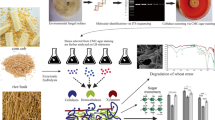
Abstract
A widespread and hitherto by far underexploited potential among ecologically diverse fungi to pretreat wheat straw and digestate from maize silage in the future perspective of using such lignocellulosic feedstock for fermentative bioenergy production was inferred from a screening of nine freshwater ascomycetes, 76 isolates from constructed wetlands, nine peatland isolates and ten basidiomycetes. Wheat straw pretreatment was most efficient with three ascomycetes belonging to the genera Acephala (peatland isolate) and Stachybotrys (constructed wetland isolates) and two white-rot fungi (Hypholoma fasciculare and Stropharia rugosoannulata) as it increased the amounts of water-extractable total sugars by more than 50 % and sometimes up to 150 % above the untreated control. The ascomycetes delignified wheat straw at rates (lignin losses between about 31 and 40 % of the initial content) coming close to those observed with white-rot fungi (about 40 to 57 % lignin removal). Overall, fungal delignification was indicated as a major process facilitating the digestibility of wheat straw. Digestate was generally more resistant to fungal decomposition than wheat straw. Nevertheless, certain ascomycetes delignified this substrate to extents sometimes even exceeding delignification by basidiomycetes. Total sugar amounts of about 20 to 60 % above the control value were obtained with the most efficient fungi (one ascomycete of the genus Phoma, the unspecific wood-rot basidiomycete Agrocybe aegerita and one unidentified constructed wetland isolate). This was accompanied by lignin losses of about 47 to 56 % of the initial content. Overall, digestate delignification was implied to be less decisive for high yields of fermentable sugars than wheat straw delignification.





Similar content being viewed by others
References
Agbor VB, Cicek N, Sparling R, Berlin A, Levin DB (2011) Biomass pretreatment: fundamentals toward application. Biotechnol Adv 29:675–685
Artz R, Anderson I, Chapman S, Hagn A, Schloter M, Potts J, Campbell C (2007) Changes in fungal community composition in response to vegetational succession during the natural regeneration of cutover peatlands. Microb Ecol 54:508–522
Bucher VVC, Pointing SB, Hyde KD, Reddy CA (2004) Production of wood decay enzymes, loss of mass, and lignin solubilization in wood by diverse tropical freshwater fungi. Microb Ecol 48:331–337
Buta JG, Zadrazil F, Galletti GC (1989) FT-IR determination of lignin degradation in wheat straw by white rot fungus Stropharia rugosoannulata with different oxygen concentrations. J Agric Food Chem 37:1382–1384
Chen R, Blagodatskaya E, Senbayram M, Blagodatsky S, Myachina O, Dittert K, Kuzyakov Y (2012) Decomposition of biogas residues in soil and their effects on microbial growth kinetics and enzyme activities. Biomass Bioenergy 45:221–229
Dashtban M, Schraft H, Qin W (2009) Fungal bioconversion of lignocellulosic residues; opportunities & perspectives. Int J Biol Sci 5:578–595
DePasquale DA, Montville TJ (1990) Mechanism by which ammonium bicarbonate and ammonium sulfate inhibit mycotoxigenic fungi. Appl Environ Microbiol 56:3711–3717
Dias AA, Freitas GS, Marques GSM, Sampaio A, Fraga IS, Rodrigues MAM, Evtuguin DV, Bezerra RMF (2010) Enzymatic saccharification of biologically pre-treated wheat straw with white-rot fungi. Bioresour Technol 101:6045–6050
Dorado J, Almendros G, Camarero S, Martinez AT, Vares T, Hatakka A (1999) Transformation of wheat straw in the course of solid-state fermentation by four ligninolytic basidiomycetes. Enzym Microbiol Technol 25:605–612
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for determination of sugars and related substances. Anal Chem 28:350–356
