Abstract
Chitin synthases catalyze the formation of β-(1,4)-glycosidic bonds between N-acetylglucosamine residues to form the unbranched polysaccharide chitin, which is the major component of cell walls in most filamentous fungi. Several studies have shown that chitin synthases are structurally and functionally divergent and play crucial roles in the growth and morphogenesis of the genus Aspergillus although little research on this topic has been done in Penicillium chrysogenum. We used BLAST to find the genes encoding chitin synthases in P. chrysogenum related to chitin synthase genes in Aspergillus nidulans. Three homologous sequences coding for a class III chitin synthase CHS4 and two hypothetical proteins in P. chrysogenum were found. The gene which product showed the highest identity and encoded the class III chitin synthase CHS4 was studied in detail. To investigate the role of CHS4 in P. chrysogenum morphogenesis, we developed an RNA interference system to silence the class III chitin synthase gene chs4. After transformation, mutants exhibited a slow growth rate and shorter and more branched hyphae, which were distinct from those of the original strain. The results also showed that the conidiation efficiency of all transformants was reduced sharply and indicated that chs4 is essential in conidia development. The morphologies of all transformants and the original strain in penicillin production were investigated by light microscopy, which showed that changes in chs4 expression led to a completely different morphology during fermentation and eventually caused distinct penicillin yields, especially in the transformants PcRNAi1-17 and PcRNAi2-1 where penicillin production rose by 27 % and 41 %, respectively.









Similar content being viewed by others
References
Borgia PT, Iartchouk N, Riggle PJ, Winter KR, Koltin Y, Bulawa CE (1996) The chsB gene of Aspergillus nidulans is necessary for normal hyphal growth and development. Fungal Genet Biol 20:193–203
Chain EB, Gualandi G, Morisi G (1966) Aeration studies. IV. Aeration conditions in 3000-liter submerged fermentations with various microorganisms. Biotechnol Bioeng 8:595–619
Chen GC, Johnson BR (1983) Improved colorimetric determination of cell wall chitin in wood decay fungi. Appl Environ Microbiol 46:13–16
Choquer M, Boccara M, Gonçalves IR, Soulié MC, Vidal-Cros A (2004) Survey of the Botrytis cinerea chitin synthase multigenic family through the analysis of six euascomycetes genomes. Eur J Biochem 271:2153–2164
Crawford L, Stepan AM, McAda PC, Rambosek JA, Conder MJ, Vinci VA, Reeves CD (1995) Production of cephalosporin intermediates by feeding adipic acid to recombinant Penicillium chrysogenum strains expressing ring expansion activity. Biotechnology (N Y) 13:58–62
Dang Y, Yang Q, Xue Z, Liu Y (2011) RNA interference in fungi: pathways, functions, and applications. Eukaryot Cell 10:1148–1155
Douma RD, Verheijen PJ, de Laat WT, Heijnen JJ, van Gulik WM (2010) Dynamic gene expression regulation model for growth and penicillin production in Penicillium chrysogenum. Biotechnol Bioeng 106:608–618
Elmayergi H, Scharer JM, Moo-Young M (1973) Effects of polymer additives on fermentation parameters in a culture of A. niger. Biotechnol Bioeng 15:845–859
Fukuda K, Yamada K, Deoka K, Yamashita S, Ohta A, Horiuchi H (2009) Class III chitin synthase ChsB of Aspergillus nidulans localizes at the sites of polarized cell wall synthesis and is required for conidial development. Eukaryot Cell 8:945–956
Jami MS, Barreiro C, García-Estrada C, Martín JF (2010) Proteome analysis of the penicillin producer Penicillium chrysogenum: characterization of protein changes during the industrial strain improvement. Mol Cell Proteomics 9:1182–1198
Janus D, Hoff B, Kück U (2009) Evidence for Dicer-dependent RNA interference in the industrial penicillin producer Penicillium chrysogenum. Microbiology 155:3946–3956
Kristiansen B, Bullock JD (1988) Developments in industrial fungal biotechnology. In: Smith JE, Berry DR, Kristiansen B (eds) Fungal biotechnology. Academic, London, pp 203–223
Kück U, Hoff B (2010) New tools for the genetic manipulation of filamentous fungi. Appl Microbiol Biotechnol 86:51–62
Luengo JM, Domínguez A, Cantroal JM, Martín JF (1986) Formation of bulges associated with penicillin production in high-producing stains of Penicillium chrysogenum. Curr Microbiol 13:203–207
