Abstract
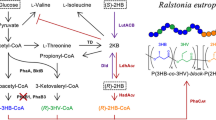
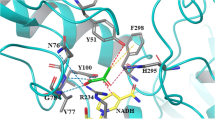
Class I polyhydroxyalkanoate (PHA) synthase from Ralstonia eutropha (PhaCRe) was engineered so as to acquire an unusual lactate (LA)-polymerizing activity. To achieve this, the site-directed saturation mutagenesis of PhaCRe was conducted at position 510, which corresponds to position 481 in the initially discovered class II LA-polymerizing PHA synthase (PhaC1PsSTQK), a mutation in which (Gln481Lys) was shown to be essential to its LA-polymerizing activity (Taguchi et al., Proc Natl Acad Sci USA 105(45):17323–17327, 2008). The LA-polymerizing activity of the PhaCReA510X mutants was evaluated based on the incorporation of LA units into the P[3-hydroxybutyrate(3HB)] backbone in vivo using recombinant Escherichia coli LS5218. Among 19 PhaCRe(A510X) mutants, 15 synthesized P (LA-co-3HB), indicating that the 510 residue plays a critical role in LA polymerization. The polymer synthesized by PhaCReA510S was fractionated using gel permeation chromatography in order to remove the low molecular weight fractions. The 13C and 1H NMR analyses of the high molecular weight fraction revealed that the polymer was a P(7 mol% LA-co-3HB) copolymer with a weight-averaged molecular weight of 3.2 × 105 Da. Interestingly, the polymer contained an unexpectedly high ratio of an LA-LA*-LA triad sequence, suggesting that the polymer synthesized by PhaCRe mutant may not be a random copolymer, but presumably had a block sequence.



Similar content being viewed by others
References
Auras R, Harte B, Selke S (2004) An overview of polylactides as packaging materials. Macromol Biosci 4(9):835–864
Doi Y (1990) Microbial polyesters. VCH, New York
Elsden SR, Volcani BE, Gilchrist FMC, Lewis D (1956) Properties of a fatty acid forming organism isolated from the rumen of sheep. J Bacteriol 72(5):681–689
Han X, Satoh Y, Satoh T, Matsumoto K, Kakuchi T, Taguchi S, Dairi T, Munekata M, Tajima K (2011) Chemo-enzymatic synthesis of polyhydroxyalkanoate (PHA) incorporating 2-hydroxybutyrate by wild-type class I PHA synthase from Ralstonia eutropha. Appl Microbiol Biotechnol 92(3):509–517
Jia Y, Yuan W, Wodzinska J, Park C, Sinskey AJ, Stubbe JA (2001) Mechanistic studies on class I polyhydroxybutyrate (PHB) synthase from Ralstonia eutropha: class I and III synthases share a similar catalytic mechanism. Biochemistry 40(4):1011–1019
Kataoka K, Harada A, Nagasaki Y (2001) Block copolymer micelles for drug delivery: design, characterization and biological significance. Adv Drug Deliv Rev 47(1):113–131
Matsumoto K, Taguchi S (2010) Enzymatic and whole-cell synthesis of lactate-containing polyesters: toward the complete biological production of polylactate. Appl Microbiol Biotechnol 85(4):921–932
Matsumoto K, Takase K, Yamamoto Y, Doi Y, Taguchi S (2009) Chimeric enzyme composed of polyhydroxyalkanoate (PHA) synthases from Ralstonia eutropha and Aeromonas caviae enhances production of PHAs in recombinant Escherichia coli. Biomacromolecules 10(4):682–685
Matsumoto K, Ishiyama A, Sakai K, Shiba T, Taguchi S (2011) Biosynthesis of glycolate-based polyesters containing medium-chain-length 3-hydroxyalkanoates in recombinant Escherichia coli expressing engineered polyhydroxyalkanoate synthase. J Biotechnol 156(3):214–217
Nampoothiri KM, Nair NR, John RP (2010) An overview of the recent developments in polylactide (PLA) research. Bioresour Technol 101(22):8493–8501
Pederson EN, McChalicher CWJ, Srienc F (2006) Bacterial synthesis of PHA block copolymers. Biomacromolecules 7(6):1904–1911
Peoples OP, Sinskey AJ (1989) Poly-β-hydroxybutyrate (PHB) biosynthesis in Alcaligenes eutrophus H16. Identification and characterization of the PHB polymerase gene (phbC). J Biol Chem 264(26):15298–15303
Rehm BHA (2003) Polyester synthases: natural catalysts for plastics. Biochem J 376(Pt 1):15–33
Schubert P, Steinbüchel A, Schlegel HG (1988) Cloning of the Alcaligenes eutrophus genes for synthesis of poly-β-hydroxybutyric acid (PHB) and synthesis of PHB in Escherichia coli. J Bacteriol 170(12):5837–5847
Shozui F, Matsumoto K, Motohashi R, Sun J, Satoh T, Kakuchi T, Taguchi S (2011) Biosynthesis of a lactate (LA)-based polyester with a 96 mol% LA fraction and its application to stereocomplex formation. Polym Degrad Stab 96(4):499–504
Song Y, Matsumoto K, Yamada M, Gohda A, Brigham CJ, Sinskey AJ, Taguchi S (2012) Engineered Corynebacterium glutamicum as an endotoxin-free platform strain for lactate-based polyester production. Appl Microbiol Biotechnol 93(5):1917–1925
Spratt SK, Ginsburgh CL, Nunn WD (1981) Isolation and genetic characterization of Escherichia coli mutants defective in propionate metabolism. J Bacteriol 146(3):1166–1169
