Abstract
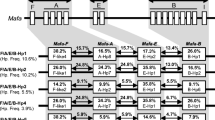
High-frequency alleles and/or co-occurring human leukocyte antigen alleles across loci appear to be more important than individual alleles as markers of disease risk and have clinical value as biomarkers for targeted screening or the development of new disease therapies. To better elucidate the major histocompatibility complex (MHC) background and to facilitate the experimental use of cynomolgus macaques, Mafa-DPA1, Mafa-DQA1, Mafa-DRA, and Mafa-DOA alleles were characterized, and their combinations were investigated in 30 Vietnamese macaques by gene cloning and sequencing. A total of 26 Mafa-DPA1, 18 Mafa-DQA1, 9 Mafa-DRA, and 15 Mafa-DOA alleles, including 7 high-frequency alleles, were identified in this study, respectively. In addition, 15 Mafa-DQA1, 17 Mafa-DPA1, 15 Mafa-DOA, and 2 Mafa-DRA alleles represented novel sequences that had not been documented in earlier studies. Our results also showed that the Vietnamese macaques might be valuable because no less than 30 % of the test animals possessed Mafa-DRA*01:02:01 (90 %), -DQA1*26:01:03 (37 %), -DOA*01:02:07 (34 %), and -DQA1*01:03:03 (30 %). We previously reported that the combinations of MHC class II alleles, including the combination of DOA*01:02:07-DPA1*02:09 and DOA*01:02:07-DQA1*01:03:03, were detected in 17 and 14 % of the animals, respectively. Interestingly, more than two Mafa-DQA1 and Mafa-DPA1 alleles were detected in one animal in this study, which suggested that they might be caused by a chromosomal duplication. If our findings can be validated by other studies, it will further enrich the number of known Mafa-DPA1 and Mafa-DQA1 polymorphisms. Our results identified the co-occurring MHC alleles across loci in a cohort of Vietnamese cynomolgus macaques, which emphasized the value of this species as a model for biomedical research.





Similar content being viewed by others
References
Aarnink A, Estrade L, Apoil PA, Kita YF, Saitou N, Shiina T, Blancher A (2010) Study of cynomolgus monkey (Macaca fascicularis) DRA polymorphism in four populations. Immunogenetics 62:123–136
Bontrop RE, Otting N, de Groot NG, Doxiadis GG (1999) Major histocompatibility complex class II polymorphisms in primates. Immunol Rev 167:339–350
Brown JH, Jardetzky T, Saper MA, Samroui B, Bjorkman PJ, Wiley DC (1998) A hypothetical model of the foreign antigen binding site of class II histocompatibility molecules. Nature 332:845–850
Creager HM, Becker EA, Sandman KK, Karl JA, Lank SM, Bimber BN, Wiseman RW, Hughes AL, O’Connor SL, O’Connor DH (2011) Characterization of full-length MHC class II sequences in Indonesian and Vietnamese cynomolgus macaques. Immunogenetics 63:611–618
de Groot N, Doxiadis GG, De Groot NG, Otting N, Heijmans C, Rouweler AJ, Bontrop RE (2004) Genetic makeup of the DR region in rhesus macaques: gene content, transcripts, and pseudogenes. J Immunol 172:6152–6157
Doxiadis GG, Otting N, de Groot NG, Bontrop RE (2001) Differential evolutionary MHC class II strategies in humans and rhesus macaques: relevance for biomedical studies. Immunol Rev 183:76–85
Doxiadis GG, de Vos-Rouweler AJ, de Groot N, Otting N, Bontrop RE (2012) DR haplotype diversity of the cynomolgus macaque as defined by its transcriptome. Immunogenetics 64:31–37
Florese RH, Wiseman RW, Venzon D, Karl JA, Demberg T, Larsen K, Flanary L, Kalyanaraman VS, Pal R, Titti F, Patterson LJ, Heath MJ, O’Connor DH, Cafaro A, Ensoli B, Robert-Guroff M (2008) Comparative study of Tat vaccine regimens in Mauritian cynomolgus and Indian rhesus macaques: influence of Mauritian MHC haplotypes on susceptibility/resistance to SHIV(89.6P) infection. Vaccine 26:3312–3321
Gyllensten UB, Erlich HA (1988) Generation of single-stranded DNA by the polymerase chain reaction and its application to direct sequencing of the HLA-DQA locus. Proc Natl Acad Sci U S A 85:7652–7656
