Abstract
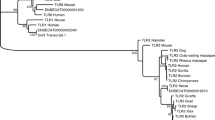
Toll-like receptors 3, 7, and 8 (TLR3, TLR7, and TLR8) were studied in the genomes of the domestic horse and several other mammals. The messenger RNA sequences and exon/intron structures of these TLR genes were determined. An equine bacterial artificial chromosome clone containing the TLR3 gene was assigned by fluorescent in situ hybridization to the horse chromosomal location ECA27q16–q17 and this map location was confirmed using an equine radiation hybrid panel. Direct sequencing revealed 13 single-nucleotide polymorphisms in the coding regions of the equine TLR 3, 7, and 8 genes. Of these polymorphisms, 12 were not previously reported. The allelic frequency was estimated for each single-nucleotide polymorphism from genotyping data obtained for 154 animals from five horse breeds. Some of these frequencies varied significantly among different horse breeds. Domain architecture predictions for the three equine TLR protein sequences revealed several conserved regions within the variable leucine-rich repeats between the corresponding horse and cattle TLR proteins. A phylogenetic analysis did not indicate that any significant exchanges had occurred between paralogous TLR7 and TLR8 genes in 20 vertebrate species analyzed.



Similar content being viewed by others
References
Bagnall RD, Ayres KL, Green PM, Giannelli F (2005) Gene conversion and evolution of Xq28 duplicons involved in recurring inversions causing severe hemophilia A. Genome Res 15:214–223
Baoprasertkul P, Peatman E, Somridhivej B, Liu Z (2006) Toll-like receptor 3 and TICAM genes in catfish: species-specific expression profiles following infection with Edwardsiella ictaluri. Immunogenetics 58:817–830
Cargill EJ, Womack JE (2007) Detection of polymorphisms in bovine toll-like receptors 3, 7, 8, and 9. Genomics 89:745–755
Chuang TH, Ulevitch RJ (2000) Cloning and characterization of a sub-family of human toll-like receptors: hTLR7, hTLR8 and hTLR9. Eur Cytokine Netw 11:372–378
Cook D, Brooks S, Bellone R, Bailey E (2008) Missense mutation in exon 2 of SLC36A1 responsible for champagne dilution in horses. PLoS Genet 4:e1000195
Daffis S, Samuel MA, Suthar MS, Gale M Jr, Diamond MS (2008) Toll-like receptor 3 has a protective role against West Nile virus infection. J Virol 82:10349–10358
Du X, Poltorak A, Wei Y, Beutler B (2000) Three novel mammalian toll-like receptors: gene structure, expression, and evolution. Eur Cytokine Netw 11:362–371
Gay J, Myers S, McVean G (2007) Estimating meiotic gene conversion rates from population genetic data. Genetics 177:881–894
Gowen BB, Hoopes JD, Wong MH, Jung KH, Isakson KC, Alexopoulou L, Flavell RA, Sidwell RW (2006) TLR3 deletion limits mortality and disease severity due to infection. J Immunol 177:6301–6307
Hardarson HS, Baker JS, Yang Z, Purevjav E, Huang CH, Alexopoulou L, Li N, Flavell RA, Bowles NE, Vallejo JG (2007) Toll-like receptor 3 is an essential component of the innate stress response in virus-induced cardiac injury. Am J Physiol Heart Circ Physiol 292:H251–H258
Heil F, Hemmi H, Hochrein H, Ampenberger F, Kirschning C, Akira S, Lipford G, Wagner H, Bauer S (2004) Species-specific recognition of single-stranded RNA via toll-like receptor 7 and 8. Science 303:1526–1529
Hemmi H, Kaisho T, Takeuchi O, Sato S, Sanjo H, Hoshino K, Horiuchi T, Tomizawa H, Takeda K, Akira S (2002) Small anti-viral compounds activate immune cells via the TLR7 MyD88-dependent signaling pathway. Nat Immunol 3:196–200
