Abstract
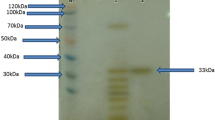
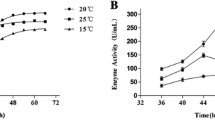
A new alkaline protease (AcpII) was purified from a culture of the deep-sea bacterium Alkalimonas collagenimarina AC40T. AcpII degraded collagen three times faster than it degraded casein. The optimal pH was 8.5–9, and the optimal temperature was 45°C for the degradation of collagen. AcpII was completely inhibited by phenylmethylsulfonyl fluoride and partially by EDTA. Cloning and sequencing the gene for AcpII revealed a 2,283-bp open reading frame encoding a protein of 760 amino acids. AcpII comprises a prepropeptide, a catalytic domain that includes a protease-associated domain (PA domain), and tandem repeat prepeptidase C-terminal domains. To elucidate the role of the PA domain of AcpII, we constructed genes for two enzyme derivatives that possessed the catalytic domains with or without the PA domain and expressed them in Escherichia coli. The derivative without the PA domain showed increased specific activities toward all proteinaceous substrates tested, including gelatin, casein, and collagen, compared with those of the derivative with the PA domain.




Similar content being viewed by others
References
Brown CK, Gu ZY, Matsuka YV, Purushothaman SS, Winter LA, Cleary PP, Olmsted SB, Ohlendorf DH, Earhart CA (2005) Structure of the streptococcal cell wall C5a peptidase. Proc Natl Acad Sci USA 102:18391–18396
Bruinenberg PG, Doesburg P, Alting AC, Exterkate FA, de Vos WM, Siezen RJ (1994) Evidence for a large dispensable segment in the subtilisin-like catalytic domain of the Lactococcus lactis cell-envelope proteinase. Protein Eng 7:991–996
Fu X, Inouye M, Shinde U (2000) Folding pathway mediated by an intramolecular chaperone. The inhibitory and chaperone functions of the subtilisin propeptide are not obligatorily linked. J Biol Chem 275:16871–16878
Helland R, Larsen AN, Smalas AO, Willassen NP (2006) The 1.8 Å crystal structure of a proteinase K-like enzyme from a psychrotroph Serratia species. Febs J 273:61–71
Ishikawa H, Amasaki H, Dohguchi H, Furuya A, Suzuki K (1999) Immunohistological distributions of fibronectin, tenascin, type I, III and IV collagens, and laminin during tooth development and degeneration in fetuses of minke whale, Balaenoptera acutorostrata. J Vet Med Sci 61:227–232
Itoi Y, Horinaka M, Tsujimoto Y, Matsui H, Watanabe K (2006) Characteristic features in the structure and collagen-binding ability of a thermophilic collagenolytic protease from the thermophile Geobacillus collagenovorans MO-1. J Bacteriol 188:6572–6579
Kang S, Jang Y, Choi Y, Kong J (2005) Purification and properties of a collagenolytic protease produced by marine bacterium Vibrio vulnificus CYK279H. Biotechnol Bioprocess Eng 10:593–598
Kim DW, Matsuzawa H (2000) Requirement for the COOH-terminal pro-sequence in the translocation of aqualysin I across the cytoplasmic membrane in Escherichia coli. Biochem Biophys Res Commun 277:216–220
Kim DW, Lee YC, Matsuzawa H (1997) Role of the COOH-terminal pro-sequence of aqualysin I (a heat-stable serine protease) in its extracellular secretion by Thermus thermophilus. FEMS Microbiol Lett 157:39–45
Kulakova L, Galkin A, Kurihara T, Yoshimura T, Esaki N (1999) Cold-active serine alkaline protease from the psychrotrophic bacterium Shewanella strain ac10: gene cloning and enzyme purification and characterization. Appl Environ Microbiol 65:611–617
Kurata A, Miyazaki M, Kobayashi T, Nogi Y, Horikoshi K (2007a) Alkalimonas collagenimarina sp. nov., a psychrotolerant, obligate alkaliphile isolated from deep-sea sediment. Int J Syst Evol Microbiol 57:1549–1553
Kurata A, Uchimura K, Shimamura S, Kobayashi T, Horikoshi K (2007b) Nucleotide and deduced amino acid sequences of a subtilisin-like serine protease from a deep-sea bacterium, Alkalimonas collagenimarina AC40(T). Appl Microbiol Biotechnol 77:311–319
Kwon Y, Kim J, Moon S, Lee H, Rho H (1994) Extracellular alkaline proteases from alkalophilic Vibrio metschnikovii strain RH530. Biotechnol Lett 16:413–418
