Abstract
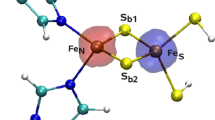
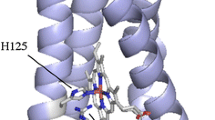
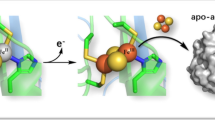
Iron–sulfur centers are widespread in living organisms, mostly performing electron transfer functions, either in electron transfer chains or as part of multi-enzymatic complexes, while being also present in enzyme active sites, handling substrate catalysis. Rubredoxin is the simplest iron–sulfur containing protein constituted by a single polypeptide chain of 50 to 60 amino acids, of which four cysteine residues are responsible for metal binding in a tetrahedral coordination sphere. In this manuscript we explore the structure and stability of both apo- and holo-forms of a Rubredoxin from Marinobacter hydrocarbonoclasticus using Synchrotron Radiation Circular Dichroism (SRCD) in combination with other biochemical and spectroscopic techniques. The results are consistent with a holo-protein form containing a monomeric iron center with UV–visible maxima at 760, 578, 494, 386, 356 and 279 nm, an intense EPR resonance with a g value around 4.3 and Mössbauer spectroscopy parameters of δ equal to 0.69 mm/s and ΔEQ equal to 3.25 mm/s, for the ferrous reconstituted state. SRCD data, obtained for the first time for the apo-form, show a quite defined structure with ∆ε maximum at 191 nm and minima at 203 and 231 nm. Most significantly, the presence of isosbestic points at 189 and 228 nm made the interconversion between the two stable apo- and holo-form solution structures clear. SRCD temperature dependence data shows that for both forms the denaturation process proceeds through an intermediate species.










Similar content being viewed by others
References
Archer M, Carvalho AL, Teixeira S et al (1999) Structural studies by X-ray diffraction on metal substituted desulforedoxin, a rubredoxin-type protein. Protein Sci 8:1536–1545. https://doi.org/10.1110/ps.8.7.1536
Beinert H, Holm RH, Münck E (1997) Iron-sulfur clusters: nature’s modular, multipurpose structures. Science 277:653–659. https://doi.org/10.1126/science.277.5326.653
Benson DA, Cavanaugh M, Clark K et al (2013) GenBank. Nucleic Acids Res 41:36–42. https://doi.org/10.1093/nar/gks1195
Blake PR, Park JB, Adams MWW, Summers MF (1992) Novel observation of NH–S(Cys) hydrogen-bond-mediated scalar coupling in cadmium-113 substituted rubredoxin from Pyrococcus furiosus. J Am Chem Soc 114:4931–4933. https://doi.org/10.1021/ja00038a084
Bonin P, Barbotin JN, Dhulster P, Bertrand JC (1987a) Nitrate reduction in simulated microniches by a denitrifying marine bacterium. Can J Microbiol 33:276–279. https://doi.org/10.1139/m87-047
Bonin P, Gilewicz M, Bertrand JC (1987b) Denitrification by a marine bacterium Pseudomonas nautica strain 617. Ann Inst Pasteur Microbiol. https://doi.org/10.1016/0769-2609(87)90125-6
Bonomi F, Iametti S, Mazzini S et al (2000) Thermal stability of Clostridium pasteurianum rubredoxin: deconvoluting the contributions of the metal site and the protein. Protein Sci 9:2413–2426. https://doi.org/10.1110/ps.9.12.2413
Cavaenero S, Zhou ZH, Adams MWW, Chan SI (1998) Unfolding mechanism of rubredoxin from Pyrococcus furiosus. Biochemistry 37:3377–3385. https://doi.org/10.1021/bi9721804
Cooper SJ, Garner CD, Hagen WR et al (2000) Hybrid-cluster protein (HCP) from Desulfovibrio vulgaris (Hildenborough) at 1.6 Å resolution. Biochemistry 39:15044–15054. https://doi.org/10.1021/bi001483m
Debrunner P, Schulz C (1976) Mössbauer parameters of rubredoxin, a one-iron–sulfur protein. Mössbauer effect methodology. Springer US, Boston, pp 155–167
Eidsness MK, O’Dell SE, Kurtz DM et al (1992) Expression of a synthetic gene coding for the amino acid sequence of Clostridium pasteurianum rubredoxin. Protein Eng Des Sel 5:367–371. https://doi.org/10.1093/protein/5.4.367