Fackler K, Schwanninger M, Gradinger C, Hinterstoisser B, Messner K (2007) Qualitative and quantitative changes of beech wood degraded by wood-rotting basidiomycetes monitored by Fourier transform infrared spectroscopic methods and multivariate data analysis. FEMS Microbiol Lett 271:162–169
Fahr K, Wetzstein H-G, Grey R, Schlosser D (1999) Degradation of 2,4-dichlorophenol and pentachlorophenol by two brown rot fungi. FEMS Microbiol Lett 175:127–132
Fütterer N, Seeger EM, van Afferden M, Reiche N, Borsdorf H, Rakoczy J, Müller RA, Kästner M, Weiß H (2010) Development of a low-cost technique for the treatment of large-scale contaminated groundwater by using constructed wetlands and aerated trenches. Proceedings CD of ConSoil 2010, 22–24 September 2010, Salzburg, Austria (11th International UFZ—Deltares/TNO Conference on Management of Soil, Groundwater and Sediment), Theme A: Restoration—ThS A19: Special subjects. UFZ, Leipzig (ISBN 978-3-00-032099-6)
Giraud F, Guiraud P, Kadri M, Blake G, Steiman R (2001) Biodegradation of anthracene and fluoranthene by fungi isolated from an experimental constructed wetland for wastewater treatment. Water Res 35:4126–4136
Grünig CR, Sieber TN (2005) Molecular and phenotypic description of the widespread root symbiont Acephala applanata gen. et sp. nov., formerly known as dark-septate endophyte type 1. Mycologia 97:628–640
Isikhuemhen O, Mikiashvili N, Kelkar V (2009) Application of solid waste from anaerobic digestion of poultry litter in Agrocybe aegerita cultivation: mushroom production, lignocellulolytic enzymes activity and substrate utilization. Biodegradation 20:351–361
Janssen GG, Baldwin TM, Winetzky DS, Tierney LM, Wang H, Murray CJ (2004) Selective targeting of a laccase from Stachybotrys chartarum covalently linked to a carotenoid-binding peptide. J Pept Res 64:10–24
Junghanns C, Moeder M, Krauss G, Martin C, Schlosser D (2005) Degradation of the xenoestrogen nonylphenol by aquatic fungi and their laccases. Microbiology 151:45–57
Junghanns C, Krauss G, Schlosser D (2008) Potential of aquatic fungi derived from diverse freshwater environments to decolourise synthetic azo and anthraquinone dyes. Bioresour Technol 99:1225–1235
Junghanns C, Pecyna MJ, Bohm D, Jehmlich N, Martin C, von Bergen M, Schauer F, Hofrichter M, Schlosser D (2009) Biochemical and molecular genetic characterisation of a novel laccase produced by the aquatic ascomycete Phoma sp. UHH 5-1-03. Appl Microbiol Biotechnol 84:1095–1105
Jurgens JA, Blanchette RA, Filley TR (2009) Fungal diversity and deterioration in mummified woods from the ad Astra Ice Cap region in the Canadian High Arctic. Polar Biol 32:751–758
Kaparaju P, Serrano M, Thomsen AB, Kongjan P, Angelidaki I (2009) Bioethanol, biohydrogen and biogas production from wheat straw in a biorefinery concept. Bioresour Technol 100:2562–2568
Klimiuk E, Pokój T, Budzyński W, Dubis B (2010) Theoretical and observed biogas production from plant biomass of different fibre contents. Bioresour Technol 101:9527–9535
Kluczek-Turpeinen B, Steffen K, Tuomela M, Hatakka A, Hofrichter M (2005) Modification of humic acids by the compost-dwelling deuteromycete Paecilomyces inflatus. Appl Microbiol Biotechnol 66:443–449
Krasznai DJ, Champagne P, Cunningham MF (2012) Quantitative characterization of lignocellulosic biomass using surrogate mixtures and multivariate techniques. Bioresour Technol 110:652–661
Krauss G-J, Solé M, Krauss G, Schlosser D, Wesenberg D, Bärlocher F (2011) Fungi in freshwaters: ecology, physiology and biochemical potential. FEMS Microbiol Rev 35:620–651
Liers C, Ullrich R, Steffen K, Hatakka A, Hofrichter M (2006) Mineralization of 14C-labelled synthetic lignin and extracellular enzyme activities of the wood-colonizing ascomycetes Xylaria hypoxylon and Xylaria polymorpha. Appl Microbiol Biotechnol 69:573–579
