Abstract
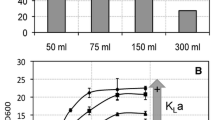
Halomonas boliviensis LC1 is able to accumulate poly(β-hydroxybutyrate) (PHB) under conditions of excess carbon source and depletion of essential nutrients. This study was aimed at an efficient production of PHB by growing H. boliviensis to high cell concentrations in batch cultures. The effect of ammonium, phosphate, and yeast extract concentrations on cell concentration [cell dry weight (CDW)] and PHB content of H. boliviensis cultured in shake flasks was assayed using a factorial design. High concentrations of these nutrients led to increments in cell growth but reduced the PHB content to some extent. Cultivations of H. boliviensis under controlled conditions in a fermentor using 1.5% (w/v) yeast extract as N source, and intermittent addition of sucrose to provide excess C source, resulted in a polymer accumulation of 44 wt.% and 12 g l−1 CDW after 24 h of cultivation. Batch cultures in a fermentor with initial concentrations of 2.5% (w/v) sucrose and 1.5% (w/v) yeast extract, and with induced oxygen limitation, resulted in an optimum PHB accumulation, PHB concentration and CDW of 54 wt.%, 7.7 g l−1 and 14 g l−1, respectively, after 19 h of cultivation. The addition of casaminoacids in the medium increased the CDW to 14.4 g l−1 in 17 h but reduced the PHB content in the cells to 52 wt.%.


Similar content being viewed by others
References
Choi J, Lee SY (1999) Factors affecting the economics of polyhydroxyalkanoate production by bacterial fermentation. Appl Microbiol Biotechnol 51:13–21
Doi Y, Tamaki A, Kunioka M, Soga K (1988) Production of copolyesters of 3-hydroxybutyrate and 3-hydroxyvalerate by Alcaligenes eutrophus from butyric and pentanoic acids. Appl Microbiol Biotechnol 28:330–334
Heinzle E, Lafferty RM (1980) A kinetic model for growth and synthesis of poly-β-hydroxybutyric acid (PHB) in Alcaligenes eutrophus. Eur J Appl Microbiol Biotechnol 11:8–16
Kim BS, Lee SC, Lee SY, Chang HN, Chang YK, Woo SI (1994) Production of poly(3-hydroxybutyric acid) by fed-batch culture of Alcaligenes eutrophus with glucose concentration control. Biotechnol Bioeng 43:892–898
Kunte HJ (2005) K+ transport and its role for osmoregulation in a halophilic member of the Bacteria domain: characterization of the K+ uptake systems from Halomonas elongata. In: Gunde-Cimerman N, Oren A, Plenemitas A (eds) Adaptation to life at high salt concentrations in Archaea, Bacteria, and Eukarya. Springer, Berlin Heidelberg New York, pp 289–300
Lageveen RG, Huisman GW, Preusting H, Ketelaar P, Gerrit E, Witholt B (1988) Formation of polyesters by Pseudomonas oleovorans: effect of substrates on formation and composition of poly-(R)-3-hydroxyalkanoates and poly-(R)-3-hydroxyalkenoates. Appl Environ Microbiol 54:2924–2932
Law JH, Slepecky RA (1961) Assay of poly-β-hydroxybutyric acid. J Bacteriol 82:33–36
Lee SY (1996a) Bacterial polyhydroxyalkanoates. Biotechnol Bioeng 49:1–14
Lee SY (1996b) Plastic bacteria? Progress and prospects for polyhydroxyalkanoate production in bacteria. Trends Biotechnol 14:431–438
Lee SY, Chang HN (1995) Production of poly(3-hydroxybutyric acid) by recombinant Escherichia coli strains: genetic and fermentation studies. Can J Microbiol 14(Suppl 1):207–215
Lee SY, Lee KM, Chang HN, Steinbüchel A (1994) Comparison of recombinant Escherichia coli strains for synthesis and accumulation of poly-(3-hydroxybutyric acid) and morphological changes. Biotechnol Bioeng 44:1337–1347
Lee SY, Wong HH, Choi J, Lee SH, Lee SC, Han CS (2000) Production of medium-chain-length polyhydroxyalkanoates by high-cell-density cultivation of Pseudomonas putida under phosphorus limitation. Biotechnol Bioeng 68:466–470
Lillo JG, Rodriguez-Valera F (1990) Effects of culture conditions on poly(β-hydroxybutyric) acid production by Haloferax mediterranei. Appl Environ Microbiol 56:2517–2521
Oren A (1999) Bioenergetic aspects of halophilism. Microbiol Mol Biol Rev 63:334–348
Page WJ (1992) Production of poly-β-hydroxybutyrate by Azotobacter vinelandii UWD in media containing sugars and complex nitrogen. Appl Microbiol Biotechnol 38:117–121
Page WJ, Cornish A (1993) Growth of Azotobacter vinelandii UWD in fish peptone medium and simplified extraction of poly-β-hydroxybutyrate. Appl Environ Microbiol 59:4236–4244
Quillaguamán J, Hatti-Kaul R, Mattiasson B, Alvarez MT, Delgado O (2004) Halomonas boliviensis sp. nov., an alkalitolerant, moderate halophile bacterium isolated from soil around a Bolivian hypersaline lake. Int J Syst Evol Microbiol 54:721–725
Quillaguamán J, Hashim S, Bento F, Mattiasson B, Hatti-Kaul R (2005) Poly(β-hydroxybutyrate) production by a moderate halophile, Halomonas boliviensis LC1 using starch hydrolysate as substrate. J Appl Microbiol 99:151–157
Quillaguamán J, Delgado O, Mattiasson B, Hatti-Kaul R (2006) Poly(β-hydroxybutyrate) production by a moderate halophile, Halomonas boliviensis LC1. Enzyme Microb Technol 38:148–154
Reddy CSK, Ghai R, Kalia V (2003) Polyhydroxyalkanoates: an overview. Bioresour Technol 87:137–146
Rodriguez-Valera F, Lillo JAG (1992) Halobacteria as producers of polyhydroxyalknoates. FEMS Microbiol Rev 103:181–186
Steinbüchel A, Füchtenbush B (1998) Bacterial and other biological systems for polyester production. Trends Biotechnol 16:419–427
Wang F, Lee SY (1997) Poly(3-hydroxybutyrate) production with high productivity and high polymer content by a fed-batch culture of Alcaligenes latus under nitrogen limitation. Appl Environ Microbiol 63:3703–3706
Acknowledgements
The authors want to express their gratitude to the Swedish International Development Cooperation Agency (Sida) for supporting this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Quillaguamán, J., Muñoz, M., Mattiasson, B. et al. Optimizing conditions for poly(β-hydroxybutyrate) production by Halomonas boliviensis LC1 in batch culture with sucrose as carbon source. Appl Microbiol Biotechnol 74, 981–986 (2007). https://doi.org/10.1007/s00253-006-0754-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-006-0754-2