Abstract
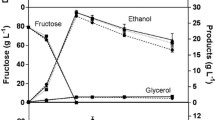
There is a growing consumer demand for wines containing lower levels of alcohol and chemical preservatives. The objectives of this study were to express the Aspergillus niger gene encoding a glucose oxidase (GOX; β-d-glucose:oxygen oxidoreductase, EC 1.1.3.4) in Saccharomyces cerevisiae and to evaluate the transformants for lower alcohol production and inhibition of wine spoilage organisms, such as acetic acid bacteria and lactic acid bacteria, during fermentation. The A. niger structural glucose oxidase (gox) gene was cloned into an integration vector (YIp5) containing the yeast mating pheromone α-factor secretion signal (MFα1 S ) and the phosphoglycerate-kinase-1 gene promoter (PGK1 P ) and terminator (PGK1 T ). The PGK1 P -MFα1 S -gox-PGK1 T cassette (designated GOX1) was introduced into a laboratory strain (Σ1278) of S. cerevisiae. Yeast transformants were analysed for the production of biologically active glucose oxidase on selective agar plates and in liquid assays. The results indicated that the recombinant glucose oxidase was active and was produced beginning early in the exponential growth phase, leading to a stable level in the stationary phase. The yeast transformants also displayed antimicrobial activity in a plate assay against lactic acid bacteria and acetic acid bacteria. This might be explained by the fact that a final product of the GOX enzymatic reaction is hydrogen peroxide, a known antimicrobial agent. Microvinification with the laboratory yeast transformants resulted in wines containing 1.8–2.0% less alcohol. This was probably due to the production of d-glucono-δ-lactone and gluconic acid from glucose by GOX. These results pave the way for the development of wine yeast starter culture strains for the production of wine with reduced levels of chemical preservatives and alcohol.







Similar content being viewed by others
Abbreviations
- GOX:
-
Glucose oxidase enzyme
- gox :
-
Aspergillus niger glucose oxidase gene
- GOX1 :
-
Aspergillus niger gox gene expressed in Saccharomyces cerevisiae as the PGK1 P -MFα1 S -gox-PGK1 T gene cassette
- MFα1 S :
-
Yeast mating pheromone α-factor secretion signal
- PGK1 P :
-
Yeast phosphoglycerate-kinase-1 gene promoter
- PGK1 T :
-
Yeast phosphoglycerate-kinase-1 gene terminator
References
Armstrong GO, Lambrechts MG, Mansvelt EPG, Van Velden DP, Pretorius IS (2001) Wine and health. S Afr J Sci 97:279–282
Bui K, Dick R, Moulin G, Galzy P (1986) A reverse osmosis for the production of low ethanol content wine. Am J Enol Vitic 37:297–300
Chung W, Hancock REW (2000) Action of lysozyme and nisin mixtures against lactic acid bacteria. Int J Food Microbiol 60:25–32
De Baetselier A, Vasavada A, Dohet P, Ha-Thi V, De Beukelaer M, Erpicum T, De Clerck L, Hanotier J, Rosenberg S (1991) Fermentation of a yeast producing A. niger glucose oxidase: scale-up, purification and characterization of the recombinant enzyme. Biotechnology 9:559–561
De Barros Lopes M, Rehman A-U, Gockowiak H, Heinrich AJ, Langridge P, Henschke PA (2000) Fermentation properties of a wine yeast overexpressing the Saccharomyces cerevisiae glycerol 3-phosphate dehydrogenase gene (GPD2). Aust J Grape Wine Res 6:208–215
De Vuyst L, Vandamme EJ (1994) Antimicrobial potential of lactic acid bacteria. In: De Vuyst L, Vandamme EJ (eds) Bacteriocins of lactic acid bacteria. Blackie, New York, pp 99–103
Drysdale GS, Fleet GH (1988) Acetic acid bacteria in winemaking: a review. Am J Enol Vitic 39:143–154
Du Toit M, Pretorius IS (2000) Microbial spoilage and preservation of wine: using weapons from nature's own arsenal a review. S Afr J Enol Vitic 21:74–96
Eglinton JM, Heinrich AJ, Pollnitz AP, Langridge P, Henschke PA, De Barros Lopes M (2002) Decreasing acetic acid accumulation by a glycerol overproducing strain of Saccharomyces cerevisiae by deleting the ALD6 aldehyde dehydrogenase gene. Yeast 19:295–301
