Abstract
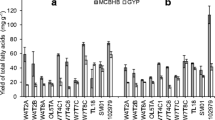
Heterotrophic growth of thraustochytrids has potential in co-producing a feedstock for biodiesel and long-chain (LC, ≥C20) omega-3 oils. Biodiscovery of thraustochytrids from Tasmania (temperate) and Queensland (tropical), Australia, covered a biogeographic range of habitats including fresh, brackish, and marine waters. A total of 36 thraustochytrid strains were isolated and separated into eight chemotaxonomic groups (A–H) based on fatty acid (FA) and sterol composition which clustered closely with four different genera obtained by 18S rDNA molecular identification. Differences in the relative proportions (%FA) of long-chain C20, C22, omega-3, and omega-6 polyunsaturated fatty acids (PUFA), including docosahexaenoic acid (DHA), docosapentaenoic acid, arachidonic acid, eicosapentaenoic acid (EPA), and saturated FA, as well as the presence of odd-chain PUFA (OC-PUFA) were the major factors influencing the separation of these groups. OC-PUFA were detected in temperate strains of groups A, B, and C (Schizochytrium and Thraustochytrium). Group D (Ulkenia) had high omega-3 LC-PUFA (53% total fatty acids (TFA)) and EPA up to 11.2% TFA. Strains from groups E and F (Aurantiochytrium) contained DHA levels of 50–61% TFA after 7 days of growth in basal medium at 20 °C. Groups G and H (Aurantiochytrium) strains had high levels of 15:0 (20–30% TFA) and the sum of saturated FA was in the range of 32–51%. β,β-Carotene, canthaxanthin, and astaxanthin were identified in selected strains. Phylogenetic and chemotaxonomic groupings demonstrated similar patterns for the majority of strains. Our results demonstrate the potential of these new Australian thraustochytrids for the production of biodiesel in addition to omega-3 LC-PUFA-rich oils.




Similar content being viewed by others
References
Abe E, Hayashi Y, Hama Y, Hayashi M, Inagaki M, Ito M (2006) A novel phosphatidylcholine which contains pentadecanoic acid at sn-1 and docosahexaenoic acid at sn-2 in Schizochytrium sp. F26-b. J Biochem 140(2):247–253
Barclay WR, Meager KM, Abril JR (1994) Heterotrophic production of long chain omega-3 fatty acids utilizing algae and algae-like microorganisms. J Appl Phycol 6(2):123–129
Boussiba S (2000) Carotenogenesis in the green alga Haematococcus pluvialis: cellular physiology and stress response. Physiol Plant 108(2):111–117
Boussiba S, Vonshak A (1991) Astaxanthin accumulation in the green alga Haematococcus pluvialis1. Plant Cell Physiol 32(7):1077–1082
Bowles RD, Hunt AE, Bremer GB, Duchars MG, Eaton RA (1999) Long-chain n-3 polyunsaturated fatty acid production by members of the marine protistan group the thraustochytrids: screening of isolates and optimisation of docosahexaenoic acid production. J Biotechnol 70(1–3):193–202
Carmona ML, Naganuma T, Yamaoka Y (2003) Identification by HPLC-MS of carotenoids of the Thraustochytrium CHN-1 strain isolated from the Aeto Inland Sea. Biosci Biotechnol Biochem 67(4):884–888
Cavalier-Smith T, Allsopp MTEP, Chao EE (1994) Thraustochytrids are chromists, not fungi: 18 s rRNA signatures of Heterokonta. Philosophical Transactions of the Royal Society of London. Ser B: Biol Sci 346(1318):387–397
Chisti Y (2007) Biodiesel from microalgae. Biotechnol Adv 25(3):294–306
Cox SL, Hulston D, Maas EW (2009) Cryopreservation of marine thraustochytrids (Labyrinthulomycetes). Cryobiology 59(3):363–365
