi.org/10.1016/j.envpol.2016.03.038
Sui QW, Zhang JY, Tong J, Chen MX, Wei YS (2017) Seasonal variation and removal efficiency of antibiotics resistance genes during wastewater treatment of swine farms. Environ Sci Pollut Res 24(10):9048–9057. https://doi.org/10.1007/s11356-015-5891-7
Sun W, Qian X, Gu J, Wang XJ, Duan ML (2016) Mechanism and effect of temperature on variations in antibiotics resistance genes during anaerobic digestion of dairy manure. Sci Rep 6:30237. https://doi.org/10.1038/srep30237
Szekeres E, Baricz A, Chiria CM, Farkas A, Opris O, Soran ML, Andrei AS, Rudi K, Balcazar JL, Dragos N, Coman C (2017) Abundance of antibiotics, antibiotics resistance genes and bacterial community composition in wastewater effluents from different Romanian hospitals. Environ Pollut 225:304–315. https://doi.org/10.1016/j.envpol.2017.01.054
Tang X, Lou C, Wang S, Lu Y, Liu M, Hashmi MZ, Liang X, Li Z, Liao Y, Qin W, Fan F, Xu J, Brookes PC (2015) Effects of long-term manure applications on the occurrence of antibiotics and antibiotics resistance genes (ARGs) in paddy soils: evidence from four field experiments in south of China. Soil Biol Biochem 90:179–187. https://doi.org/10.1016/j.soilbio.2015.07.027
Tao CW, Hsu BM, Ji WT, Hsu TK, Kao PM, Hsu CP, Shen SM, Shen TY, Wan TJ, Huang YL (2014) Evaluation of five antibiotics resistance genes in wastewater treatment systems of swine farms by real-time PCR. Sci Total Environ 496:116–121. https://doi.org/10.1016/j.scitotenv.2014.07.024
Tao WD, Zhang XX, Zhao FZ, Huang KL, Ma HJ, Wang Z, Ye L, Ren HQ (2016) High levels of antibiotics resistance genes and their correlations with bacterial community and mobile genetic elements in pharmaceutical wastewater treatment bioreactors. PLoS One 11(6):e0156854. https://doi.org/10.1371/journal.pone.0156854
Tian Z, Zhang Y, Yu B, Yang M (2016) Changes of resistome, mobilome and potential hosts of antibiotics resistance genes during the transformation of anaerobic digestion from mesophilic to thermophilic. Water Res 98:261–269. https://doi.org/10.1016/j.watres.2016.04.031
Tuckfield RC, McArthur JV (2008) Spatial analysis of antibiotics resistance along metal contaminated streams. Microb Ecol 55(4):595–607. https://doi.org/10.1007/s00248-007-9303-5
Venturini C, Beatson SA, Djordjevic SP, Walker MJ (2010) Multiple antibiotics resistance gene recruitment onto the enterohemorrhagic Escherichia coli virulence plasmid. FASEB J 24(4):1160–1166. https://doi.org/10.1096/fj.09-144972
Wang FH, Qiao M, Lv ZE, Guo GX, Jia Y, Su YH, Zhu YG (2014) Impact of reclaimed water irrigation on antibiotics resistance in public parks, Beijing, China. Environ Pollut 184(S1):247–253. https://doi.org/10.1016/j.envpol.2013.08.038
Wang R, Zhang JY, Sui QW, Wan HF, Tong J, Chen MX, Wei YS, Wei DB (2016a) Effect of red mud addition on tetracycline and copper resistance genes and microbial community during the full scale swine manure composting. Bioresour Technol 216:1049–1057. https://doi.org/10.1016/j.biortech.2016.06.012
Wang J, Ben WW, Yang M, Zhang Y, Qiang ZM (2016b) Dissemination of veterinary antibiotics and corresponding resistance genes from a concentrated swine feedlot along the waste treatment paths. Environ Int 92-93:317–323. https://doi.org/10.1016/j.envint.2016.04.020
WHO (2015) Antimicrobial resistance: global report on surveillance. In World Health Organization. Available at http://apps.who.Int/iris/bitstream/10665/112642/1/9789241564748eng.pdf. Accessed 1 May 2015
Wu D, Huang ZT, Yang K, Graham D, Xie B (2015) Relationships between antibiotics and antibiotics resistance gene levels in municipal solid waste leachates in Shanghai, China. Environ Sci Technol 49(7):4122–4128. https://doi.org/10.1021/es506081z