Frederick KR, Tung J, Emerick RS, Masiarz FR, Chamberlain SH, Vasavada A, Rosenberg S, Chakraborty S, Schopter LM, Massey V (1990) Glucose oxidase from Aspergillus niger: Cloning, gene sequence, secretion from Saccharomyces cerevisiae and kinetic analysis of a yeast-derived enzyme. J Biol Chem 265:3793–3802
Geisen R (1995) Expression of the Aspergillus niger glucose oxidase gene in Penicillium nalgiovense. World J Microbiol Biotechnol 11:322–325
Geisen R (1999) Inhibition of food-related pathogenic bacteria by god-transformed Penicillium nalgiovense strains. J Food Protect 62:940–943
Gerbaux V, Villa A, Monamy C, Bertrand A (1997) Use of lysozyme to inhibit malolactic fermentation and to stabilize wine after malolactic fermentation. Am J Enol Vitic 48:49–54
Gietz RD, Schiestl RH (1991) Applications of high efficiency lithium acetate transformation of intact yeast cells using single-stranded nucleic acids as carrier. Yeast 3:253–263
Gladstones J (2000) Implications of lowering wine alcohol content. Wine Industry J 15:45–46
Gladstones J, Tomlinson B (1999) A proposal by the Independent Winemakers Association for volumetric taxation based on alcohol content. Wine Industry J 14:92–99
Hammer FE (1998) Oxidoreductases. In: Nagodawithana T, Reed G (eds) Enzymes in food processing. Academic , New York, pp 251–254
Hodgkins M, Mead D, Ballance DJ, Goodey A, Sudbery P (1993) Expression of the glucose oxidase gene from Aspergillus niger in Hansenula polymorpha and its use as a reporter gene to isolate regulatory mutation. Yeast 9:625–635
Hong SY, Choi SK, Lee YH, Pek UH, Jung JK (1998) Overproduction and high level secretion of glucose oxidase in Saccharomyces cerevisiae. Korean J Appl Microbiol Biotechnol 26:68–75
Iland P, Eward A, Sitters J, Markides A, Bruer N (2000) Determination of alcohol by ebulliometry. In: Techniques for chemical analysis and quality monitoring during winemaking. Patrick Iland Wine Promotions, Australia, pp 34–35
Kapat A, Jung J-K, Park Y-H (2001) Enhancement of glucose oxidase production in batch cultivation of recombinant Saccharomyces cerevisiae: optimization of oxygen transfer condition. J Appl Microbiol 90:216–222
Lee Y-H, Lin T-I (1996) Determination of carbohydrates by high-performance capillary electrophoresis with indirect absorbance detection. J Chrom B 681:87–97
Mermelstein NH (2000) Removing alcohol from wine. Food Technol 54:89–92
Michnick S, Roustan J-L, Remize F, Barre P, Dequin S (1997) Modulation of glycerol and ethanol yields during alcoholic fermentation in Saccharomyces cerevisiae strains overexpressed or disrupted for GPD1 encoding glycerol 3-phosphate dehydrogenase. Yeast 13:783–793
Nevoigt E, Pilger R, Mast-Gerlach E, Schmidt U, Freihammer S, Eschenbrenner M, Garbe L, Stahl U (2002) Genetic engineering of brewing yeast to reduce the content of ethanol in beer. FEMS Yeast Res 2:225–232
Ohlmeyer DW (1957) Use of glucose oxidase to stabilize beer. Food Technol October, pp 503–507
Park EH, Shin Y-M, Lim Y-Y, Kwon T-H, Kim d-H, Tang M-S (2000) Expression of glucose oxidase by using recombinant yeast. J Biotechnol 81:35–44
Pickering G (1998) The use of enzymes to stabilise colour and flavour in wine—an alternative to SO2. Aust Grapegrower and Winemaker September, pp 101–103
Pickering G, Heatherbell DA (1996) Characterisation of reduced alcohol wine made from glucose oxidase treated must. Food Technol 26:101–107
Pickering GJ, Heatherbell DA, Barnes MF (1998) Optimising glucose conversion in the production of reduced alcohol wine using glucose oxidase. Food Res Int 31:685–692
Pickering GJ, Heatherbell DA, Barnes MF (1999a) The production of reduced-alcohol wine using glucose oxidase treated juice. Part I. Composition. Am J Enol Vitic 50:291–298