Ellenbogen BB, Aaronson S (1969) Polyunsaturated fatty acids of aquatic fungi: possible phylogenetic significance. Comp Biochem Physiol 29:805–811
Fan KW, Chen F (2007) Production of high-value products by marine microalgae thraustochytrids. In: Shang-Tian Y (ed) Bioprocessing for value-added products from renewable resources. Elsevier, Amsterdam, pp 293–323
Fan KW, Jiang Y, Faan YW, Chen F (2007) Lipid characterization of mangrove thraustochytrid—Schizochytrium mangrovei. J Agric Food Chem 55(8):2906–2910
Fan K-W, Jiang Y, Ho L-T, Chen F (2009) Differentiation in fatty acid profiles of pigmented and nonpigmented Aurantiochytrium isolated from Hong Kong mangroves. J Agric Food Chem 57(14):6334–6341
Forsyth JS, Carlson SE (2001) Long-chain polyunsaturated fatty acids in infant nutrition: effects on infant development. Curr Opin Clin Nutr Metab Care 4(2):123–126
Gavrilescu M, Chisti Y (2005) Biotechnology—a sustainable alternative for chemical industry. Biotechnol Adv 23(7–8):471–499
Guil-Guerrero J, López-Martínez J, Rincón-Cervera M, Campra-Madrid P (2007) One-step extraction and concentration of polyunsaturated fatty acids from fish liver. J Am Oil Chem Soc 84(4):357–361
Honda D, Yokochi T, Nakahara T, Erata M, Higashihara T (1998) Schizochytrium limacinum sp. nov., a new thraustochytrid from a mangrove area in the west Pacific Ocean. Mycol Res 102(Part 4):439–448
Honda D, Yokochi T, Nakahara T, Raghukumar S, Nakagiri A, Schaumann K, Higashihara T (1999) Molecular phylogeny of labyrinthulids and thraustochytrids based on the sequencing of 18S ribosomal RNA gene. J Eukaryot Microbiol 46(6):637–647
Horrocks L, Farooqui A (2004) Docosahexaenoic acid in the diet: its importance in maintenance and restoration of neural membrane function. Prostaglandins Leukot Essent Fatty Acids 70:361–372
Horrocks LA, Yeo YK (1999) Health benefits of docosahexaenoic acid (DHA). Pharmacol Res 40(3):211–225
Huang J, Aki T, Yokochi T, Nakahara T, Honda D, Kawamoto S, Shigeta S, Ono K, Suzuki O (2003) Grouping newly isolated docosahexaenoic acid-producing thraustochytrids based on their polyunsaturated fatty acid profiles and comparative analysis of 18S rRNA genes. Mar Biotechnol 5(450):450–457
Iida I, Nakahara T, Yokochi T, Kamisaka Y, Yagi H, Yamaoka M, Suzuki O (1996) Improvement of docosahexaenoic acid production in a culture of Thraustochytrium aureum by medium optimization. J Ferment Bioeng 81(1):76–78
Jain R, Raghukumar S, Sambaiah K, Kumon Y, Nakahara T (2007) Docosahexaenoic acid accumulation in thraustochytrids: search for the rationale. Mar Biol 151(5):1657–1664
Jiang Y, Fan KW, Tsz-YeungWong R, Chen F (2004) Fatty acid composition and squalene content of the marine microalga Schizochytrium mangrovei. J Agric Food Chem 52(5):1196–1200
Johnson EA, An G-H (1991) Astaxanthin from microbial sources. Crit Rev Biotechnol 11(4):297–326
Johnson MB, Wen Z (2009) Production of biodiesel fuel from the microalga Schizochytrium limacinum by direct transesterification of algal biomass. Energy Fuel 23(10):5179–5183
Knothe G (2007) Some aspects of biodiesel oxidative stability. Fuel Process Technol 88(7):669–677
Kris-Etherton PM, Hecker KD, Binkoski AE (2004) Polyunsaturated fatty acids and cardiovascular health. Nutr Rev 62(11):414–426
Leander CA, Porter D, Leander BS (2004) Comparative morphology and molecular phylogeny of aplanochytrids (Labyrinthulomycota). Eur J Protistol 40(4):317–328
