Abstract
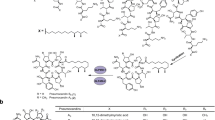
Pimaricin is an important antifungal antibiotic for antifungal therapy and prevention of mould contamination in the food industry. In this study, three new pimaricin derivatives, 12-decarboxy-12-methyl pimaricin (1), 4,5-desepoxy-12-decarboxy-12-methyl pimaricin (2), and 2-hydro-3-hydroxy-4,5-desepoxy-12-decarboxy-12-methyl pimaricin (3), were generated through the inactivation of P450 monooxygenase gene scnG in Streptomyces chattanoogensis L10. Compared with pimaricin, 1 displayed a twofold increase in antifungal activity against Candida albicans ATCC 14053 and a 4.5-fold decrease in cytotoxicity with erythrocytes, and 2 had comparable antifungal activity and reduced cytotoxicity, whereas 3 showed nearly no antifungal and hemolytic activities. Genetic and biochemical analyses proved that 1 is converted from 2 by P450 monooxygenase ScnD. Therefore, the overexpression of scnD in scnG-null strain eliminated the accumulation of 2 and improved the yield of 1 by 20 %. Conversely, scnG/scnD double mutation abolished the production of 1 and improved the yield of 2 to 2.3-fold. These results indicate that the pimaricin derivatives with improved pharmacological properties obtained by genetic engineering can be further developed into antifungal agents for potential clinical application.




Similar content being viewed by others
References
Anton N, Mendes MV, Martin JF, Aparicio JF (2004) Identification of PimR as a positive regulator of pimaricin biosynthesis in Streptomyces natalensis. J Bacteriol 186(9):2567–2575
Anton N, Santos-Aberturas J, Mendes MV, Guerra SM, Martin JF, Aparicio JF (2007) PimM, a PAS domain positive regulator of pimaricin biosynthesis in Streptomyces natalensis. Microbiology 153(Pt 9):3174–3183
Aparicio JF, Colina AJ, Ceballos E, Martin JF (1999) The biosynthetic gene cluster for the 26-membered ring polyene macrolide pimaricin. A new polyketide synthase organization encoded by two subclusters separated by functionalization genes. J Biol Chem 274(15):10133–10139
Aparicio JF, Fouces R, Mendes MV, Olivera N, Martin JF (2000) A complex multienzyme system encoded by five polyketide synthase genes is involved in the biosynthesis of the 26-membered polyene macrolide pimaricin in Streptomyces natalensis. Chem Biol 7(11):895–905
Baginski M, Resat H, Borowski E (2002) Comparative molecular dynamics simulations of amphotericin B-cholesterol/ergosterol membrane channels. Biochim Biophys Acta 1567(1–2):63–78
Borgos SE, Tsan P, Sletta H, Ellingsen TE, Lancelin JM, Zotchev SB (2006) Probing the structure-function relationship of polyene macrolides: engineered biosynthesis of soluble nystatin analogues. J Med Chem 49(8):2431–2439
Brautaset T, Sekurova ON, Sletta H, Ellingsen TE, StrLm AR, Valla S, Zotchev SB (2000) Biosynthesis of the polyene antifungal antibiotic nystatin in Streptomyces noursei ATCC 11455: analysis of the gene cluster and deduction of the biosynthetic pathway. Chem Biol 7(6):395–403
Brautaset T, Sletta H, Nedal A, Borgos SE, Degnes KF, Bakke I, Volokhan O, Sekurova ON, Treshalin ID, Mirchink EP, Dikiy A, Ellingsen TE, Zotchev SB (2008) Improved antifungal polyene macrolides via engineering of the nystatin biosynthetic genes in Streptomyces noursei. Chem Biol 15(11):1198–1206
Caffrey P, Lynch S, Flood E, Finnan S, Oliynyk M (2001) Amphotericin biosynthesis in Streptomyces nodosus: deductions from analysis of polyketide synthase and late genes. Chem Biol 8(7):713–723
Cannon RD, Lamping E, Holmes AR, Niimi K, Tanabe K, Niimi M, Monk BC (2007) Candida albicans drug resistance another way to cope with stress. Microbiology 153(Pt 10):3211–3217
Carmody M, Murphy B, Byrne B, Power P, Rai D, Rawlings B, Caffrey P (2005) Biosynthesis of amphotericin derivatives lacking exocyclic carboxyl groups. J Biol Chem 280(41):34420–34426
Chen S, Huang X, Zhou X, Bai L, He J, Jeong KJ, Lee SY, Deng Z (2003) Organizational and mutational analysis of a complete FR-008/candicidin gene cluster encoding a structurally related polyene complex. Chem Biol 10(11):1065–1076
Chen S, Mao X, Shen Y, Zhou Y, Li J, Wang L, Tao X, Yang L, Wang Y, Zhou X, Deng Z, Wei D (2009) Tailoring the P450 monooxygenase gene for FR-008/candicidin biosynthesis. Appl Environ Microbiol 75(6):1778–1781
De Poire E, Stephens N, Rawlings B, Caffrey P (2013) Engineered biosynthesis of disaccharide-modified polyene macrolides. Appl Environ Microbiol 79(19):6156–6159