Horn GT, Bugawan TL, Long DM, Erlich HA (1988) Allelic sequence variation of the HLA-DQ loci: relationship to serology and insulin-dependent diabetes susceptibility. Proc Natl Acad Sci U S A 85:6012–6016
Klein J, Bontrop RE, Dawkins RL, Erlich HA, Gyllensten UB, Heise ER, Jones PP, Parham P, Wakeland EK, Watkins DI (1990) Nomenclature for the major histocompatibility complexes of different species: a proposal. Immunogenetics 31:217–219
Krebs KC, Jin ZY, Rudersdorf R, Hughes AL, O’Connor DH (2005) Unusually high frequency MHC class I alleles in Mauritian origin cynomolgus macaques. J Immunol 175:5230–5239
Ling F, Wei LQ, Wang T, Wang HB, Zhuo M, Du HL, Wang JF, Wang XN (2011) Characterization of the major histocompatibility complex class II DOB, DPB1, and DQB1 alleles in cynomolgus macaques of Vietnamese origin. Immunogenetics 63:155–166
Ling F, Zhuo M, Ni C, Zhang GQ, Wang T, Li W, Wei LQ, Du HL, Wang JF, Wang XN (2012) Comprehensive identification of high-frequency and co-occurring Mafa-B, Mafa-DQB1, and Mafa-DRB alleles in cynomolgus macaques of Vietnamese origin. Hum Immunol 73:547–553
Lith M, Ham W, Neefjes J (2002) Novel polymorphisms in HLA-DOA and HLA-DOB in B-cell malignancies. Immunogenetics 54:591–595
Naruse TK, Kawata H, Anzai T, Takashige N, Kagiya M, Nose Y, Nabeya N, Isshiki G, Tatsumi N, Inoko H (1999) Limited polymorphism in the HLA-DOA gene. Tissue Antigens 53:359–365
O’Connor SL, Blasky AJ, Pendley CJ, Becker EA, Wiseman RW, Karl JA, Hughes AL, O’Connor DH (2007) Comprehensive characterization of MHC class II haplotypes in Mauritian cynomolgus macaques. Immunogenetics 59:449–462
O’Sullivan MG, Anderson DK, Goodrich JA, Tulli H, Green SW, Young NS, Brown KE (1997) Experimental infection of cynomolgus monkeys with simian parvovirus. J Virol 71:4517–4521
Robinson J, Waller MJ, Stoehr P, Marsh SG (2005) IPD—the immuno polymorphism database. Nucleic Acids Res 33:D523–D526
Sano K, Shiina T, Kohara S, Yanagiya K, Hosomichi K, Shimizu S, Anzai T, Watanabe A, Ogasawara K, Torii R, Kulski JK, Inoko H (2006) Novel cynomolgus macaque MHC-DPB1 polymorphisms in three South-East Asian populations. Tissue Antigens 67:297–306
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599
Todd JA, Acha-Orbea H, Bel JI, Chao N, Fronek Z, Jacob CO, McDermott M, Sinha AA, Timmerman L, Steinmen L, Mcdevitt HO (1988) A molecular basis for MHC class II-associated autoimmunity. Science 240:1003–1009
Walsh GP, Tan EV, dela Cruz EC, Abalos RM, Villahermosa LG, Young LJ, Cellona RV, Nazareno JB, Horwitz MA (1996) The Philippine cynomolgus monkey (Macaca fasicularis) provides a new non-human primate model of tuberculosis that resembles human disease. Nat Med 2:430–436
Wiseman RW, Karl JA, Bimber BN, O’Leary CE, Lank SM, Tuscher JJ, Detmer AM, Bouffard P, Levenkova N, Turcotte C, Jr ES, Wright C, Harkims T, O’Connor DH (2009) Major histocompatibility complex genotyping with massively parallel pyrosequencing. Nat Med 15:1322–1326
Acknowledgments
We thank the Primate Research Center of South China for providing blood samples from animals. We thank Dr Natasja de Groot, Dr Nel Otting, and IMGT Non-human Primate Nomenclature Committee for naming the Mafa-DPA1, Mafa-DQA1, Mafa-DRA, and Mafa-DOA sequences. This project was granted by the National Natural Science Foundation of China (31271322), the Natural Science Foundation of Guangdong, China (S2011040005261), and the Fundamental Research Funds for the Central Universities of South China University of Technology (2012ZZ0093).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Ruirui Xiang and Huiling Zhang contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 169 kb)
Rights and permissions
About this article
Cite this article
Xiang, R., Zhang, H., Deng, Q. et al. Comprehensive identification of high-frequency and co-occurring Mafa-DPA1, Mafa-DQA1, Mafa-DRA, and Mafa-DOA alleles in Vietnamese cynomolgus macaques. Immunogenetics 65, 667–674 (2013). https://doi.org/10.1007/s00251-013-0713-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00251-013-0713-x