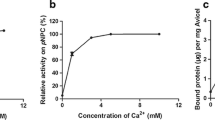
Abstract
Cytophaga hutchinsonii is an abundant aerobic cellulolytic bacterium that rapidly digests crystalline cellulose in a contact-dependent manner. The different roles of various predicted glycoside hydrolases and the detailed mechanism used by C. hutchinsonii in cellulose digestion are, however, not known. In this study, an endoglucanase belonging to glycoside hydrolase family 5 (GH5) named as ChCel5A was isolated from the outer membrane of C. hutchinsonii. The catalytic domain of ChCel5A exhibited typical endoglucanase activity and was capable of hydrolyzing insoluble cellulose with cellobiose and cellotriose as the predominant digestion products. Site-directed mutagenesis identified two aromatic amino acids in ChCle5A, W61 and W308, that dramatically decreased its hydrolytic activity toward filter paper while causing only a slight decrease in carboxymethylcellulase (CMCase) activity. Disruption of chu_1107 encoding ChCel5A caused no drastic effect on the growth parameters on cellulose for the resulting mutant strain with negligible reduction in the specific CMCase activities for intact cells. The demonstration of targeted gene inactivation capability for C. hutchinsonii has provided an opportunity to improve understanding of the details of the mechanism underlying its efficient utilization of cellulose.







Similar content being viewed by others
References
Bortoli-German I, Haiech J, Chippaux M, Barras F (1995) Informational suppression to investigate structural functional and evolutionary aspects of the Erwinia chrysanthemi cellulase EGZ. J Mol Biol 246(1):82–94
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chang WT, Thayer DW (1977) The cellulase system of a Cytophaga species. Can J Microbiol 23(9):1285–1292
Chapon V, Czjzek M, El Hassouni M, Py B, Juy M, Barras F (2001) Type II protein secretion in gram-negative pathogenic bacteria: the study of the structure/secretion relationships of the cellulase Cel5 (formerly EGZ) from Erwinia chrysanthemi. J Mol Biol 310(5):1055–1066
Divne C, Stahlberg J, Reinikainen T, Ruohonen L, Pettersson G, Knowles JK, Teeri TT, Jones TA (1994) The three-dimensional crystal structure of the catalytic core of cellobiohydrolase I from Trichoderma reesei. Science 265(5171):524–528
Gong J, Forsberg CW (1993) Separation of outer and cytoplasmic membranes of Fibrobacter succinogenes and membrane and glycogen granule locations of glycanases and cellobiase. J Bacteriol 175(21):6810–6821
Li LY, Shoemaker NB, Salyers AA (1995) Location and characteristics of the transfer region of a bacteroides conjugative transposon and regulation of transfer genes. J Bacteriol 177(17):4992–4999
Lynd LR, Weimer PJ, van Zyl WH, Pretorius IS (2002) Microbial cellulose utilization: fundamentals and biotechnology. Microbiol Mol Biol Rev 66(3):506–577
Lynd LR, van Zyl WH, McBride JE, Laser M (2005) Consolidated bioprocessing of cellulosic biomass: an update. Curr Opin Biotechnol 16(5):577–583
McBride MJ, Baker SA (1996) Development of techniques to genetically manipulate members of the genera Cytophaga, Flavobacterium, Flexibacter, and Sporocytophaga. Appl Environ Microbiol 62(8):3017–3022
Miller GL (1959) Use of dinitrosalicylic acid reagent for determination of reducing sugar. Anal Biochem 31:426–427
Olson DG, Tripathi SA, Giannone RJ, Lo J, Caiazza NC, Hogsett DA, Hettich RL, Guss AM, Dubrovsky G, Lynd LR (2010) Deletion of the Cel48S cellulase from Clostridium thermocellum. Proc Natl Acad Sci U S A 107(41):17727–17732
