Abstract
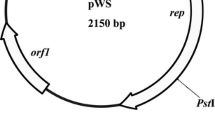
The strain Rhodococcus erythropolis CCM2595, which was shown to degrade phenol, was chosen for genetic studies. To facilitate strain improvement using the methods of gene manipulation, the technique of genetic transfer was introduced and cloning vectors were constructed. Using the plasmid pFAJ2574, an electrotransformation procedure yielding up to 7×104 transformants/μg DNA was optimized. Escherichia coli-R. erythropolis shuttle vectors were constructed using the replicons pSR1 and pGA1 from Corynebacterium glutamicum. The small vector pSRK21 (5.8 kb) provides six unique cloning sites and selection of recombinant clones using α-complementation of β-galactosidase in E. coli. This vector, exhibiting high segregational stability under non-selective conditions in R. erythropolis CCM2595, was applied to cloning and efficient expression of the gene coding for green fluorescent protein (gfpuv).


Similar content being viewed by others
References
Archer JA, Sinskey AJ (1993) The DNA sequence and minimal replicon of the Corynebacterium glutamicum plasmid pSR1: evidence of a common ancestry with plasmids from C. diphtheriae. J Gen Microbiol 139:1753–1759
Chion CK, Duran R, Arnaud A, Galzy P (1991) Cloning vectors and antibiotic-resistance markers for Brevibacterium sp. R312. Gene 105:119–124
De Mot R, Nagy I, De Schrijver A, Pattanapipitpaisal P, Schoofs G, Vanderleyden J (1997) Structural analysis of the 6 kb cryptic plasmid pFAJ2600 from Rhodococcus erythropolis NI86/21 and construction of Escherichia coli-Rhodococcus erythropolis shuttle vectors. Microbiology 143:3137–3147
Denis-Larose C, Bergeron H, Labbe D, Greer CW, Hawari J, Grossman MJ, Sankey BM, Lau PC (1998) Characterization of the basic replicon of Rhodococcus plasmid pSOX and development of a Rhodococcus-Escherichia coli shuttle vector. Appl Environ Microbiol 64:4363–4367
Desomer J, Dhaese P, van Montagu M (1990) Transformation of Rhodococcus fascians by high-voltage electroporation and development of R. fascians cloning vectors. Appl Environ Microbiol 56:2818–2825
Eikmanns BJ, Kleinertz E, Liebl W, Sahm H (1991) A family of Corynebacterium glutamicum/Escherichia coli shuttle vectors for cloning, controlled gene expression, and promoter probing. Gene 102:93–98
Goodfellow M, Alderson G, Chun J (1998) Rhodococcal systematics: problems and developments. Antonie Van Leeuwenhoek 74:3–20
Hashimoto Y, Nishiyama M, Yu F, Watanabe I, Horinouchi S, Beppu T (1992) Development of a host-vector system in a Rhodococcus strain and its use for expression of the cloned nitrile hydratase gene cluster. J Gen Microbiol 138:1003–1010
Haynes JA, Britz ML (1989) Electrotransformation of Brevibacterium lactofermentum and Corynebacterium glutamicum: growth in Tween 80 increases transformation frequencies. FEMS Microbiol Lett 61:329–334
Hughes J, Armitage YC, Symes KC (1998) Application of whole cell rhodococcal biocatalysts in acrylic polymer manufacture. Antonie Van Leeuwenhoek 74:107–118
Kalscheuer R, Arenskotter M, Steinbüchel A (1999) Establishment of a gene transfer system for Rhodococcus opacus PD630 based on electroporation and its application for recombinant biosynthesis of poly(3-hydroxyalkanoic acids). Appl Microbiol Biotechnol 52:508–515
Lang S, Philip JC (1998) Surface-active lipids in rhodococci. Antonie Van Leeuwenhoek 74:59–70
