Abstract
We report the discovery of previously unrecognised short consensus repeats (SCRs) within human and chimpanzee CR1 and CR1L. Analysis of available genomic, protein and expression databases suggests that these are actually genomic remnants of SCRs previously reported in other complement control proteins (CCPs). Comparison with the nucleotide motifs of the 11 defined subfamilies of SCRs justifies the designation g-like because of the close similarity to the g subfamily found in CR2 and MCP.
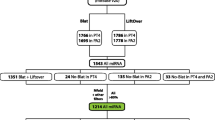
To date, we have identified five such SCRs in human and chimpanzee CR1, one in human and chimpanzee CR1L, but none in either rat or mouse Crry in keeping with the number of internal duplications of the long homologous repeat (LHR) found in CR1 and CR1L. In fact, at the genomic level, the ancestral LHR must have contained eight SCRs rather than seven as previously thought. Since g-like SCRs are found immediately downstream of d SCRs, we suggest that there must have been a functional dg set which has been retained by CR2 and MCP but which is degenerate in CR1 or CR1L. Interestingly, dg is also present in the CR2 component of mouse CR1. The degeneration of the g SCR must have occurred prior to the formation of primate CR1L and prior to the duplication events which resulted in primate CR1. In this context, the apparent conservation of g-like SCRs may be surprising and may suggest the existence of mechanisms unrelated to protein coding.
These results provide examples of the many processes which have contributed to the evolution of the extensive repertoire of CCPs.




Similar content being viewed by others
References
Birmingham DJ (2001) CR1 and CR1-like: the primate immune adherence receptors. Immunol Rev 180:100–111
Dawkins RL, Christiansen FT, Kay PH, Garlepp M, McCluskey J, Hollingsworth PN, Zilko PJ (1983) Disease associations with complotypes, supratypes and haplotypes. Immunol Rev 70:1–22
Dawkins R, Leelayuwat C, Gaudieri S, Tay G, Hui J, Cattley S, Martinez P, Kulski J (1999) Genomics of the major histocompatibility complex: haplotypes, duplication, retroviruses and disease. Immunol Rev 167:275–304
Gaudieri S, Kulski JK, Dawkins RL, Gojobori T (1999) Extensive nucleotide variability within a 370 kb sequence from the central region of the Major Histocompatibility Complex. Gene 238:157–161
Hourcade D, Holers VM, Atkinson JP (1989) The regulators of complement activation (RCA) gene cluster. Adv Immunol 45:381–416
Hourcade D, Liszewski M, Krych-Goldberg M, Atkinson J (2000) Functional domains, structural variations and pathogen interactions of MCP, DAF and CR1. Immunopharmacology 49:103–116
Krushkal J, Bat O, Gigli I (2000) Evolutionary relationships among proteins encoded by the regulator of complement activation gene cluster. Mol Biol Evol 17(11):1718–1730
Kumar S, Tamura K, Jacobsen I, Nei M (2001) MEGA2: molecular evolutionary genetics analysis software. Bioinformatics 17:1244–1245
Leelayuwat C, Abraham LJ, Tabarias H, Christiansen FT, Dawkins RL (1992) Genomic organization of a polymorphic duplicated region centromeric of HLA-B. Immunogenetics 36:208–212
Longman-Jacobsen N, Williamson JF, Dawkins RL, Gaudieri S (2003) In polymorphic genomic regions indels cluster with nucleotide polymorphism: quantum genomics. Gene 312:257–261
McLure CA, Williamson JF, Dawkins RL, Davies R, Berry J, Laird R, Longman-Jacobsen N, Gaudieri S (2004) Amino acid patterns within short consensus repeats define conserved duplicons shared by genes of the RCA complex. J Mol Evol 59(2):143–157
Reid K, Bentley D, Campbell R, Chung L, Sim R, Kristensen T, Tack B (1986) Complement system proteins which interact with C3b or C4b. A superfamily of structurally related proteins. Immunol. Today 7:230–234
Rutherford K, Parkhill J, Crook J, Horsnell T, Rice P, Rajandream M, Barrell B (2000) Artemis: sequence visualisation and annotation. Bioinformatics 16:944–945
Sonnhammer E, Durbin R (1995) A dot-matrix program with dynamic threshold control suited for genomic DNA and protein sequence analysis. Gene 167(1–2):GC1–GC10
Acknowledgements
The authors gratefully acknowledge the support of the Immunogenetics Research Foundation, Genetic Technologies, the National Health and Medical Research Council of Australia, the Australian Research Council and Equitech. C.M. is supported by an APA scholarship
Author information
Authors and Affiliations
Corresponding author
Additional information
Manuscript number 0406 of the Centre for Molecular Immunology and Instrumentation of the University of Western Australia
Rights and permissions
About this article
Cite this article
McLure, C.A., Williamson, J.F., Stewart, B.J. et al. Genomic analysis reveals a duplication of eight rather than seven short consensus repeats in primate CR1 and CR1L: evidence for an additional set shared between CR1 and CR2. Immunogenetics 56, 631–638 (2004). https://doi.org/10.1007/s00251-004-0731-9
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00251-004-0731-9