Kwon YT, Kim JO, Moon SY, Yoo YD, Rho HM (1995) Cloning and characterization of the gene encoding an extracellular alkaline serine protease from Vibrio metschnikovii strain RH530. Gene 152:59–63
Le Guellec D, Morvan-Dubois G, Sire JY (2004) Skin development in bony fish with particular emphasis on collagen deposition in the dermis of the zebrafish (Danio rerio). Int J Dev Biol 48:217–231
Lee YC, Koike H, Taguchi H, Ohta T, Matsuzawa H (1994) Requirement of a COOH-terminal pro-sequence for the extracellular secretion of aqualysin I (a thermophilic subtilisin-type protease) in Thermus thermophilus. FEMS Microbiol Lett 120:69–74
Luo X, Hofmann K (2001) The protease-associated domain: a homology domain associated with multiple classes of proteases. Trends Biochem Sci 26:147–148
Mahon P, Bateman A (2000) The PA domain: a protease-associated domain. Protein Sci 9:1930–1934
Minakuchi Y, Takeshita F, Kosaka N, Sasaki H, Yamamoto Y, Kouno M, Honma K, Nagahara S, Hanai K, Sano A, Kato T, Terada M, Ochiya T (2004) Atelocollagen-mediated synthetic small interfering RNA delivery for effective gene silencing in vitro and in vivo. Nucleic Acids Res 32:e109
Miyamoto K, Tsujibo H, Nukui E, Itoh H, Kaidzu Y, Inamori Y (2002) Isolation and characterization of the genes encoding two metalloproteases (MprI and MprII) from a marine bacterium, Alteromonas sp. strain O-7. Biosci Biotechnol Biochem 66:416–421
Neidhart DJ, Petsko GA (1988) The refined crystal structure of subtilisin Carlsberg at 2.5 Å resolution. Protein Eng 2:271–276
Okamoto M, Yonejima Y, Tsujimoto Y, Suzuki Y, Watanabe K (2001) A thermostable collagenolytic protease with a very large molecular mass produced by thermophilic Bacillus sp. strain MO-1. Appl Microbiol Biotechnol 57:103–108
Semenova SA, Rudenskaya GN, Lyutova LV, Nikitina OA (2008) Isolation and properties of collagenolytic serine proteinase isoenzyme from King Crab Paralithodes camtschatica. Biochemistry (Mosc) 73:1125–1133
Siezen RJ, Leunissen JA (1997) Subtilases: the superfamily of subtilisin-like serine proteases. Protein Sci 6:501–523
Toyoshima T, Matsushita O, Minami J, Nishi N, Okabe A, Itano T (2001) Collagen-binding domain of a Clostridium histolyticum collagenase exhibits a broad substrate spectrum both in vitro and in vivo. Connect Tissue Res 42:281–290
Tsujibo H, Miyamoto K, Tanaka K, Kawai M, Tainaka K, Imada C, Okami Y, Inamori Y (1993) Cloning and sequence of an alkaline serine protease-encoding gene from the marine bacterium Alteromonas sp. strain O-7. Gene 136:247–251
Tsujibo H, Miyamoto K, Tanaka K, Kaidzu Y, Imada C, Okami Y, Inamori Y (1996) Cloning and sequence analysis of a protease-encoding gene from the marine bacterium Alteromonas sp. strain O-7. Biosci Biotechnol Biochem 60:1284–1288
Tsuruoka N, Nakayama T, Ashida M, Hemmi H, Nakao M, Minakata H, Oyama H, Oda K, Nishino T (2003) Collagenolytic serine-carboxyl proteinase from Alicyclobacillus sendaiensis strain NTAP-1: purification, characterization, gene cloning, and heterologous expression. Appl Environ Microbiol 69:162–169
Wandersman C (1989) Secretion, processing and activation of bacterial extracellular proteases. Mol Microbiol 3:1825–1831
Watanabe K (2004) Collagenolytic proteases from bacteria. Appl Microbiol Biotechnol 63:520–526
Yeats C, Bentley S, Bateman A (2003) New knowledge from old: in silico discovery of novel protein domains in Streptomyces coelicolor. BMC Microbiol 3:3
Zhao GY, Chen XL, Zhao HL, Xie BB, Zhou BC, Zhang YZ (2008) Hydrolysis of insoluble collagen by deseasin MCP-01 from deep-sea Pseudoalteromonas sp. SM9913. J Biol Chem 283:36100–36107
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kurata, A., Uchimura, K., Kobayashi, T. et al. Collagenolytic subtilisin-like protease from the deep-sea bacterium Alkalimonas collagenimarina AC40T . Appl Microbiol Biotechnol 86, 589–598 (2010). https://doi.org/10.1007/s00253-009-2324-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-009-2324-x