Abstract
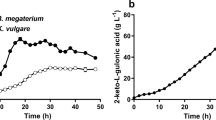
The fermentation process of 2-keto-L-gulonic acid (2KGA) from L-sorbose was developed using a two-stage continuous fermentation system. The mixed culture of Ketogulonicigenium vulgare DSM 4025 and Bacillus megaterium DSM 4026 produced 90 g/L of 2KGA from 120 g/L of L-sorbose at the dilution rate of 0.01 h−1 in a single-stage continuous fermentation process. But after the production period was beyond 150 h, the significant decrease of 2KGA productivity was observed. When the non-spore forming bacteria Xanthomonas maltophilia IFO 12692 was used instead of B. megaterium DSM 4026 as a partner strain for K. vulgare DSM 4025, the 2KGA productivity was significantly improved in a two-stage continuous culture mode, in which two fermentors of the same size and volume were connected in series. In this mode, with two sets of 3-L jar fermentors, the steady state could be continued to over 1,331.5 h at least, when the dilution rates were 0.0382 h−1 and 0.0380 hour−1, respectively, for the first and second fermentors. The overall productivity was calculated to be 2.15 g/L/h at 113.1 g/L and a molar conversion yield of 90.1%. In the up-scaling fermentation to 30-L jar fermentors, 118.5 g/L of 2KGA was produced when dilution rates in both stages were 0.0430 hour−1, and the overall productivity was calculated to be 2.55 g/L/h.










Similar content being viewed by others
References
Afshar AS, Biebl H, Schaller K, Schügerl K (1985) Production of acetone and butanol by Clostridium acetobutylicum in continuous culture with cell recycle. Appl Microbiol Biotechnol 22:394–398
Aiba S, Humphrey AE, Millis NF (1984) Biochemical engineering, 2nd edn. University of Tokyo Press and Academic Press, Tokyo, pp 139–172
Bahl H, Andersch W, Braun K, Gottschalk G (1982) Effect of pH and butyrate concentration on the production of acetone and butanol by Clostridium acetobutylicum grown in continuous culture. Eur J Appl Microbiol Biotechnol 14:17–20
Godin C, Engasser JM (1988) Improved stability of the continuous production of acetone–butanol by Clostridium acetobutylicum in a two-stage process. Biotechnol Lett 10:389–392
Godin C, Engasser JM (1989) Acid in the first stage is a determinant factor for the solvent production in the two-stage continuous fermentation Clostridium acetobutylicum. Biotechnol Lett 11:903–906
Godin C, Engasser JM (1990) Two-stage continuous fermentation of Clostridium acetobutylicum: effects of pH and dilution rate. Appl Microbiol Biotechnol 33:269–273
Goto S, Kitai A, Ozaki A (1973) Continuous yeast cell production from ethanol with a multi-stage tower fermentor. J Ferment Technol 51:582–593
Hoshino T, Sugisawa T, Tazoe M, Shinjoh M, Fujiwara A (1990) Metabolic pathway for 2-keto-L-gulonic acid formation from L-sorbose and d-sorbitol by Gluconobacter melanogenes IFO 3293. Agric Biol Chem 54:1211–1218
Hoshino T, Sugisawa T, Takagi Y (2004) Production of 2-KGA. International publication number WO2004/029265 A2
Imanaka T, Kaieda T, Taguchi H (1973) Optimization of α-galactosidase production in multi-stage continuous culture of mold. J Ferment Technol 51:431–439
Maiorella B, Wike CR, Blanch HW (1981) Alcohol production and recovery. Adv Biochem Eng 20:43–92
Mori A (1993) Industrial application of immobilized catalyst. Marcel Dekker Inc, New York, pp 291–313
Mori A, Takemoto H (1994) Better living through innovative biochemical engineering. Department of Chemical Engineering and Bioprocessing Technoly Unit, National University of Singapore, Singapore, pp 474–476
Nishiwaki A (1997) Analysis of a two stage fermentor with cell recycling for continuous acetic acid production. J Ferment Bioeng 83:565–570
Nogami I, Shirafuji H, Oka M, Yamaguchi T (1995) Method for producing 2-keto-L-gulonic acid. United States Patent 5474924
Park YS, Toda K (1992) Multi-stage biofilm reactor for acetic acid production at high concentration. Biotechnol Lett 14:609–612
Reichstein T, Gruessner A, Oppenauer R (1934) Eine ergiebige synthese der L-ascorbinsaeure (C-vitamin). Helv Chim Acta 17:311–328
Stanbury PF, Whitaker A (1987a) Principles of fermentation technology. Pergamon Press, New York. pp 12–18
Stanbury PF, Whitaker A (1987b) Principles of fermentation technology. Pergamon Press, New York. pp 174–176
Sugisawa T, Hoshino T, Masuda S, Nomura S, Setoguchi Y, Tazoe M, Shinjoh M, Someha S, Fujiwara A (1990) Microbial production of 2-keto-L-gulonic acid from L-sorbose and d-sorbitol by Gluconobacter melanogenes. Agric Biol Chem 54:1201–1209
Takagi Y, Sugisawa T, Hoshino T (2009) Continuous 2-keto-L-gulonic acid fermentation from L-sorbose by Ketogulonigenium vulgare DSM 4025. Appl Microbiol Biotechnol 82:1049–1056
Urbance JW, Bratina BJ, Stoddard SF, Schmidt TM (2001) Taxonomic characterization of Ketogulonigenium vulgare gen. nov., sp. nov. and Ketogulonigenium robustum sp. nov., which oxidize L-sorbose to 2-keto-L-gulonic acid. Int J Syst Evol Microbiol 51:1059–1070
Yin G, Tao Z, Yan Z, Ning W, Wang C, Wang S (1990) Fermentation process. United States Patent 4935359
Acknowledgement
The author wishes to thank Dr. Hideki Aoyagi, the associate professor of University of Tsukuba, for stimulating discussions and helpful and valuable advice.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Takagi, Y., Sugisawa, T. & Hoshino, T. Continuous 2-Keto-l-gulonic acid fermentation by mixed culture of Ketogulonicigenium vulgare DSM 4025 and Bacillus megaterium or Xanthomonas maltophilia . Appl Microbiol Biotechnol 86, 469–480 (2010). https://doi.org/10.1007/s00253-009-2312-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-009-2312-1