Maier FJ, Malz S, Lösch AP, Lacour T, Schäfer W (2005) Development of a highly efficient gene targeting system for Fusarium graminearum using the disruption of a polyketide synthase gene as a visible marker. FEMS Yeast Res 5:653–662
Makagiansar HY, Shamalou PA, Thomas CR, Lilly MD (1993) The influence of mechanical forces on the morphology and penicillin production of Penicillium chrysogenum. Bioproc Eng 9:83–90
Mellado E, Aufauvre-Brown A, Gow NA, Holden DW (1996) The Aspergillus fumigatus chsC and chsG genes encode Class III chitin synthases with different functions. Mol Microbiol 20:667–679
Nakayashiki H, Hanada S, Nguyen BQ, Kadotani N, Tosa Y, Mayama S (2005) RNA silencing as a tool for exploring gene function in ascomycete fungi. Fungal Genet Biol 42:275–283
Nguyen QB, Kadotani N, Kasahara S, Tosa Y, Mayama S, Nakayashiki H (2008) Systematic functional analysis of calcium-signalling proteins in the genome of the rice-blast fungus, Magnaporthe oryzae, using a high-throughput RNA silencing system. Mol Microbiol 68:1348–1365
Papagianni M (2004) Fungal morphology and metabolite production in submerged mycelial process. Biotechnol Adv 22:189–259
Paul GC, Thomas CR (1996) A structured model for hyphal differentiation and penicillin production using Penicillium chrysogenum. Biotechnol Bioeng 51:558–572
Pessoni RA, Freshour G, RdCL F-R, Hahn MG, Braga MR (2005) Cell-wall structure and composition of Penicillium janczewskii as affected by inulin. Mycologia 97:304–311
Phillips DH (1966) Oxygen transfer into mycelial pellets. Biotechnol Bioeng 8:456–460
Rowlands RT, Turner G (1973) Nuclear and extranuclear inheritance of oligomycin resistance in Aspergillus nidulans. Mol Gen Genet 126:201–216
Salame TM, Ziv C, Hadar Y, Yarden O (2011) RNAi as a potential tool for biotechnological applications in fungi. Appl Microbiol Biotechnol 89:501–512
Shamlou PA, Makagiansar HY, Ison AP, Lilly MD (1994) Turbulent breakage of filamentous microorganisms in submerged culture in mechanically stirred bioreactors. Chem Eng Sci 49:2621–2631
Smith JJ, Lilly MD, Fox RI (1990) The effect of agitation on the morphology and penicillin production of Penicillium chrysogenum. Biotechnol Bioeng 35:1011–1023
Steel R, Lentz CP, Martin SM (1954) A standard inoculum for citric acid production in submerged culture. Can J Microbiol 1:150–157
Tucker KG, Thomas CR (1993) Effect of biomass concentration and morphology on the rheological parameters of Penicillium chrysogenum fermentation broths. Trans Inst Chem Eng 71:111–117
Ullán RV, Godio RP, Teijeira F, Vaca I, García-Estrada C, Feltrer R, Kosalkova K, Martín JF (2008) RNA-silencing in Penicillium chrysogenum and Acremonium chrysogenum: validation studies using β-lactam genes expression. J Microbiol Methods 75:209–218
United States Pharmacopeial Convention (2006) United States Pharmacopoeia 29 National Formulary 24 (USP 29-NF 24). United State Pharmacopeia, Rockville
van den Berg MA, Albang R, Albermann K, Badger JH, Daran JM, Driessen AJ, Garcia-Estrada C, Fedorova ND, Harris DM, Heijne WH, Joardar V, Kiel JA, Kovalchuk A, Martín JF, Nierman WC, Nijland JG, Pronk JT, Roubos JA, van der Klei IJ, van Peij NN, Veenhuis M, von Döhren H, Wagner C, Wortman J, Bovenberg RA (2008) Genome sequencing and analysis of the filamentous fungus Penicillium chrysogenum. Nat Biotechnol 26:1161–1168
van Suijdam JC, Kossen NWF, Paul PG (1980) An inoculum technique for the production of fungal pellets. Eur J Appl Microbiol Biotechnol 10:211–221
Yanai K, Kojima N, Takaya N, Horiuchi H, Ohta A, Takagi M (1994) Isolation and characterization of two chitin synthase genes from Aspergillus nidulans. Biosci Biotechnol Biochem 58:1828–1835
Yarden O, Yanofsky C (1991) Chitin synthase 1 plays a major role in cell wall biogenesis in Neurospora crassa. Genes Dev 5:2420–2430
Zangirolami TC, Johansen CL, Nielsen J, Jørgensen SB (1997) Simulation of penicillin production in fed-batch cultivations using a morphologically structured model. Biotechnol Bioeng 56:593–604
Acknowledgments
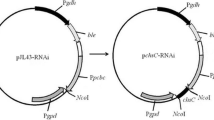
We appreciate the kind donation of plasmid pJL43-RNAi by Professor Juan F. Martín. The present work was supported by key 863 fund of China (no. 2009AA02Z305).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, H., Zheng, Z., Wang, P. et al. Morphological changes induced by class III chitin synthase gene silencing could enhance penicillin production of Penicillium chrysogenum . Appl Microbiol Biotechnol 97, 3363–3372 (2013). https://doi.org/10.1007/s00253-012-4581-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-4581-3