Du YL, Chen SF, Cheng LY, Shen XL, Tian Y, Li YQ (2009) Identification of a novel Streptomyces chattanoogensis L10 and enhancing its natamycin production by overexpressing positive regulator ScnRII. J Microbiol 47(4):506–513
Du YL, Li SZ, Zhou Z, Chen SF, Fan WM, Li YQ (2011) The pleiotropic regulator AdpAch is required for natamycin biosynthesis and morphological differentiation in Streptomyces chattanoogensis. Microbiology 157(Pt 5):1300–1311
Georgopapadakou NH, Walsh TJ (1996) Antifungal agents: chemotherapeutic targets and immunologic strategies. Antimicrob Agents Chemother 40(2):279–291
Hutchinson E, Murphy B, Dunne T, Breen C, Rawlings B, Caffrey P (2010) Redesign of polyene macrolide glycosylation: engineered biosynthesis of 19-O-perosaminyl-amphoteronolide B. Chem Biol 17(2):174–182
Kells PM, Ouellet H, Santos-Aberturas J, Aparicio JF, Podust LM (2010) Structure of cytochrome P450 PimD suggests epoxidation of the polyene macrolide pimaricin occurs via a hydroperoxoferric intermediate. Chem Biol 17(8):841–851
Khutorsky VE (1992) Structures of amphotericin B-cholesterol complex. Biochim Biophys Acta 1108(2):123–127
Kieser T, Bibb MJ, Buttner MJ, Charter KF, Hopwood DA (eds)(2000) Practical Streptomyces Genetics. The John Innes Foundation, Norwich, United Kingdom
Matsumori N, Eiraku N, Matsuoka S, Oishi T, Murata M, Aoki T, Ide T (2004) An amphotericin B-ergosterol covalent conjugate with powerful membrane permeabilizing activity. Chem Biol 11(5):673–679
Mendes MV, Recio E, Fouces R, Luiten R, Martin JF, Aparicio JF (2001) Engineered biosynthesis of novel polyenes: a pimaricin derivative produced by targeted gene disruption in Streptomyces natalensis. Chem Biol 8(7):635–644
Mendes MV, Tunca S, Anton N, Recio E, Sola-Landa A, Aparicio JF, Martin JF (2007) The two-component phoR-phoP system of Streptomyces natalensis: inactivation or deletion of phoP reduces the negative phosphate regulation of pimaricin biosynthesis. Metab Eng 9(2):217–227
Mora-Duarte J, Betts R, Rotstein C, Colombo AL, Thompson-Moya L, Smietana J, Lupinacci R, Sable C, Kartsonis N, Perfect J (2002) Comparison of caspofungin and amphotericin B for invasive candidiasis. N Engl J Med 347(25):2020–2029
Nedal A, Sletta H, Brautaset T, Borgos SE, Sekurova ON, Ellingsen TE, Zotchev SB (2007) Analysis of the mycosamine biosynthesis and attachment genes in the nystatin biosynthetic gene cluster of Streptomyces noursei ATCC 11455. Appl Environ Microbiol 73(22):7400–7407
Paquet V, Carreira EM (2006) Significant improvement of antifungal activity of polyene macrolides by bisalkylation of the mycosamine. Org Lett 8(9):1807–1809
Recio E, Aparicio JF, Rumbero A, Martin JF (2006) Glycerol, ethylene glycol and propanediol elicit pimaricin biosynthesis in the PI-factor-defective strain Streptomyces natalensis npi287 and increase polyene production in several wild-type actinomycetes. Microbiology 152(Pt 10):3147–3156
Schagger H (2006) Tricine-SDS-PAGE. Nat Protoc 1(1):16–22
Seco EM, Cuesta T, Fotso S, Laatsch H, Malpartida F (2005) Two polyene amides produced by genetically modified Streptomyces diastaticus var. 108. Chem Biol 12(5):535–543
Stephens N, Rawlings B, Caffrey P (2013) Versatility of enzymes catalyzing late steps in polyene 67-121C biosynthesis. Biosci Biotechnol Biochem 77(4):880–883
Zhou ZX, Xu QQ, Bu QT, Liu SP, Yu P, Li YQ (2015) Transcriptome-guided identification of SprA as a pleiotropic regulator in Streptomyces chattanoogensis. Appl Microbiol Biotechnol 99(3):1287–1298
Acknowledgments
This work was supported by grants from the Ministry of Science and Technology of the People’s Republic of China (No. 2012AA02A706, 2012AA022107, and 2012CB721005), the National Natural Science Foundation of China (No. 31400030 and 31470157), and the Program of University of Michigan—Shanghai Jiao Tong University Collaboration on Biomedical Technology. We are grateful to Prof. Yongquan Li (Zhejiang University) for providing the strain S. chattanoogensis L10.
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Zhen Qi and Qianjin Kang contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 519 kb)
Rights and permissions
About this article
Cite this article
Qi, Z., Kang, Q., Jiang, C. et al. Engineered biosynthesis of pimaricin derivatives with improved antifungal activity and reduced cytotoxicity. Appl Microbiol Biotechnol 99, 6745–6752 (2015). https://doi.org/10.1007/s00253-015-6635-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-015-6635-9