Fairbanks BD, Schwartz MP, Bowman CN, Anseth KS (2009) Photoinitiated polymerization of PEG-diacrylate with lithium phenyl-2,4,6-trimethylbenzoylphosphinate: polymerization rate and cytocompatibility. Biomaterials 30:6702–6707. https://doi.org/10.1016/j.biomaterials.2009.08.055
Folgosa F, Cordas CM, Santos JA et al (2011) New spectroscopic and electrochemical insights on a class I superoxide reductase: evidence for an intramolecular electron-transfer pathway. Biochem J 438:485–494. https://doi.org/10.1042/BJ20110836
Gauthier MJ, Lafay B, Christen R et al (1992) Marinobacter hydrocarbonoclasticus gen. nov., sp. nov., a new, extremely halotolerant, hydrocarbon-degrading marine bacterium. Int J Syst Bacteriol 42:568–576. https://doi.org/10.1099/00207713-42-4-568
Goodsell DS (2011) PDB Pioneers. RCSB Protein Data Bank. https://doi.org/10.2210/rcsb_pdb/mom_2011_10
Henehan CJ, Pountney DL, Vašák M, Zerbe O (1993) Identification of cysteine ligands in metalloproteins using optical and NMR spectroscopy: cadmium-substituted rubredoxin as a model [Cd(CysS) 4 ] 2- center. Protein Sci 2:1756–1764. https://doi.org/10.1002/pro.5560021019
Henriques BJ, Saraiva LM, Gomes CM (2005) Probing the mechanism of rubredoxin thermal unfolding in the absence of salt bridges by temperature jump experiments. Biochem Biophys Res Commun 333:839–844. https://doi.org/10.1016/j.bbrc.2005.06.004
Johnson DC, Dean DR, Smith AD, Johnson MK (2005) Structure, function, and formation of biological iron–sulfur clusters. Annu Rev Biochem 74:247–281. https://doi.org/10.1146/annurev.biochem.74.082803.133518
Kabsch W, Sander C (1983) Dictionary of protein secondary structure: pattern recognition of hydrogen-bonded and geometrical features. Biopolymers 75:413–429. https://doi.org/10.1002/bip.360221211
Kowal AT, Zambrano IC, Moura I et al (1988) Electronic and magnetic properties of nickel-substituted rubredoxin: a variable-temperature magnetic circular dichroism study. Inorg Chem 27:1162–1166. https://doi.org/10.1021/ic00280a015
Lees JG, Miles AJ, Wien F, Wallace BA (2006) A reference database for circular dichroism spectroscopy covering fold and secondary structure space. Bioinformatics 22:1955–1962. https://doi.org/10.1093/bioinformatics/btl327
Li Y, Pan Liu P, Ni X (2019) Molecular evolution and functional analysis of rubredoxin-like proteins in plants. Biomed Res Int. https://doi.org/10.1155/2019/2932585
Lode ET, Coon MJ (1971) Enzymatic omega-oxidation. V. Forms of Pseudomonas oleovorans rubredoxin containing one or two iron atoms: structure and function in omega-hydroxylation. J Biol Chem 246:791–802
Lubitz W, Ogata H, Ru O, Reijerse E (2014) Hydrogenases. Chem Rev 114:4081–4148. https://doi.org/10.1021/cr4005814
May SW, Kuo J-Y (1978) Preparation and properties of cobalt(II) rubredoxin. Biochemistry 17:3333–3338. https://doi.org/10.1021/bi00609a025
Merrouch M, Benvenuti M, Lorenzi M et al (2018) Maturation of the [Ni–4Fe–4S] active site of carbon monoxide dehydrogenases. JBIC J Biol Inorg Chem 23:613–620. https://doi.org/10.1007/s00775-018-1541-0
Meyer J, Moulis J-M (2006) Rubredoxin. Handbook of metalloproteins. Wiley, Chichester, pp 1–13
Moura I, Teixeira M, Moura JJG, LeGall J (1991) Spectroscopic studies of cobalt and nickel substituted rubredoxin and desulforedoxin. J Inorg Biochem 44:127–139. https://doi.org/10.1016/0162-0134(91)84025-5
Petillot Y, Forest E, Mathieu I et al (1993) Analysis, by electrospray ionization mass spectrometry, of several forms of Clostridium pasteurianum rubredoxin. Biochem J 296:657–661. https://doi.org/10.1042/bj2960657
Prakash S, Sundd M, Guptasarma P (2014) The key to the extraordinary thermal stability of P. furiosus holo-rubredoxin: Iron binding-guided packing of a core aromatic cluster responsible for high kinetic stability of the native structure. PLoS ONE. https://doi.org/10.1371/journal.pone.0089703
Rees DC, Akif Tezcan F, Haynes CA et al (2005) Structural basis of biological nitrogen fixation. Philos Trans R Soc A Math Phys Eng Sci 363:971–984. https://doi.org/10.1098/rsta.2004.1539