Hidaka F, Matsuo S, Muta T, Takeshige K, Mizukami T, Nunoi H (2006) A missense mutation of the Toll-like receptor 3 gene in a patient with influenza-associated encephalopathy. Clin Immunol 119:188–194
Hutchens M, Luker KE, Sottile P, Sonstein J, Lukacs NW, Nunez G, Curtis JL, Luker GD (2008) TLR3 increases disease morbidity and mortality from vaccinia infection. J Immunol 180:483–491
Kruithof EK, Satta N, Liu JW, Dunoyer-Geindre S, Fish RJ (2007) Gene conversion limits divergence of mammalian TLR1 and TLR6. BMC Evol Biol 7:148
Le Goffic R, Balloy V, Lagranderie M, Alexopoulou L, Escriou N, Flavell R, Chignard M, Si-Tahar M (2006) Detrimental contribution of the Toll-like receptor (TLR)3 to influenza A virus-induced acute pneumonia. PLoS Pathog 2:e53
Letunic I, Copley RR, Schmidt S, Ciccarelli FD, Doerks T, Schultz J, Ponting CP, Bork P (2004) SMART 4.0: towards genomic data integration. Nucleic Acids Res 32:D142–D144
Li M, Zhou Y, Feng G, Su SB (2009) The critical role of Toll-like receptor signaling pathways in the induction and progression of autoimmune diseases. Curr Mol Med 9:365–374
MacDonald MR, Xia J, Smith AL, Magor KE (2008) The duck toll like receptor 7: genomic organization, expression and function. Mol Immunol 45:2055–2061
Moue M, Tohno M, Shimazu T, Kido T, Aso H, Saito T, Kitazawa H (2008) Toll-like receptor 4 and cytokine expression involved in functional immune response in an originally established porcine intestinal epitheliocyte cell line. Biochim Biophys Acta 1780:134–144
Oh DY, Taube S, Hamouda O, Kucherer C, Poggensee G, Jessen H, Eckert JK, Neumann K, Storek A, Pouliot M, Borgeat P, Oh N, Schreier E, Pruss A, Hattermann K, Schumann RR (2008) A functional toll-like receptor 8 variant is associated with HIV disease restriction. J Infect Dis 198:701–709
Okui Y, Kano R, Maruyama H, Hasegawa A (2008) Cloning of canine Toll-like receptor 7 gene and its expression in dog tissues. Vet Immunol Immunopathol 121:156–160
Perelygin AA, Lear TL, Zharkikh AA, Brinton MA (2005) Structure of equine 2′-5′oligoadenylate synthetase (OAS) gene family and FISH mapping of OAS genes to ECA8p15–>p14 and BTA17q24–>q25. Cytogenet Genome Res 111:51–56
Perelygin AA, Lear TL, Zharkikh AA, Brinton MA (2006) Comparative analysis of vertebrate EIF2AK2 (PKR) genes and assignment of the equine gene to ECA15q24–q25 and the bovine gene to BTA11q12–q15. Genet Sel Evol 38:551–563
Philbin VJ, Iqbal M, Boyd Y, Goodchild MJ, Beal RK, Bumstead N, Young J, Smith AL (2005) Identification and characterization of a functional, alternatively spliced Toll-like receptor 7 (TLR7) and genomic disruption of TLR8 in chickens. Immunology 114:507–521
Raudsepp T, Lee EJ, Kata SR, Brinkmeyer C, Mickelson JR, Skow LC, Womack JE, Chowdhary BP (2004) Exceptional conservation of horse-human gene order on X chromosome revealed by high-resolution radiation hybrid mapping. Proc Natl Acad Sci U S A 101:2386–2391
Raudsepp T, Gustafson-Seabury A, Durkin K, Wagner ML, Goh G, Seabury CM, Brinkmeyer-Langford C, Lee EJ, Agarwala R, Stallknecht-Rice E, Schaffer AA, Skow LC, Tozaki T, Yasue H, Penedo MC, Lyons LA, Khazanehdari KA, Binns MM, MacLeod JN, Distl O, Guerin G, Leeb T, Mickelson JR, Chowdhary BP (2008) A 4,103 marker integrated physical and comparative map of the horse genome. Cytogenet Genome Res 122:28–36
Sang Y, Yang J, Ross CR, Rowland RR, Blecha F (2008) Molecular identification and functional expression of porcine Toll-like receptor (TLR) 3 and TLR7. Vet Immunol Immunopathol 125:162–167