Leańo EM, Gapasin RSJ, Polohan B, Vrijmoed LLP (2003) Growth and fatty acid production of thraustochytrids from Panay mangroves, Philippines. Fungal Diversity 12:111–122
Lee Chang KJ, Mansour MP, Dunstan GA, Blackburn SI, Koutoulis A, Nichols PD (2011) Odd-chain polyunsaturated fatty acids in thraustochytrids. Phytochem 72(11–12):1460–1465
Lewis TE, Nichols PD, McMeekin TA (1999) The biological potential of thraustochytrids. Mar Biotechnol 1:580–587
Lewis TE, Nichols PD, McMeekin TA (2001) Sterol and squalene content of a DHA-producing thraustochytrid: influence of culture age, temperature and dissolved oxygen. Mar Biotechnol 3(5):439–447
Lippmeier J, Crawford K, Owen C, Rivas A, Metz J, Apt K (2009) Characterization of both polyunsaturated fatty acid biosynthetic pathways in Schizochytrium sp. Lipids 44(7):621–630
Lorenz RT, Cysewski GR (2000) Commercial potential for Haematococcus microalgae as a natural source of astaxanthin. Trends Biotechnol 4:160–167
Ludwig W, Strunk O, Westram R, Richter L, Meier H, Yadhukumar BA, Lai T, Steppi S, Jobb G, Förster W, Brettske I, Gerber S, Ginhart AW, Gross O, Grumann S, Hermann S, Jost R, König A, Liss T, Lüßmann R, May M, Nonhoff B, Reichel B, Strehlow R, Stamatakis A, Stuckmann N, Vilbig A, Lenke M, Ludwig T, Bode A, Schleifer KH (2004) ARB: a software environment for sequence data. Nucleic Acids Res 32(4):1363–1371
Maas PAY, Kleinschuster SJ, Dykstra MJ, Smolowitz R, Parent J (1999) Molecular characterization of QPX (quahog parasite unknown), a pathogen of Mercenaria mercenaria. J Shellfish Res 18(2):561–567
Metz JG, Roessler P, Facciotti D, Levering C, Dittrich F, Lassner M, Valentine R, Lardizabal K, Domergue F, Yamada A, Yazawa K, Knauf V, Browse J (2001) Production of polyunsaturated fatty acids by polyketide synthases in both prokaryotes and eukaryotes. Science 293(5528):290–293
Mo C, Douek J, Rinkevich B (2002) Development of a PCR strategy for thraustochytrid identification based on 18S rDNA sequence. Mar Biol 140(5):883–889
Monyem A, Canakci M, Van Gerpen JH (2000) Investigation of biodiesel thermal stability under simulated in-use conditions. Appl Eng Agric 16(4):373–378
Pauly D, Christensen V, Guenette S, Pitcher TJ, Sumaila UR, Walters CJ, Watson R, Zeller D (2002) Towards sustainability in world fisheries. Nature 418(6898):689–695
Porter D (1990) Phylum Labyrinthulomycota. In: Margulis L, Corliss JO, Melkonian M, Chapman DJ (eds) Handbook of protoctista. Jones and Bartlett, Boston, pp 388–398
Pruesse E, Quast C, Knittel K, Fuchs BM, Ludwig W, Peplies J, Glöckner FO (2007) SILVA: a comprehensive online resource for quality checked and aligned ribosomal RNA sequence data compatible with ARB. Nucleic Acids Res 35(21):7188–7196
Raghukumar S (1988) Schizochytrium mangrovei sp. nov., a thraustochytrid from mangroves in India. Transcr British Mycol Soc 90(4):627–631
Raghukumar S (1992) Bactivory: a novel dual role for thraustochytrids in the sea. Mar Biol 113:165–169
Raghukumar S (2002) Ecology of the marine portists, the Labyrinthulomycetes (thraustochytrids and labyrinthulids). Eur J Protistol 38(2):127–145
Ratledge C (2004) Fatty acid biosynthesis in microorganisms being used for single cell oil production. Biochimie 86(11):807–815
Rosa S, Galvagno M, Vélez C (2010) Adjusting culture conditions to isolate thraustochytrids from temperate and cold environments in southern Argentina. Mycoscience:1-11
