Abstract
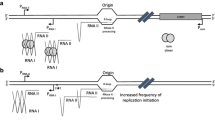
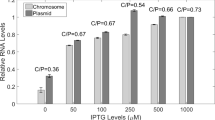
ColE1-like plasmids are widely used as expression vectors and as gene delivery vehicles. We have recently described a naturally occurring plasmid deletion phenomenon in the ColE1-type plasmid, pCI-neo, which leads to the detectable expression of an apparently promotorless kanamycin resistance gene. In the current work, we found that the expression of that aminoglycoside phosphotransferase (aph) gene is regulated by an RNAII preprimer promoter located within the plasmid ColE1 replication origin, as a consequence of the extension of the RNA II species for at least 1.5 kb, up to the aph gene. This mechanism is dependent on the nonformation and/or dissociation of the hybrid between plasmid DNA and RNA II preprimer transcript. This is the first in vivo description of RNA II transcription beyond ori in wild-type Escherichia coli strains and nonmutated RNAII plasmid sequences resulting in productive transcription of distant downstream genes. Our results raise questions about unwanted expression of genes from expression or cloning vectors of ColE1 type and highlight the importance of a more careful design of ColE1-derived plasmid vectors.



Similar content being viewed by others
References
Actis LA, Tolmasky ME, Crosa JH (1999) Bacterial plasmids: replication of extrachromosomal genetic elements encoding resistance to antimicrobial compounds. Front Biosci 4:D43–D62
Ambrazi M, Fellas G, Kapetaniou E, Kotsifaki D, Providak IM, Kokkinidis M (2008) Purification, crystallization and preliminary X-ray diffraction analysis of a variant of the ColE1 Rop protein. Acta Crystallogr Sect F Struct Biol Cryst Commun 1:432–434
Clewel DB (1972) Nature of ColE1 plasmid replication in the presence of chloramphenicol. J Bacteriol 110:667–676
del Solar G, Giraldo R, Ruiz-Echevarría MJ, Espinosa M, Díaz-Orejas R (1998) Replication and control of circular bacterial plasmids. Microbiol Mol Biol Rev 62:434–464
Di Primo C (2008) Real time analysis of the RNAI-RNAII-Rop complex by surface plasmon resonance: from a decaying surface to a standard kinetic analysis. J Mol Recogn 21:37–45
Ederth J, Isaksson LA, Abdulkarim F (2002) Origin-specific reduction of ColE1 plasmid copy number due to mutations in a distinct region of the Escherichia coli RNA polymerase. Mol Genet Genom 267:587–592
Gowrishankar J, Harinarayanan R (2004) Why is transcription coupled to translation in bacteria? Mol Microbiol 54:598–603
Grabherr R, Bayer K (2002) Impact of targeted vector design on ColE1 plasmid replication. Trends Biotechnol 20:257–260
Harinarayanan R, Gowrishankar J (2003) Host factor titration by chromosomal R-loops as a mechanism for runaway plasmid replication in transcription termination-defective mutants of Escherichia coli. J Mol Biol 332:31–46
Inoue N, Uchida H (1991) Transcription initiation of ColE1 replication in Escherichia coli K-12. J Bacteriol 173:1208–1214
Itoh T, Tomizawa JI (1980) Formation of an RNA primer for initiation of replication of ColE1 DNA by ribonuclease H. Proc Natl Acad Sci USA 77:2450–2454
Kim D, Rhee Y, Rhodes D, Sharma V, Sorenson O, Greener A, Smider V (2005) Directed evolution and identification of control regions of ColE1 plasmid replication origins using only nucleotide deletions. J Mol Biol 351:763–775
Kües U, Stahl U (1989) Replication of plasmids in gram-negative bacteria. Microbiol Rev 53:491–516
Maniatis T, Fritsch E, Sambrook J (1982) Molecular cloning. A laboratory manual. Cold Spring Harbor, New York
Masukata H, Dasgupta S, Tomizawa J (1987) Transcriptional activation of ColE1 DNA synthesis by displacement of the nontranscribed strand. Cell 51:1123–1130
Naito S, Uchida H (1980) Initiation of DNA replication in a ColE1-type plasmid: isolation of mutations in the ori region. Proc Natl Acad Sci USA 77:6744–6748
Parada CA, Marians KJ (1989) Transcriptional activation of pBR322 can lead to duplex DNA unwinding catalysed by the Escherichia coli preprisome. J Biol Chem 264:15120–15129
Polaczeck P, Ciesla Z (1984) Effect of altered efficiency of the RNA I and RNA II promoters on in vivo replication of ColE1-like plasmids in Escherichia coli. Mol Gen Genet 194:227–231
Ribeiro S, Oliveira P, Prazeres D, Monteiro G (2008) High frequency plasmid recombination mediated by 28 bp direct repeats. Mol Biotechnol 40:252–260
Struble E, Ladner J, Brabazon D, Marino J (2008) New crystal structures of ColE1 Rom and variants resulting from mutation of a surface exposed residue: implications for RNA-recognition. Proteins 72:761–768
Tomizawa J, Masukata H (1987) Factor-independent termination of transcription in a stretch of deoxyadenosine residues in the template DNA. Cell 51:623–630
Tomizawa J, Itoh T, Selzer G, Som T (1981) Inhibition of ColE1 RNA primer formation by a plasmid-specified small RNA. Proc Natl Acad Sci USA 78:1421–1425
Uptain SM, Chamberlin MJ (1997) Escherichia coli RNA polymerase terminates transcription efficiently at rho-independent terminators on single stranded DNA templates. Proc Natl Acad Sci USA 94:13548–13553
Wang Z, Yuan Z, Hengge UR (2004) Processing of plasmid DNA with ColE1-like replication origin. Plasmid 51:149–161
Wegrzyn G, Wegrzyn A (2008) Is tRNA only a translation factor or also a regulator of other processes? J Appl Genet 49:115–122
Williams JA, Carnes AE, Hodgson CP (2009) Plasmid DNA vaccine vector design: impact on efficacy, safety and upstream production. Biotechnol Adv 27:353–370
Xu FF, Gaggero C, Cohen SN (2002) Polyadenylation can regulate ColE1 type plasmid copy number independently of any effect on RNAI decay by decreasing the interaction of antisense RNAI with its RNAII target. Plasmid 48:49–58
Acknowledgments
This work was funded by Fundação para a Ciência e a Tecnologia, Portugal through project grant (POCTI/BIO/32875/2000) and a PhD grant awarded to S.C. Ribeiro (BD/2957/2000).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ribeiro, S.C., Prazeres, D.M.F. & Monteiro, G.A. Evidence for the in vivo expression of a distant downstream gene under the control of ColE1 replication origin. Appl Microbiol Biotechnol 86, 671–679 (2010). https://doi.org/10.1007/s00253-009-2339-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-009-2339-3