Lazarova V, Manem J (1995) Biofilm characterization and activity analysis in water and wastewater treatment. Water Res 29:2227–2245
Lessard PA, O'Brien XM, Ahlgren NA, Ribich SA, Sinskey AJ (1999) Characterization of IS1676 from Rhodococcus erythropolis SQ1. Appl Microbiol Biotechnol 52:811–819
Liebl W, Bayerl A, Schein B, Stillner U, Schleifer KH (1989) High efficiency electroporation of intact Corynebacterium glutamicum cells. FEMS Microbiol Lett 53:299–303
Meer JR van der, de Vos WM, Harayama S, Zehnder AJ (1992) Molecular mechanisms of genetic adaptation to xenobiotic compounds. Microbiol Rev 56:677–694
Nešvera J, Pátek M, Hochmannová J, Abrhámová Z, Bečvářová V, Jelínková M, Vohradský J (1997) Plasmid pGA1 from Corynebacterium glutamicum codes for a gene product that positively influences plasmid copy number. J Bacteriol 179:1525–1532
Osborn M, Bron S, Firth N, Holsappel, Huddleson A, Kiewiet R, Meijer W, Seegers J, Skurray R, Terpstra P, Thomas CM, Thorsted P, Tietze E, Turner SL (2000). The evolution of bacterial plasmids. In: Thomas CM (ed) The horizontal gene pool. Harwood, Amsterdam, pp 301–361
Pátek M, Eikmanns BJ, Pátek J, Sahm H (1996) Promoters from Corynebacterium glutamicum: cloning, molecular analysis and search for a consensus motif. Microbiology 142:1297–1309
Pridmore RD (1987) New and versatile cloning vectors with kanamycin-resistance marker. Gene 56:309–312
Prieto MB, Hidalgo A, Rodriguez-Fernandez C, Serra JL, Llama MJ (2002) Biodegradation of phenol in synthetic and industrial wastewater by Rhodococcus erythropolis UPV-1 immobilized in an air-stirred reactor with clarifier. Appl Microbiol Biotechnol 58:853–859
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning, a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y.
Sekizaki T, Tanoue T, Osaki M, Shimoji Y, Tsubaki S, Takai S (1998) Improved electroporation of Rhodococcus equi. J Vet Med Sci 60:277–279
Serwold-Davis TM, Groman N, Rabin M (1987) Transformation of Corynebacterium diphtheriae, Corynebacterium ulcerans, Corynebacterium glutamicum, and Escherichia coli with the C. diphtheriae plasmid pNG2. Proc Natl Acad Sci USA 84:4964–4968
Singer ME, Finnerty WR (1988) Construction of an Escherichia coli-Rhodococcus shuttle vector and plasmid transformation in Rhodococcus spp. J Bacteriol 170:638–645
Soda S, Ike M, Fujita M (1998) Effects of inoculation of a genetically engineered bacterium on performance and indigenous bacteria of a sequencing batch activated sludge process treating phenol. J Ferment Bioeng 86:90–96
Vašicová P, Abrhámová Z, Nešvera J, Pátek M, Sahm H, Eikmanns B (1998) Integrating and autonomously replicating vectors for analysis of promoters in Corynebacterium glutamicum. Biotechnol Tech 12:743–746
Venkova-Canova T, Pátek M, Nešvera J (2003) Control of rep gene expression in the plasmid pGA1 from Corynebacterium glutamicum. J Bacteriol (in press)
Acknowledgements
We thank R. De Mot for the gift of pFAJ2574 and R. Kalscheuer and A. Steinbüchel for the gift of pNC9503. This work was supported by grant 526/01/0177 from the Grant Agency of the Czech Republic and by Institutional Research Concept no. AV0Z5020903.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Veselý, M., Pátek, M., Nešvera, J. et al. Host-vector system for phenol-degrading Rhodococcus erythropolis based on Corynebacterium plasmids. Appl Microbiol Biotechnol 61, 523–527 (2003). https://doi.org/10.1007/s00253-003-1230-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-003-1230-x