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol 4(4):406
Siboni N, Rasoulouniriana D, Ben-Dov E, Kramarsky-Winter E, Sivan A, Loya Y, Hoegh-Guldberg O, Kushmaro A (2010) Stramenopile microorganisms associated with the massive coral Favia sp. J Eukaryot Microbiol 57(3):236–244
Smith TJ (2000) Squalene: potential chemopreventive agent. Expert Opin Investig Drugs 9(8):1841–1848
Takahata K, Monobe K, Tada M, Weber PC (1998) The benefits and risks of n-3 polyunsaturated fatty acids. Biosci Biotechnol Biochem 62(11):2079–2085
Tapiero H, Ba GN, Couvreur P, Tew KD (2002) Polyunsaturated fatty acids (PUFA) and eicosanoids in human health and pathologies. Biomed Pharmacother 56:215–222
Van Heukelem L, Thomas CS (2001) Computer-assisted high-performance liquid chromatography method development with applications to the isolation and analysis of phytoplankton pigments. J Chromatogr 910(1):31-49
Volkman JK, Barrett SM, Blackburn SI, Mansour MP, Sikes EL, Gelin F (1998) Microalgal biomarkers: a review of recent research developments. Org Geochem 29(5–7):1163–1179
Yamamura R, Shimomura Y (1997) Industrial high-performance liquid chromatography purification of docosahexaenoic acid ethyl ester and docosapentaenoic acid ethyl ester from single-cell oil. J Am Oil Chem Soc 74(11):1435–1440
Yang H-L, Lu C-K, Chen S-F, Chen Y-M, Chen Y-M (2010) Isolation and characterization of Taiwanese heterotrophic microalgae: screening of strains for docosahexaenoic acid (DHA) production. Mar Biotechnol 12(2):173–185
Yokochi T, Honda D, Higashihara T, Nakahara T (1998) Optimization of docosahexaenoic acid production by Schizochytrium limacinum SR21. Appl Microbiol Biotechnol 49(1):72–76
Yokoyama R, Honda D (2007) Taxonomic rearrangement of the genus Schizochytrium sensu lato based on morphology, chemotaxonomic characteristics, and 18S rRNA gene phylogeny (Thraustochytriaceae, Labyrinthulomycetes): emendation for Schizochytrium and erection of Aurantiochytrium and Oblongichytrium gen. nov. Mycoscience 48(4):199–211
Yokoyama R, Salleh B, Honda D (2007) Taxonomic rearrangement of the genus Ulkenia sensu lato based on morphology, chemotaxonomical characteristics, and 18S rRNA gene phylogeny (Thraustochytriaceae, Labyrinthulomycetes): emendation for Ulkenia and erection of Botryochytrium, Parietichytrium, and Sicyoidochytrium gen. nov. Mycoscience 48(6):329–341
Acknowledgments
The authors thank the CSIRO Energy Transformed Flagship and Food Futures Flagship for their support, Dion Frampton and Rob Gurney for assistance during sample collection and cooperation, Ian Jameson for epifluorescence microscope operation, Malcolm Brown for comments on an earlier version of this manuscript, and the helpful comments from two anonymous journal referees and Dr. Claudia Schmidt-Dannert. Kim Jye Lee Chang was supported by an Australian Postgraduate Award and a CSIRO Office of the Chief Executive top-up scholarship through the CSIRO Energy Transformed Flagship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee Chang, K.J., Dunstan, G.A., Abell, G.C.J. et al. Biodiscovery of new Australian thraustochytrids for production of biodiesel and long-chain omega-3 oils. Appl Microbiol Biotechnol 93, 2215–2231 (2012). https://doi.org/10.1007/s00253-011-3856-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-011-3856-4