Pickering GJ, Heatherbell DA, Barnes MF (1999b) The production of reduced-alcohol wine using glucose oxidase treated juice. Part II. Stability and SO2-binding. Am J Enol Vitic 50:299–306
Pickering GJ, Heatherbell DA, Barnes MF (1999c) The production of reduced-alcohol wine using glucose oxidase treated juice. Part III. Sensory. Am J Enol Vitic 50:307–316
Power J (1998) Enzymes in brewing. In: Nagodawithana T, Reed G (eds) Enzymes in food processing. Academic , New York, pp 455–456
Pretorius IS (2000) Tailoring wine yeast for the new millennium: novel approaches to the ancient art of winemaking. Yeast 16:675–729
Pretorius IS (2003) The genetic improvement of wine yeasts. In: Arora D (ed) Fungal biotechnology. Decker, New York (in press)
Pretorius IS, Bauer FF (2002) Meeting the consumer challenge through genetically customised wine yeast strains. Trends Biotechnol 20:426–432
Radler F (1990a) Possible use of nisin in winemaking. I. Action of nisin against lactic acid bacteria and wine yeasts in solid and liquid media. Am J Enol Vitic 41:1–6
Radler F (1990b) Possible use of nisin in winemaking. II. Experiments to control lactic acid bacteria in the production of wine. Am J Enol Vitic 41:7–11
Remize F, Roustan JL, Sablayrolles JM, Barre P, Dequin S (1999) Glycerol overproduction by engineered Saccharomyces cerevisiae wine yeast strains leads to substantial changes in by-product formation and to a stimulation of fermentation rate in stationary phase. Appl Environ Microbiol 65:143–149
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning: a laboratory manual, 2nd edn. Cold Spring Harbor Laboratory , Cold Spring Harbor, New York
Schoeman H, Vivier MA, Du Toit M, Dicks LMT, Pretorius IS (1999) The development of bacterial yeast strains by expressing the Pediococcus acidilactici pediocin gene (pedA) in Saccharomyces cerevisiae. Yeast 15:647–656
Scudamore-Smith P, Moran J (1997) A growing market for reduced alcohol wines. Wine Ind J 12:165–167
Struhl K, Stincomb DT, Scherer S, Davis RW (1979) High-frequency transformation of yeast: autonomous replication of hybrid DNA molecules. Proc Natl Acad Sci USA 76:1035–1039
Vemulapalli V, Miller KA, Hoseney RC (1998) Glucose oxidase in breadmaking systems. Cereal Chem 75:439–442
Villettaz JC (1987) A new method for the production of low alcohol wines and better balanced wines. Proceedings of Sixth Australian Wine Industry Technical Conference, Australian Industrial Publishers, Adelaide
Vivier MA, Pretorius IS (2002) Genetically tailored grapevines for a wine industry that is sustainable and focussed on quality. Trends Biotechnol 20:472–478
Whittington H, Kerry-Williams S, Bidgood K, Dodsworth N, Peberdy J, Dobson M, Hinchliffe E, Ballance DJ (1990) Expression of the Aspergillus niger glucose oxidase gene in A. niger, A. nidulans and Saccharomyces cerevisiae. Curr Genet 18:531–536
Acknowledgements
The authors wish to thank the South African wine industry (Winetech) and the National Research Foundation (NRF) for funding this study. We would also like to thank Dr. R. Geisen of the Federal Research Centre for Nutrition in Germany for sending us the Aspergillus niger glucose oxidase gene, and Dr. Miranda Waldron of the Electron Microscope Unit at the University of Cape Town for her time and patience with the scanning electron microscopy. In addition, we want to thank Dr. Elisabeth Rohwer of the Institute for Wine Biotechnology at Stellenbosch University, for her time and help with the high-performance capillary electrophoresis system.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Malherbe, D.F., du Toit, M., Cordero Otero, R.R. et al. Expression of the Aspergillus niger glucose oxidase gene in Saccharomyces cerevisiae and its potential applications in wine production. Appl Microbiol Biotechnol 61, 502–511 (2003). https://doi.org/10.1007/s00253-002-1208-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00253-002-1208-0