Robert X, Gouet P (2014) Deciphering key features in protein structures with the new ENDscript server. Nucleic Acids Res 42:320–324. https://doi.org/10.1093/nar/gku316
Schweimer K, Hoffmann S, Rösch P et al (2000) Solution structure of a zinc substituted eukaryotic rubredoxin from the cryptomonad alga Guillardia theta. Protein Sci 9:1474–1486. https://doi.org/10.1110/ps.9.8.1474
Sievers F, Wilm A, Dineen D et al (2011) Fast, scalable generation of high-quality protein multiple sequence alignments using Clustal Omega. Mol Syst Biol 7:539. https://doi.org/10.1038/msb.2011.75
Sreerama N, Woody RW (2000) Estimation of protein secondary structure from circular dichroism spectra: comparison of CONTIN, SELCON, and CDSSTR methods with an expanded reference set. Anal Biochem 287:252–260. https://doi.org/10.1006/abio.2000.4880
Tavares P, Pereira AS, Krebs C et al (1998) Spectroscopic characterization of a novel tetranuclear Fe cluster in an iron–sulfur protein isolated from Desulfovibrio desulfuricans. Biochemistry 37:2830–2842. https://doi.org/10.1021/bi9723008
Thapper A, Rizzi AC, Brondino CD et al (2013) Copper-substituted forms of the wild type and C42A variant of rubredoxin. J Inorg Biochem 127:232–237. https://doi.org/10.1016/j.jinorgbio.2013.06.003
Vrajmasu VV, Bominaar EL, Meyer J, Münck E (2002) Mössbauer study of reduced rubredoxin as purified and in whole cells. Structural correlation analysis of spin Hamiltonian parameters. Inorg Chem 41:6358–6371. https://doi.org/10.1021/ic020508y
Waerenborgh JC, Tavares P, Pereira AS (2019) Mössbauer spectroscopy. Principles of nuclear magnetic resonance and selected biological applications. Springer, Cham, pp 213–244
Whitmore L, Wallace BA (2004) DICHROWEB, an online server for protein secondary structure analyses from circular dichroism spectroscopic data. Nucleic Acids Res 32:668–673. https://doi.org/10.1093/nar/gkh371
Whitmore L, Wallace BA (2008) Protein secondary structure analyses from circular dichroism spectroscopy: methods and reference databases. Biopolymers 89:392–400. https://doi.org/10.1002/bip.20853
Xiao Z, Lavery MJ, Ayhan M et al (1998) The rubredoxin from Clostridium pasteurianum: mutation of the iron cysteinyl ligands to serine. Crystal and molecular structures of oxidized and dithionite-treated forms of the Cys42Ser mutant. J Am Chem Soc 120:4135–4150. https://doi.org/10.1021/ja973162c
Zheng P, Wang Y, Li H (2014) Reversible unfolding-refolding of rubredoxin: a single-molecule force spectroscopy study. Angew Chem Int Ed 53:14060–14063. https://doi.org/10.1002/anie.201408105
Acknowledgements
This research was supported by Fundação para a Ciência e Tecnologia, Ministério da Ciência, Tecnologia e Ensino Superior (FCT-MCTES), grant PTDC/BIA-PRO/111485/2009 (to P.T.), PTDC/QUI/64248/2006 (to A.S.P). This work was also supported by Radiation Biology and Biophysics Doctoral Training Programme (RaBBiT, PD/00193/2012, Applied Molecular Biosciences Unit – UCIBIO (UIDB/04378/2020), CEFITEC (UIDB/00068/2020) and C2TN (UID/Multi/04349/2019), all financed by national funds from FCT-MCTES). A.V.A. (PD/BD/135477/2017), J.P.J. (SFRH/BD/135056/2017), and J.P.L.G. (PD/BD/135476/2017) are supported by the RaBBiT programme. This work benefited from STSM funding by COST Action (CA15126 MOBIEU) and by the project CALIPSOplus under the Grant Agreement 730872 from the EU Framework Programme for Research and Innovation HORIZON 2020.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Special Issue: COST Action CA15126, MOBIEU: between atom and cell.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Almeida, A.V., Jacinto, J.P., Guerra, J.P.L. et al. Structural features and stability of apo- and holo-forms of a simple iron–sulfur protein. Eur Biophys J 50, 561–570 (2021). https://doi.org/10.1007/s00249-021-01546-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00249-021-01546-0