Xu Y, Guo CS, Luo Y, Lv JP, Zhang Y, Lin HX, Wang L, Xu J (2016) Occurrence and distribution of antibiotics, antibiotics resistance genes in the urban rivers in Beijing, China. Environ Pollut 213:833–840. https://doi.org/10.1016/j.envpol.2016.03.054
Xu Y, Xu J, Mao DQ, Luo Y (2017) Effect of the selective pressure of sub-lethal level of heavy metals on the fate and distribution of ARGs in the catchment scale. Environ Pollut 220:900–908. https://doi.org/10.1016/j.envpol.2016.10.074
Yang Y, Li B, Ju F, Zhang T (2013) Exploring variation of antibiotics resistance genes in activated sludge over a four-year period through a metagenomic approach. Environ Sci Technol 47(18):10197–10205. https://doi.org/10.1021/es4017365
Yi QZ, Zhang Y, Gao YX, Tian Z, Yang M (2017) Anaerobic treatment of antibiotics production wastewater pretreated with enhanced hydrolysis: simultaneous reduction of COD and ARGs. Water Res 110:211–217. https://doi.org/10.1016/j.watres.2016.12.020
Zhang XX, Zhang T, Fang HH (2009) Antibiotics resistance genes in water environment. Appl Microbiol Biotechnol 82(3):397–414. https://doi.org/10.1007/s00253-008-1829-z
Zhang YJ, Boyd SA, Teppen BJ, Tiedje JM, Li H (2014) Role of tetracycline speciation in the bioavailability to Escherichia Coli for uptake and expression of antibiotics resistance. Environ Sci Technol 48(9):4893–4900. https://doi.org/10.1021/es5003428
Zhang ZZ, Zhang QQ, Guo Q, Chen QQ, Jiang XY, Jin RC (2015) Anaerobic ammonium-oxidizing bacteria gain antibiotic resistance during long-term acclimatization. Bioresour Technol 192:756–764. https://doi.org/10.1016/j.biortech.2015.06.044
Zhang JY, Sui QW, Tong J, Buhe C, Wang R, Chen MX, Wei YS (2016a) Sludge bio-drying: effective to reduce both antibiotics resistance genes and mobile genetic elements. Water Res 106:62–70. https://doi.org/10.1016/j.watres.2016.09.055
Zhang JY, Chen MX, Sui QW, Tong J, Jiang C, Lu XT, Zhang YX, Wei YS (2016b) Impacts of addition of natural zeolite or a nitrification inhibitor on antibiotics resistance genes during sludge composting. Water Res 91:339–349. https://doi.org/10.1016/j.watres.2016.01.010
Zhang QQ, Yang GF, Sun KK, Tian GM, Jin RC (2018) Insights into the effects of bio-augmentation on the granule-based anammox process under continuous oxytetracycline stress: performance and microflora structure. Chem Eng J 348:503–513. https://doi.org/10.1016/j.cej.2018.04.204
Zhu YG, Johnson TA, Su JQ, Qiao M, Guo GX, Stedtfeld RD, Hashsham SA, Tiedje JM (2013) Diverse and abundant antibiotics resistance genes in Chinese swine farms. Proc Natl Acad Sci 110(9):3435–3440. https://doi.org/10.1073/pnas.1222743110
Zou W, Li XK, Lai ZY, Zhang XL, Hu XG, Zhou QX (2016) Graphene oxide inhibits antibiotics uptake and antibiotics resistance gene propagation. Appl Mater Interfaces 8(48):33165–33174. https://doi.org/10.1021/acsami.6b09981
Acknowledgments
The authors wish to thank the Natural Science Foundation of China (No. 51578204) and the National Key Research and Development Program of China (Project No. 2016YFD0800605) for their partial support of this study.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Zhang, QQ., Tian, GM. & Jin, RC. The occurrence, maintenance, and proliferation of antibiotic resistance genes (ARGs) in the environment: influencing factors, mechanisms, and elimination strategies. Appl Microbiol Biotechnol 102, 8261–8274 (2018). https://doi.org/10.1007/s00253-018-9235-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-018-9235-7