Payne CM, Bomble YJ, Taylor CB, McCabe C, Himmel ME, Crowley MF, Beckham GT (2011) Multiple functions of aromatic–carbohydrate interactions in a processive cellulase examined with molecular simulation. J Biol Chem 286(47):41028–41035
Perret S, Maamar H, Belaich JP, Tardif C (2004) Use of antisense RNA to modify the composition of cellulosomes produced by Clostridium cellulolyticum. Mol Microbiol 51(2):599–607
Russell JB (1985) Fermentation of cellodextrins by cellulolytic and noncellulolytic rumen bacteria. Appl Environ Microbiol 49(3):572–576
Stanier RY (1942) The Cytophaga group: a contribution to the biology of myxobacteria. Bacteriol Rev 6(3):143–196
Tolonen AC, Chilaka AC, Church GM (2009) Targeted gene inactivation in Clostridium phytofermentans shows that cellulose degradation requires the family 9 hydrolase Cphy3367. Mol Microbiol 74(6):1300–1313
Walker E, Warren FL (1938) Decomposition of cellulose by Cytophaga. I. Biochem J 32(1):31–43
Watson BJ, Zhang H, Longmire AG, Moon YH, Hutcheson SW (2009) Processive endoglucanases mediate degradation of cellulose by Saccharophagus degradans. J Bacteriol 191(18):5697–5705
Wilson DB (2008) Three microbial strategies for plant cell wall degradation. Ann N Y Acad Sci 1125:289–297
Xie G, Bruce DC, Challacombe JF, Chertkov O, Detter JC, Gilna P, Han CS, Lucas S, Misra M, Myers GL, Richardson P, Tapia R, Thayer N, Thompson LS, Brettin TS, Henrissat B, Wilson DB, McBride MJ (2007) Genome sequence of the cellulolytic gliding bacterium Cytophaga hutchinsonii. Appl Environ Microbiol 73(11):3536–3546
Xu Y, Ji X, Chen N, Li P, Liu W, Lu X (2011) Development of replicative oriC plasmids and their versatile use in genetic manipulation of Cytophaga hutchinsonii. Appl Microbiol Biotechnol 93(2):697–705
Yao Q, Sun TT, Liu WF, Chen GJ (2008) Gene cloning and heterologous expression of a novel endoglucanase, swollenin, from Trichoderma pseudokoningii S38. Biosci Biotechnol Biochem 72(11):2799–2805
Zhang YH, Lynd LR (2004) Toward an aggregated understanding of enzymatic hydrolysis of cellulose: noncomplexed cellulase systems. Biotechnol Bioeng 88(7):797–824
Zhang YH, Lynd LR (2005a) Cellulose utilization by Clostridium thermocellum: bioenergetics and hydrolysis product assimilation. Proc Natl Acad Sci U S A 102(20):7321–7325
Zhang YH, Lynd LR (2005b) Determination of the number-average degree of polymerization of cellodextrins and cellulose with application to enzymatic hydrolysis. Biomacromolecules 6(3):1510–1515
Zhu Y, Li H, Zhou H, Chen G, Liu W (2010) Cellulose and cellodextrin utilization by the cellulolytic bacterium Cytophaga hutchinsonii. Bioresour Technol 101(16):6432–6437
Acknowledgments
We are grateful for Dr. MJ McBride for providing the plasmids and strains and critical reading of this manuscript. This work is supported by grants from the National Basic Research Program (2011CB707402), the National Natural Science Foundation of China (31170762), New Century Excellent Talents in University (NCET-10-0546), Shandong Provincial Funds for Distinguished Young Scientists (JQ201108), and Independent Innovation Foundation of Shandong University, IIFSDU.
Author information
Authors and Affiliations
Corresponding author
Additional information
Yongtao Zhu and Hong Zhou contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 4093 kb)
Rights and permissions
About this article
Cite this article
Zhu, Y., Zhou, H., Bi, Y. et al. Characterization of a family 5 glycoside hydrolase isolated from the outer membrane of cellulolytic Cytophaga hutchinsonii . Appl Microbiol Biotechnol 97, 3925–3937 (2013). https://doi.org/10.1007/s00253-012-4259-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-4259-x