Sanghavi SK, Shankarappa R, Reinhart TA (2004) Genetic analysis of Toll/Interleukin-1 Receptor (TIR) domain sequences from rhesus macaque Toll-like receptors (TLRs) 1–10 reveals high homology to human TLR/TIR sequences. Immunogenetics 56:667–674
Schott E, Witt H, Neumann K, Bergk A, Halangk J, Weich V, Muller T, Puhl G, Wiedenmann B, Berg T (2008) Association of TLR7 single nucleotide polymorphisms with chronic HCV-infection and response to interferon-a-based therapy. J Viral Hepat 15:71–78
Schultz J, Milpetz F, Bork P, Ponting CP (1998) SMART, a simple modular architecture research tool: identification of signaling domains. Proc Natl Acad Sci U S A 95:5857–5864
Tabeta K, Georgel P, Janssen E, Du X, Hoebe K, Crozat K, Mudd S, Shamel L, Sovath S, Goode J, Alexopoulou L, Flavell RA, Beutler B (2004) Toll-like receptors 9 and 3 as essential components of innate immune defense against mouse cytomegalovirus infection. Proc Natl Acad Sci U S A 101:3516–3521
Town T, Bai F, Wang T, Kaplan AT, Qian F, Montgomery RR, Anderson JF, Flavell RA, Fikrig E (2009) Toll-like receptor 7 mitigates lethal West Nile encephalitis via interleukin 23-dependent immune cell infiltration and homing. Immunity 30:242–253
Walsh JB (1987) Sequence-dependent gene conversion: can duplicated genes diverge fast enough to escape conversion? Genetics 117:543–557
Werling D, Piercy J, Coffey TJ (2006) Expression of TOLL-like receptors (TLR) by bovine antigen-presenting cells-potential role in pathogen discrimination? Vet Immunol Immunopathol 112:2–11
Ye S, Dhillon S, Ke X, Collins AR, Day IN (2001) An efficient procedure for genotyping single nucleotide polymorphisms. Nucleic Acids Res 29:E88–E88
Zhang SY, Jouanguy E, Ugolini S, Smahi A, Elain G, Romero P, Segal D, Sancho-Shimizu V, Lorenzo L, Puel A, Picard C, Chapgier A, Plancoulaine S, Titeux M, Cognet C, von Bernuth H, Ku CL, Casrouge A, Zhang XX, Barreiro L, Leonard J, Hamilton C, Lebon P, Heron B, Vallee L, Quintana-Murci L, Hovnanian A, Rozenberg F, Vivier E, Geissmann F, Tardieu M, Abel L, Casanova JL (2007) TLR3 deficiency in patients with herpes simplex encephalitis. Science 317:1522–1527
Zhang Y, Guo Y, Lv K, Wang K, Sun S (2008) Molecular cloning and functional characterization of porcine toll-like receptor 7 involved in recognition of single-stranded RNA virus/ssRNA. Mol Immunol 45:1184–1190
Zhu J, Lai K, Brownile R, Babiuk LA, Mutwiri GK (2008) Porcine TLR8 and TLR7 are both activated by a selective TLR7 ligand, imiquimod. Mol Immunol 45:3238–3243
Acknowledgments
This work was supported by grant CI000216 from the National Center for Infectious Diseases, Centers for Disease Control and Prevention, to MAB and AAP.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary materials
Below is the link to the electronic supplementary material.
Supplemental Table 1
PCR primers and restriction enzymes used to genotype SNPs within the coding regions of the equine TLR3, TLR7, and TLR8 genes (DOC 28 kb)
Supplemental Table 2
PCR primers used to amplify mammalian TLR3 genes (DOC 24 kb)
Supplemental Table 3
PCR primers used to amplify the middle regions of various mammalian TLR7 mRNAs (DOC 24 kb)
Rights and permissions
About this article
Cite this article
Astakhova, N.M., Perelygin, A.A., Zharkikh, A.A. et al. Characterization of equine and other vertebrate TLR3, TLR7, and TLR8 genes. Immunogenetics 61, 529–539 (2009). https://doi.org/10.1007/s00251-009-0381-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00251-009-0381-z