Liers C, Arnstadt T, Ullrich R, Hofrichter M (2011) Patterns of lignin degradation and oxidative enzyme secretion by different wood- and litter-colonizing basidiomycetes and ascomycetes grown on beech-wood. FEMS Microbiol Ecol 78:91–102
Menardo S, Gioelli F, Balsari P (2011) The methane yield of digestate: effect of organic loading rate, hydraulic retention time, and plant feeding. Bioresour Technol 102:2348–2351
Middelhoven WJ, Scorzetti G, Fell JW (2001) Trichosporon porosum comb. nov., an anamorphic basidiomycetous yeast inhabiting soil, related to the loubieri/laibachii group of species that assimilate hemicelluloses and phenolic compounds. FEMS Yeast Res 1:15–22
Salvachúa D, Prieto A, López-Abelairas M, Lu-Chau T, Martínez ÁT, Martínez MJ (2011) Fungal pretreatment: an alternative in second-generation ethanol from wheat straw. Bioresour Technol 102:7500–7506
Seeger EM, Kuschk P, Fazekas H, Grathwohl P, Kaestner M (2011) Bioremediation of benzene-, MTBE- and ammonia-contaminated groundwater with pilot-scale constructed wetlands. Environ Pollut 159:3769–3776
Shafiei M, Karimi K, Taherzadeh MJ (2010) Palm date fibers: analysis and enzymatic hydrolysis. Int J Mol Sci 11:4285–4296
Šnajdr J, Steffen KT, Hofrichter M, Baldrian P (2010) Transformation of 14C-labelled lignin and humic substances in forest soil by the saprobic basidiomycetes Gymnopus erythropus and Hypholoma fasciculare. Soil Biol Biochem 42:1541–1548
Steffen KT, Hofrichter M, Hatakka A (2000) Mineralisation of 14C-labelled synthetic lignin and ligninolytic enzyme activities of litter-decomposing basidiomycetous fungi. Appl Microbiol Biotechnol 54:819–825
Tuyen VD, Cone JW, Baars JJP, Sonnenberg ASM, Hendriks WH (2012) Fungal strain and incubation period affect chemical composition and nutrient availability of wheat straw for rumen fermentation. Bioresour Technol 111:336–342
Veverka K, Štolcová J, Rùžek P (2007) Sensitivity of fungi to urea, ammonium nitrate and their equimolar solution UAN. Plant Prot Sci 43:157–164
Yadav RS, Patel VK, Yadav KD, Sharma JK, Singh NP (2010) Lignin peroxidases of some indigenous ligninolytic fungi: secretion and enzymatic characteristics. Indian J Microbiol 50(Suppl 1):132–138
Yang X, Ma F, Zeng Y, Yu H, Xu C, Zhang X (2010) Structure alteration of lignin in corn stover degraded by white-rot fungus Irpex lacteus CD2. Int Biodeterior Biodegrad 64:119–123
Yelle DJ, Ralph J, Lu F, Hammel KE (2008) Evidence for cleavage of lignin by a brown rot basidiomycete. Environ Microbiol 10:1844–1849
Acknowledgments
We very much appreciate the provision and processing of peat samples from the peatland “Hollweger Moor” by J. Müller and U. Riegel (both Westerstede-Ocholt, Germany), as well as excellent technical assistance by R. Bötz (Leipzig, Germany). The help of W. Geyer (Leipzig, Germany) with the FT-mIR spectroscopy measurements is also gratefully acknowledged. Furthermore, we want to thank C. Decock and P. Massart (Louvain-la-Neuve, Belgium) for providing nucleotide sequences of newly isolated and identified strains. Resources for this research were provided by the Helmholtz Centre for Environmental Research—UFZ (Leipzig, Germany) research topic “Refining Biomass into Chemical Energy”.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 331 kb)
Rights and permissions
About this article
Cite this article
Singh, S., Harms, H. & Schlosser, D. Screening of ecologically diverse fungi for their potential to pretreat lignocellulosic bioenergy feedstock. Appl Microbiol Biotechnol 98, 3355–3370 (2014). https://doi.org/10.1007/s00253-014-5563-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-014-5563-4