Taguchi S, Doi Y (2004) Evolution of polyhydroxyalkanoate (PHA) production system by “enzyme evolution”: successful case studies of directed evolution. Macromol Biosci 4(3):145–156
Taguchi S, Nakamura H, Hiraishi T, Yamato I, Doi Y (2002) In vitro evolution of a polyhydroxybutyrate synthase by intragenic suppression-type mutagenesis. J Biochem 131(6):801–806
Taguchi S, Yamada M, Matsumoto K, Tajima K, Satoh Y, Munekata M, Ohno K, Kohda K, Shimamura T, Kambe H (2008) A microbial factory for lactate-based polyesters using a lactate-polymerizing enzyme. Proc Natl Acad Sci USA 105(45):17323–17327
Tajima K, Satoh Y, Satoh T, Itoh R, Han X, Taguchi S, Kakuchi T, Munekata M (2009) Chemo-enzymatic synthesis of poly (lactate-co-(3-hydroxybutyrate)) by a lactate-polymerizing enzyme. Macromolecules 42(6):1985–1989
Tajima K, Han X, Satoh Y, Ishii A, Araki Y, Munekata M, Taguchi S (2012) In vitro synthesis of polyhydroxyalkanoate (PHA) incorporating lactate (LA) with a block sequence by using a newly engineered thermostable PHA synthase from Pseudomonas sp. SG4502 with acquired LA-polymerizing activity. Appl Microbiol Biotechnol 94(2):365–376
Tripathi L, Wu LP, Chen J, Chen GQ (2012) Synthesis of diblock copolymer poly-3-hydroxybutyrate-block-poly-3-hydroxyhexanoate [PHB-b-PHHx] by a β-oxidation weakened Pseudomonas putida KT2442. Microbial Cell Factories 11(1):44
Tsuge T, Taguchi K, Taguchi S, Doi Y (2003) Molecular characterization and properties of (R)-specific enoyl-CoA hydratases from Pseudomonas aeruginosa: metabolic tools for synthesis of polyhydroxyalkanoates via fatty acid β-oxidation. Int J Biol Macromol 31(4–5):195–205
Tsuge T, Saito Y, Narike M, Muneta K, Normi YM, Kikkawa Y, Hiraishi T, Doi Y (2004) Mutation effects of a conserved alanine (Ala510) in type I polyhydroxyalkanoate synthase from Ralstonia eutropha on polyester biosynthesis. Macromol Biosci 4(10):963–970
Tsuji H (2002) Polylactide. In: Doi Y, Steinbüchel A (eds) Biopolymers, vol 4. Wiley, Weinheim, pp 129–177
Valentin HE, Steinbüechel A (1994) Application of enzymatically synthesized short-chain-length hydroxy fatty acid coenzyme A thioesters for assay of polyhydroxyalkanoic acid synthases. Appl Microbiol Biotechnol 40(5):699–709
Yamada M, Matsumoto K, Nakai T, Taguchi S (2009) Microbial production of lactate-enriched poly [(R)-lactate-co-(R)-3-hydroxybutyrate] with novel thermal properties. Biomacromolecules 10(4):677–681
Yamada M, Matsumoto K, Shimizu K, Uramoto S, Nakai T, Shozui F, Taguchi S (2010) Adjustable mutations in lactate (LA)-polymerizing enzyme for the microbial production of LA-based polyesters with tailor-made monomer composition. Biomacromolecules 11(3):815–819
Yang TH, Jung YK, Kang HO, Kim TW, Park SJ, Lee SY (2011) Tailor-made type II Pseudomonas PHA synthases and their use for the biosynthesis of polylactic acid and its copolymer in recombinant Escherichia coli. Appl Microbiol Biotechnol 90(2):603–614
Yuan W, Jia Y, Tian J, Snell KD, Müh U, Sinskey AJ, Lambalot RH, Walsh CT, Stubbe JA (2001) Class I and III polyhydroxyalkanoate synthases from Ralstonia eutropha and Allochromatium vinosum: characterization and substrate specificity studies. Arch Biochem Biophys 394(1):87–98
Zhang S, Yasuo T, Lenz RW, Goodwin S (2000) Kinetic and mechanistic characterization of the polyhydroxybutyrate synthase from Ralstonia eutropha. Biomacromolecules 1(2):244–251
Zhang S, Kamachi M, Takagi Y, Lenz RW, Goodwin S (2001) Comparative study of the relationship between monomer structure and reactivity for two polyhydroxyalkanoate synthases. Appl Microbiol Biotechnol 56(1):131–136
Acknowledgments
We thank Dr. Hideki Abe (RIKEN Institute) for critical discussion, and Dr. Nobuhito Kurono and Dr. Tomohiro Hirose (both Hokkaido University) for technical assistance with the NMR analysis. This work was partly supported by a Research Fellowship for Young Scientists from the Japan Society for the Promotion of Science (To A. O.), a Grant-in-Aid for Scientific Research of Japan (no. 23310059) (to S.T.), and the Global COE Program (project no. B01: Catalysis as the Basis for Innovation in Materials Science), all from the Ministry of Education, Culture, Sports, Science and Technology of Japan. Pacific Edit reviewed the manuscript prior to submission.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 19 kb)
Rights and permissions
About this article
Cite this article
Ochi, A., Matsumoto, K., Ooba, T. et al. Engineering of class I lactate-polymerizing polyhydroxyalkanoate synthases from Ralstonia eutropha that synthesize lactate-based polyester with a block nature. Appl Microbiol Biotechnol 97, 3441–3447 (2013). https://doi.org/10.1007/s00253-012-4231-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-012-4231-9